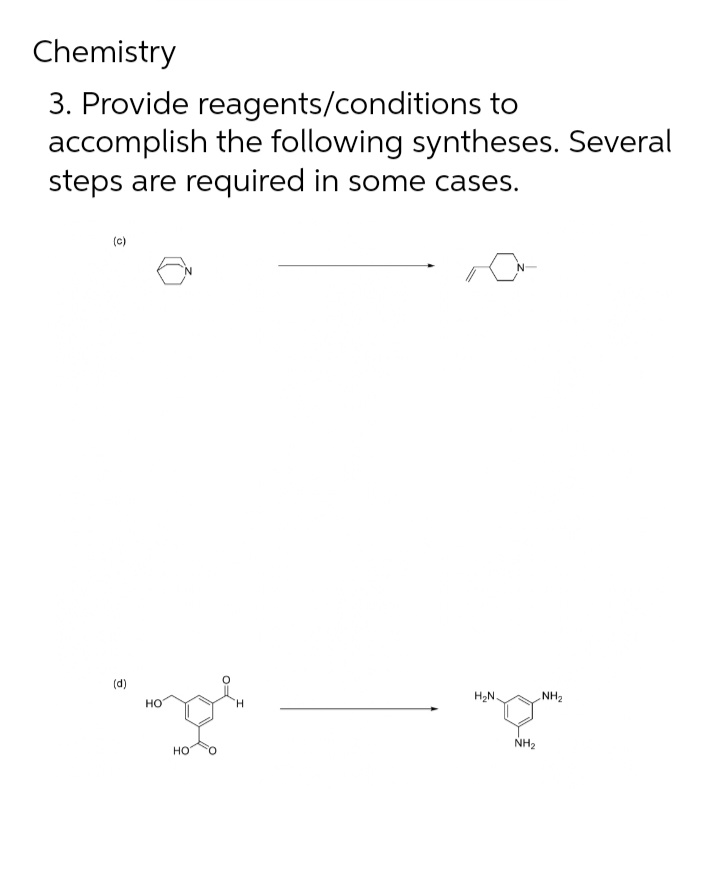
Chemistry 3. Provide reagents/conditions to accomplish the following syntheses. Several steps are required in some cases. (c) (d) H2N. NH2 но H. NH2 но
Q: To prepare the buffer, a total volume of 500 mL is required. The stock solution of the weak base is…
A: Answer: Buffer solution is the type of solution that resists the change in its pH on adding small…
Q: Evan H 3 Wing Consider the following unbalanced chemical equation: (501) Cu2 S(s) + O2(9) → Cu, O(s)…
A: The balanced equation is 2 Cu2S + 3O2 ---> 2 Cu2O + 2 SO2. Then we calculate the moles of…
Q: If the absolute entropy for O2 (g) is 126 J/(mole∙K), what is the ΔS for the complete combustion of…
A: C2H4 (g) on complete combustion gives CO2(g) and H2O(g) We need a balanced equation for the…
Q: Mass of AgNO3 used in reaction (this is the mass of just the AgNO3, without the vial) Mass of copper…
A: Values can be calculated using given data .
Q: Why would Hinsberg's Method for Characterizing Primary, Secondary, and Tertiary Amines fail with the…
A:
Q: -H
A:
Q: 1. Melting point technique
A:
Q: Compare and contrast the positive and negative effects that result from the use of the Haber…
A: The Haber-Bosch process has countless pros It supports the agriculture industry (fertilizers) It…
Q: Rank these compounds according to boiling point: a) heptanoic acid b) 1-octanol C) nonane O a)…
A:
Q: Calculate [OH⁻] and [H₃O⁺] in each solution and determine whether the solution is acidic, basic, or…
A: Autoionization of water: H2O(l) <.....> H3O+(aq) + OH-(aq) Water dissociation constant: KW =…
Q: Question 20 Consider the diagrams below. Rxn Progress Rxn Progress Rxn Progress II • Which diagram…
A: To solve this problem we have to know about the given chemical reactions diagram
Q: Describe the Bohr model of the atom, including quantization and emission spectra. (b) Discuss the…
A: SUCCESSES OF BOHR MODEL = Bohr theory modified the atomic structure model by explaining that…
Q: The activation energy for a particular reaction is 102 kJ/mol. If the rate constant is 1.35 × 10“ s1…
A: Given, Activation energy, Ea = 102 kJ/mol = 102x103 J/mol…
Q: EXPERIMENT 1 [A] 0.10 [B] 0.10 INITIAL RATE 2.45X10-3 2 0.10 0.20 9.80 X10-3 3. 0.20 0.10 4.90 X10-3…
A: Here we are required to find the rate law expression for the given reaction.
Q: 1. Describe the difference between the Rutherford model of the atom and the Bohr- Rutherford model…
A: There are different type of models for explaining the atomic model of an atom. Some of them are…
Q: Determine the concentration in molarity of a KOH solution if 21.6 g of KOH are dissolved in water to…
A:
Q: Complete Structure # of Carbons Shortcut Alkanes Structure Cycloalkanes (If applicable) 1 2 5 6 7 8…
A: 1- methane 2- enthane 3- propane 4-butane 5 - pentane 6- hexane 7- heptane 8- octane
Q: 0.700 g of an impure FeCl3 sample are dissolved in water to create 50.0 mL of solution. It took 1.54…
A:
Q: 8. Which choice best represents the relationship between wavelength and frequency? when frequency…
A: The correct answer is given below
Q: Which is the best set of reagents to carry out this transformation? CH3 H;C CH3 HC О 1. ВНЗ 2.…
A: In this question we have to complete the reaction by providing the good reagent.
Q: 9. Which of the following is not a disadvantage of nuclear fusion reactions? The sun produces a…
A: The correct answer is given below
Q: Use the correct Significant figures and Solve the following problems How many Liters…
A: The molarity of the solution is defined as the ratio of no of moles of solute to that of volume of…
Q: 16. The following data relate to the reaction A +B C. Find the order with respect to each reactant.…
A: The power of that reactants in the rate law equation is the order of that reactant.
Q: Which of the following does not describe beta particles? A. Beta particles can be stopped by a sheet…
A: Beta particle (β) is a type of high speed radiation with a negative charge.
Q: 4. Calculate E°cell and indicate whether the overall reaction shown is spontaneous or…
A: If E°cell is positive The reaction occurs spontaneously and if it comes negative it become non…
Q: A saline solution (NaCl solution) of 0.61 mg in 100 mL of solution is used in the formulation of…
A: Given, Mass of Saline (NaCl solution) = 0.61 mg…
Q: Complete Structure Shortcut Alkynes Cycloalkynes (if applicable) Structure Ethyne Propyne Butyne…
A:
Q: 16. Most nuclear reactions are controlled by substances that are good at catching neutrons. Which…
A: Control rods are used in nuclear decay process to control the neutrons Which can lead to chain…
Q: You discover a new substance and wish to determine its specific heat capacity. Your sample weighs…
A:
Q: At a certain temperature the reaction: CO(g) + Cl2(g) ⇌ COCl2 (g) Determine the Kc given the [CO]0…
A: The Kc is the equilibrium constant which is defined as the ratio of the concentration of the…
Q: 1/INO,(8)] (1/M) 5. You studied the chemical reaction, 2NO2(g) → 2NO(g) + O2(g), at 25°C by…
A: Rate constant, is a proportionality constant in an equation which depicts the relationship between…
Q: Name this according to the IUPAC system. NH CH3 CH3
A:
Q: (a) Give starting materials (including molar ratios) for synthesis of compound Z. Ph EIO2C OEt F3C…
A: This is an example of Hantz pyridine synthesis.
Q: Example 2: Carbon dioxide (2.5 mol) reacts with lithium hydroxide (3.75 mol) to form lithium…
A:
Q: HA, a weak acid with a Ka of 4.0x10, also forms the ion HA,. The reaction is :- НА (aq) +A (aq) HA2…
A: Solutions
Q: a. Given the following set of values for a gas, calculate the unknown quantity: P = 746.6 mm Hg V =…
A:
Q: Rank these hydrogens as to acidity. H;C HO. нн A C A most, C least O C most, B least O A most, B…
A:
Q: Take the reaction: NH3 + O2 ---> NO + H2O. If 3.25 g of NH3 are reacted with excess oxygen, what is…
A:
Q: Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of…
A:
Q: CAHOBR compound Which C,H, Br compound would give the following proton NMR spectrum? Ehlargements of…
A:
Q: Draw the organic products formed in each reaction.
A: In this question we have to tell the organic product of the reaction.
Q: For items 4-6, consider a one component system with a temperature-pressure phase diagram as shown…
A: Phase diagram is a graphical representation of phases that a substance can have at a particular…
Q: 34. Propose a mechanism for the following reaction: H + H20 CH, CH2CH2CH2 он
A: In this question, we will draw the mechanism steps for the formation of cyclic ether from the given…
Q: For items 10-11, the following solutions were prenared with their respective concentration: I. 0.02…
A: When solute is added to solvent , then the solution would have higher boiling point and lower…
Q: What is the pH of a solution that contains 60.0 mL of 0.100 M HCl?
A:
Q: Which salt is a strong electrolyte? O iron (III) phosphate ammonium sulfate O calcium carbonate…
A: To solve this problem we have to know about electrolyte . Electrolyte are the substance which…
Q: The theoretical yield of a product in a reaction is determined by the limiting reactant. O True…
A: 1.Theoritical yeild is the amount of product fromed from given balance equation . 2.While actual…
Q: 4.0050 g sample of chlorinated lime was mixed with enough water to make 1000 mL. A 100 mL sx of the…
A: Find the availble chlorine content of chloronated lime
Q: To do: Identify the oxidizing agent and reducing agent in the reactions. 1.…
A: Oxidising agent oxidizes the other species but itself gets reduced. reducing agent reduces the…
Q: Using 1.0x106 M as the quantitative precipitation criterion, if silver ions are added to a solution…
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- The substitution reaction below forms two ethers (which are consitutional isomers). Which ether is the major product and why?Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Alcohols can undergo a lot of different reaction mechanims. If the alcohol group (OH) is attaached to an aromatic core, how will the chemistry change as compared to a typical alkyl alcohol? A) The OH group will become more polarised and more nucleophilic. B) The OH group will become more susceptible to oxidation C) The OH group will become more polarised and therefore basic D) The OH group will become more polarised and therefore acidic.
- best condition for the reaction (1r,2s)-1 bromo-2methylcyclohexane->(s)3-methylcyclohex-1eneTunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):Provide the product/starting material/reaction conditions for each reaction below.

