as ato 9. Give an IUPAC name for each of the following hydrocarbons with the benzene ring as a substituent. CO Bran at is retu chase. I to abus AM S common acils, bal ghlighte ablish p zinforce rands 00 2014, Rese info acco 18 Page 7 of 7
as ato 9. Give an IUPAC name for each of the following hydrocarbons with the benzene ring as a substituent. CO Bran at is retu chase. I to abus AM S common acils, bal ghlighte ablish p zinforce rands 00 2014, Rese info acco 18 Page 7 of 7
Chapter12: Unsaturated Hydrocarbons
Section: Chapter Questions
Problem 12.57E
Related questions
Question
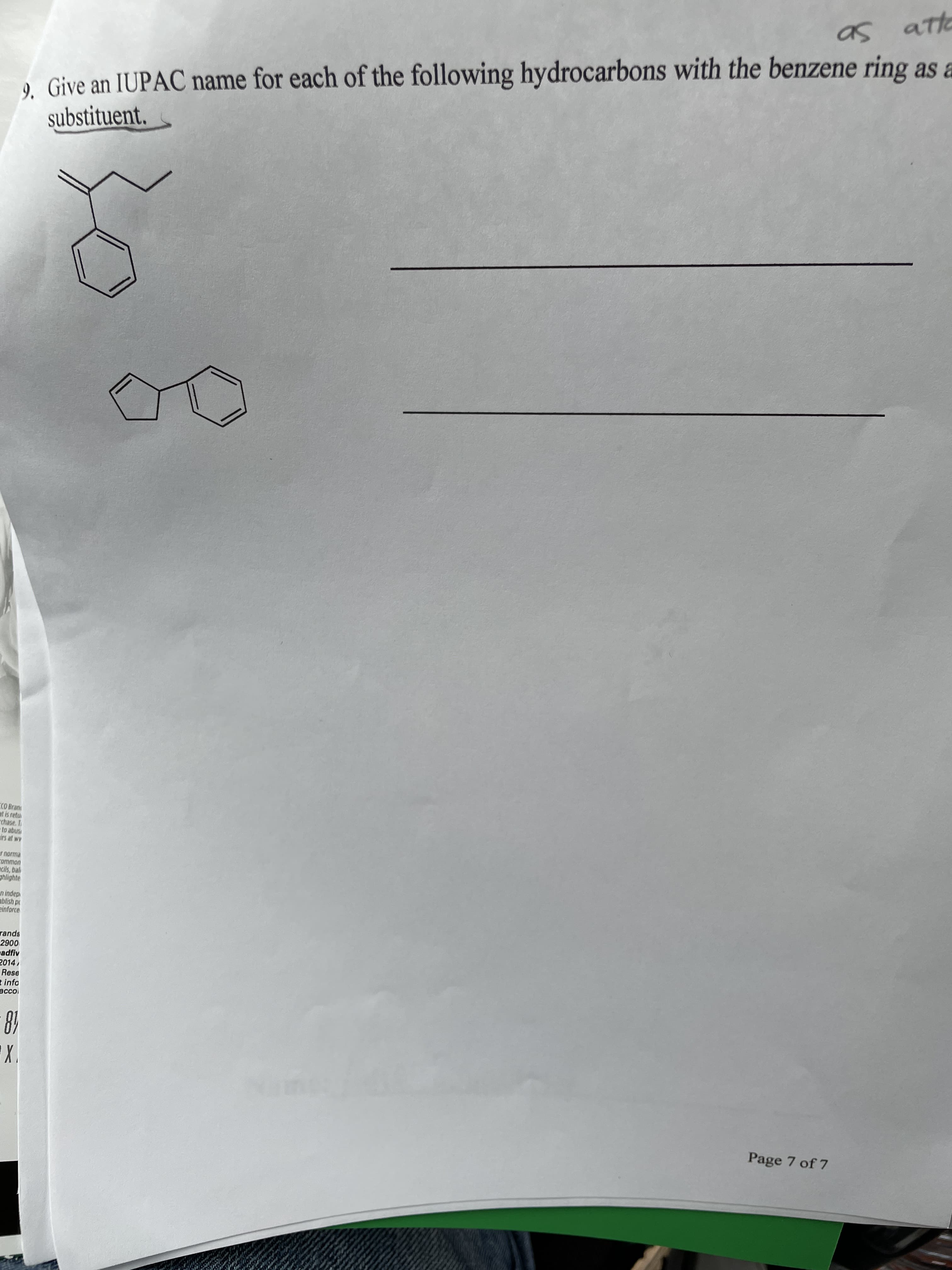
Transcribed Image Text:as ato
9. Give an IUPAC name for each of the following hydrocarbons with the benzene ring as a
substituent.
CO Bran
at is retu
chase. I
to abus
AM S
common
acils, bal
ghlighte
ablish p
zinforce
rands
00
2014,
Rese
info
acco
18
Page 7 of 7
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning