Q: A student constructs the following galvanic cell using a electrode in 1.0 M Zn(NO3)2, a silver…
A: Galvanic cell: Zn electrode dipped in 1 M Zn(NO3)2 solution Silver electrode dipped in 1 M AgNO3…
Q: What is the S for the reaction? SO3(g) + H2O(1) –> H2SO4(1) 69.9 Given: S°(J/K mol): 256.2 156.9 O +…
A: The balance chemical reaction : SO3(g) + H2O(l) ---> H2SO4(l) S°(J/k.mol):…
Q: E2 Nal acetone Br OH- Br water, heat
A: Elimination reactions are the reactions which proceeds with two types one is E1 and other is E2. E1…
Q: -Cf3 Nucleophilic Attack
A:
Q: 28. Calculate the velocity of an enzyme- catalyzed reaction A+B P+Q at [A]= ex 10 M and [B]= 6.7x 10…
A: Solutions- Data given- [A] = 2.0 x10-3 M [B] = 6.7 x 10-3 M KA = 2.2 x 10-4 M KB = 1.9 x 10-5 M and…
Q: The reaction C2H5Cl →C2H4 + HCl is first order in C2H5Cl. The rate constant is 1.60 x 10-6/s. In an…
A:
Q: Calculate the concentration of an anthracene solution which produced a fluorescence intensity (I) of…
A: Given: Fluorescence intensity (I) = 815 Irradiance of beam (P0) = 1571 Length of medium (b) = 0.868…
Q: Learning 1ask No. 3 CODH methylene blue methyl red Na+ CH3 CI- 11 CH3 methyl orange Crystal viotet
A: The given molecules contain number of heteroatoms, which results into formation of several…
Q: 8. 20.0g of sodium chloride is reacted with 32.0g of fluorine. What mass of sodium fluoride results?
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly…
Q: The specific heat of a certain type of cooking oil is 1.75 J/(g-°C). How much heat energy is needed…
A: Specific heat capacity is amount of heat needed to increase the temperature of one gram of substance…
Q: Draw all possible ways by which the following two molecules can form hydrogen bonds with each other:…
A: The molecules given are HF and .
Q: 1. Write the balanced molecular equation for the reaction of the CuSO4 with ammonia to form…
A: Dear student, since you have asked multiple questions but according to guidelines we will solve 1st…
Q: How would you prepare the following substances from 1-butanol? Butylamine N,N-dimethylbutylamine…
A:
Q: Propose starting materials for the following azo dyes
A: We have to predict the starting material for azo dyes.
Q: The complete reaction of an alkyl halide towards alcoholic silver nitrate solution results to a…
A:
Q: How does boiling point or freezing point affect the dissolution of a solid in water and its…
A: Boiling point is the temperature at which the compound starts boiling and its liquid phase converts…
Q: HW: Hess' Law Ericka Rivera · Apr 20 (Edited Apr 20) 20 points Find the AH of this reaction: C(s) +…
A:
Q: REDOX TITRIMETRY I. Balancing Redox Equations Use the Half-reaction method: a) Write the…
A:
Q: For each structure on the left, pick an identification on the right. Clear All H2N-ÇH- -CH- -OH CH2…
A:
Q: Identify the species oxidized, the species reduced, the oxidizing agent and the reducing agent in…
A: The given electron transfer reaction is: 2Cr3++Mg→2Cr2++Mg2+
Q: The rate of the reaction ; NO2 + CO ---------> NO + CO2 depends only on the concentration of…
A: As the rate is depends only on the concentration of nitrogen dioxide below 225C means the reaction…
Q: Convert each of the following. Provide your reasoning. 19) 64 cubic centimeters =…
A:
Q: Stiochiometry question: Please show all steps. How many molecules of H2O would be produced by 1.40…
A:
Q: (Give clear handwritten detailed solution) Find out oxidising agent from the mentioned reaction.
A:
Q: Compare and contrast the following general methods of argentometric titration. What are the…
A: In the given problem we need to discuss the comparison between Mohr, Volhard and Fajans method and…
Q: Chlorofluorohydrocarbons of low molecular weight are called Freons. Draw the structures of the given…
A: Trichlorofluoromethane : Methane is having once carbon. Three chlorines and one fluorine is attached…
Q: You have 100ml of a bacterial culture. After diluting your culture 1x10^5 times you counted 42 cfu.…
A: Given that, a 100 mL of bacterial culture. It was diluted by 1×105 times. So, the volume of the…
Q: Q1) Which of the following statements regarding collision and transition state theories b)…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the Keq expression for the given reaction: 2M9C12(s) → 2Mg(s) + Cl2(g) A. Keq = [MgCl2]? B.…
A: Equilibrium constant = concentration of products / concentration of starting materials. Solids,…
Q: I need to get the rf valuse from a made up value and draw a tlc plate of 30% ethyl actate and 70%…
A: TLC can easily tell whether a reaction is complete or not. On a TLC plate, a polar compound takes…
Q: The rate of the reaction ; NO2 + CO ---------> NO + CO2 depends only on the concentration of…
A: We must find out order of reaction w.r.t [NO] to solve this problem.
Q: 3. What type of bond do you have with the people around you?
A: Mainly there are four types of bonds or interactions. They are (1) ionic (2) covalent (3) hydrogen…
Q: 5. Provide an adequate route (reaction scheme) for the following reaction
A:
Q: Molecules with the composition ROH is a(n) ________ group. carbonyl hydroxyl amino
A: composition of given molecule is ROH
Q: Which of the properties below apply to the following polymer? -CH2- -CH2 -CH CH2- Choose all that…
A: Polymerization is formed by the addition of monomer unit, condensation of monomer unit.
Q: QUESTION 3: Write the following half reaction per 1 electron. 1) Cr(III) to Cr(II) 2) Zero valent Fe…
A: Here we have to predict the half reaction in which one elctron transfer occur.
Q: One potential uranium fission reaction is shown below. 235U + n- 140BA + 93Kr + 3 n [Atomic and…
A:
Q: Incorrect Mechanism (The mechanism is for the formation of the 1,4 product. Please ignore the 1,2…
A: Note : 1,3-butadiene reacts with proton produces the allylic carbocation. Allylic carbocation…
Q: How can a substance, like water, show its amphiprotic nature? What information can be derived from…
A:
Q: Solid ammonium phosphate is slowly added to 50.0 mL of a silver acetate solution until the…
A: Given, Solid (NH4)3PO4 is slowly added to 50.0 mL of a CH3COOAg solution until the concentration of…
Q: Use this to answer Qs 24-29.) Bill Nye the Science Guy is at it again and wants to show the chemistı…
A:
Q: The next phase diagram looks at the effect of changing pressure at constant temperature. Name and…
A: Here phase diagram given I have explained below with details about critical point.
Q: Show a stoichiometric solution on how glucose will be converted to ethanol.
A:
Q: When the following skeletal equation is balanced under acidic conditions, what are the coefficients…
A: The given redox reaction is: Hg+Sn2+→Hg2++Sn
Q: Complete the following data Standardization of NaOH Trial 1 Trial 2 Trial 3 Trial 4 Mass KHP, g…
A: Here we are required to find the normality of standardized NaOH in reaction 1 and then normality of…
Q: How many equivalents of Mg*2 are there in 6.00 mol of Mg3(PO4)2? First we have to convert moles into…
A: Initially we have to determine the number of equivalent of Mg2+ in the corresponding phosphate of 1…
Q: CH2 -CH2 -0- -(CH)4 One or more of the monomers below are needed to make the polymer Choose which…
A:
Q: Depict H-bonding between the molecule CH3OCH3 and water
A: The given molecule is acetone and posses an electronegative oxygen atom, hence due to this reason,…
Q: The decomposition of hydrogen peroxide in dilute sodium hydroxide at 20 °C H,0,(aq) H,0(1) + ½ 02(g)…
A:
Q: A metal object with mass of 21.4 g is heated to 97.0 °C and then transferred to an insulated…
A:
N f.
IUPAC name.
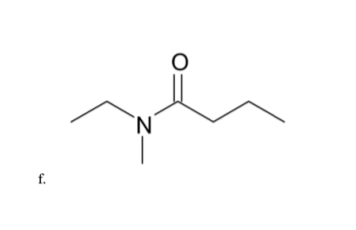
Step by step
Solved in 2 steps with 1 images


