Q: Each row of the table below describes an aqueous solution at about 25 °C. [1,0]0 to 2 significant…
A: We have given pH of three solutions. We need to find the concentration of hydronium ion. Using…
Q: A reaction that is second-order in reactant A has (A]o = 0.200 M. The half-life is 45.6 sec. What is…
A:
Q: Draw the expected 'H NMR spectrum for the following compound. Include in your answer the splitting…
A:
Q: Tonic water (20.0 mL) was added into each of two volumetric flasks (100 mL). The first flask was…
A:
Q: O ii. and iv. Question 18 How does the internal energy of the universe change during an endothermic…
A: Here we have to predict the correct statement regarding internal energy change of universe during an…
Q: The following Wittig reactions may produce a mixture of alkene stereoisomers. Write the products for…
A: Witting reaction is reaction of aldehyde with oxophosphetane, resulting the formation of alkene.
Q: • Explain Heisenberg Unte inby principie with deivahion aloy fully descaibe in word and more than…
A:
Q: Consider Na,[Co(CN)]. A coordinate covalent bond exists between: Select ong a. Со and CN 3+ o b. Na…
A:
Q: Specify the reaction conditions to favour the indicated elimination mechanism. a. Strong base, such…
A:
Q: 12. If a cube of ice at 0 °C is placed outside on a warm summer day, the ice will melt…
A: This question belong to thermochemistry. Spontaneous reaction occurs itself without applying any…
Q: What is the pH of a 2.5 M solution of HC1O4? pH
A:
Q: Explain Heisenbery Uncertainby principle with dei vahion aloy fully describe in and more than word…
A:
Q: How many mL of 0.625 M KBr are needed to prepare 2. O 200. mL O 0.0200 mL O 313 mL O 31.3 mL
A: Intial concentration (M1) = 0.625 M Initial Volume (V1) = Final concentration (M2) = 0.050 M Final…
Q: An optically inactive amino acid is a.glycine b.valine c.serine
A: Which one of the following is correct answer
Q: CH,CH,CI (5) ? AICI,
A:
Q: Carbonyls make the adjacent alkyl groups more acidic. However, the presence of electron withdrawing…
A: A question based on general chemistry that is to be accomplished.
Q: De Broglie Hypothesis (Dual Narure of matter And Radi ohou) Ex plavin weu aloug with detailed Ex…
A:
Q: According to MO theory, which of the following lists ranks the nitrogen species in terms on…
A:
Q: Determine the [H*], pH, and pOH of a solution with an [OH-] of 3.3 × 10-12 M at 25 °C. [H*] = M pH =…
A: if the pH is less than 7, then the solution is acidic. if the pH is more than 7, then the solution…
Q: Calculate the PH of a solution of 0.33 M HF solution The ka for HF is 7.2x 10^-4.
A:
Q: Main eondi non which meari fuf must satispa to solution c Eigen funetion) Detailed. Puysical…
A: ψ ( wave function) is not a physically observed quantity but using that we can calculate other…
Q: Given the standard reduction potentials of Sn and Cu: Sn2+ + 2e- Sn Eo = - 0.14 V 2 Cu2+ + 2e- 2…
A:
Q: the correct name for CuSO4·5H2O is could you explaining to me how to read and understand the…
A:
Q: H HHH н-с-с-с-с-н i iii H H H H 1. Consider the following chemical reaction for the combustion of…
A:
Q: Question 25 Consider the following electron configurations to answer the question that follows: 1.…
A: We have to predict which of the given options will have highest second ionisation energy
Q: De Broglie Hypothesis ( Dual Nahute of matter And Radi ohou) Explovn weu along with detailed Ex…
A:
Q: Chemistry IDENTIFY PRODUCT A TO C Br BugSnH, AIBN A Toluene, 80 °C Ph-
A:
Q: A solid metal object with a mass of 163 g is added to a graduated cylinder which originally contains…
A:
Q: Q6. Starting from the indicated starting material, provide eitber the product(s) or the reagents…
A: Here we have to write the major product formed in the following given elctrophilic aromatic…
Q: Component 1 on a GC column elutes before component 2. Which of the following statements is true?…
A: The true statement is - c) Component 1 has more affinity for the mobile phase than component 2.
Q: (Q77) The standard Gibbs free energy (AG°rxn) for the formation of sulfur trioxide from sulfur…
A:
Q: True or False. Support your answers For every process, AUsys = -AUsurr b. For every cyclic process,…
A: first law of thermodynamics
Q: In each row check off the boxes that apply to the highlighted reactant. The highlighted reactant…
A: Here we are required to identify the species as acid or Base.
Q: For a bond where the hybridization of the central atom is sp3d, it is true that: a) It can have…
A: For a bond where the hybridization of the central atom is sp3d it is true that:
Q: An analytical chemist is titrating 175.9 mL of a 0.1800M solution of methylamine (CH,NH,) with a…
A:
Q: Name the following compounds according to the IUPAC nomenclature and mark all asymmetric C atoms…
A:
Q: H >:0: H-C-H H-C H с н `N Н-с —н `z: I-Ó-I
A: Based on this image, classify each of the following statements as True or False. All of the…
Q: (1) mCРBA Fumeric acid product (2)H30+ (1) mCPBA फ्यूमेरिक अम्ल on. उत्पाद (2)H30+ СООН on HO (A)…
A: Organic chemistry is branch of chemistry in which organic reactant react to form organic products.
Q: What is the relationship of Ecell and Eocell if all of the concentrations are set to 1.0 M (and if…
A:
Q: Given that Ka for HC1O is 4.0 × 10-8 at 25 °C, what is the value of K, for ClO¯ at 25 °C? Kp = Given…
A: (1) Given that - Ka for HClO = 4.0×10-8 Temperature = 25°C Then, from following formula - Ka .…
Q: 2) How many grams of calcium chloride contains 4.08 x1024 ions of Cl?
A: Given that: Ions of Cl- = 4.08×1024 Formula of calcium chloride = CaCl2
Q: Below is the structural formula of altrose. To what carbohydrate class does altrose belong? HO но OH…
A:
Q: Fill in the name and empirical formula of each ionic compound that could be formed from the ions in…
A:
Q: When reacted, how does the use of potassium (K*) vs sodium (Na*) change the soap product? O Na* adds…
A:
Q: If you dissolve 50 g of NH4Cl at 30 0C, how much NH4Cl will be left undissolved or precipitate out…
A: At 30°C solubility of ammonium chloride is 41.4gm/100gm water.Therefore 8.6 gm of ammonium chloride…
Q: 2. The reaction: N2 (g) + O2 (g) 2 NO (g) with the enthalpy of reaction AH° = + 180.5 kJ was in…
A: The Le Chatelier's principle explains the movement of the equilibrium to gain another new…
Q: Explain Heisenberg Uncerlointy principle along with deivahion and fully describe in more than word…
A:
Q: Which of the following statements describes the amino acid presented below? OH CH2 O H3Ñ–CH-C-0 This…
A: The R group in an amino acid is its side chain. This side group make it hydrophilic if it can make…
Q: When non-symmetrical dienes react with non-symmetrical dienophiles, there are two possible…
A:
Q: The use of phosphates in commercial detergents is decreasing because ----. phosphates accelerate the…
A: Initially detergents were made using phosphate Compounds . But with the time it has been realised…
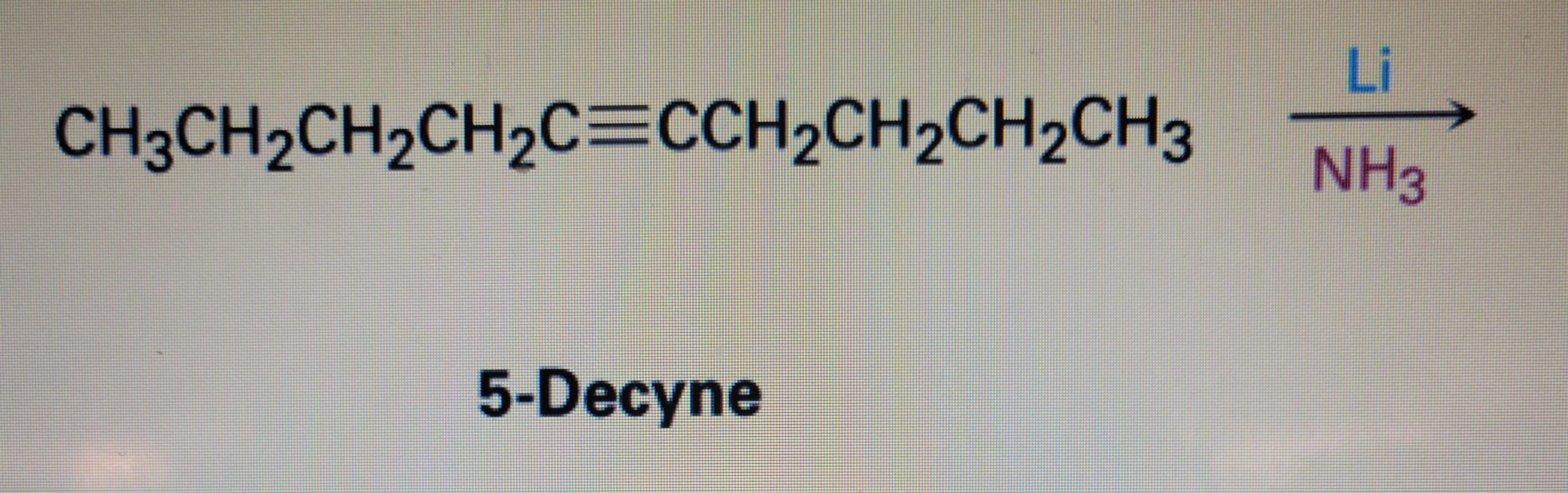
Step by step
Solved in 2 steps with 1 images


