6 Σ Att The rate constant for this first-order reaction is 0.450 s at 400 °C. A products How long, in seconds, would it take for the concentration of A to decrease from 0.800 M to 0.250 M? TOOLS O LX x10 9:32 PM 10/24/2021 F5 F7 F8 F10 F11 F12 PrtScr Insert Delete of 24 Backspace 5. Enter H B. Alt Ctrl పటరన
6 Σ Att The rate constant for this first-order reaction is 0.450 s at 400 °C. A products How long, in seconds, would it take for the concentration of A to decrease from 0.800 M to 0.250 M? TOOLS O LX x10 9:32 PM 10/24/2021 F5 F7 F8 F10 F11 F12 PrtScr Insert Delete of 24 Backspace 5. Enter H B. Alt Ctrl పటరన
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 94AE: For a first order gas phase reaction A products, k = 7.2 104s1 at 660. K and k = 1.7 102s1 at...
Related questions
Question
Transcribed Image Text:6
Σ
Att
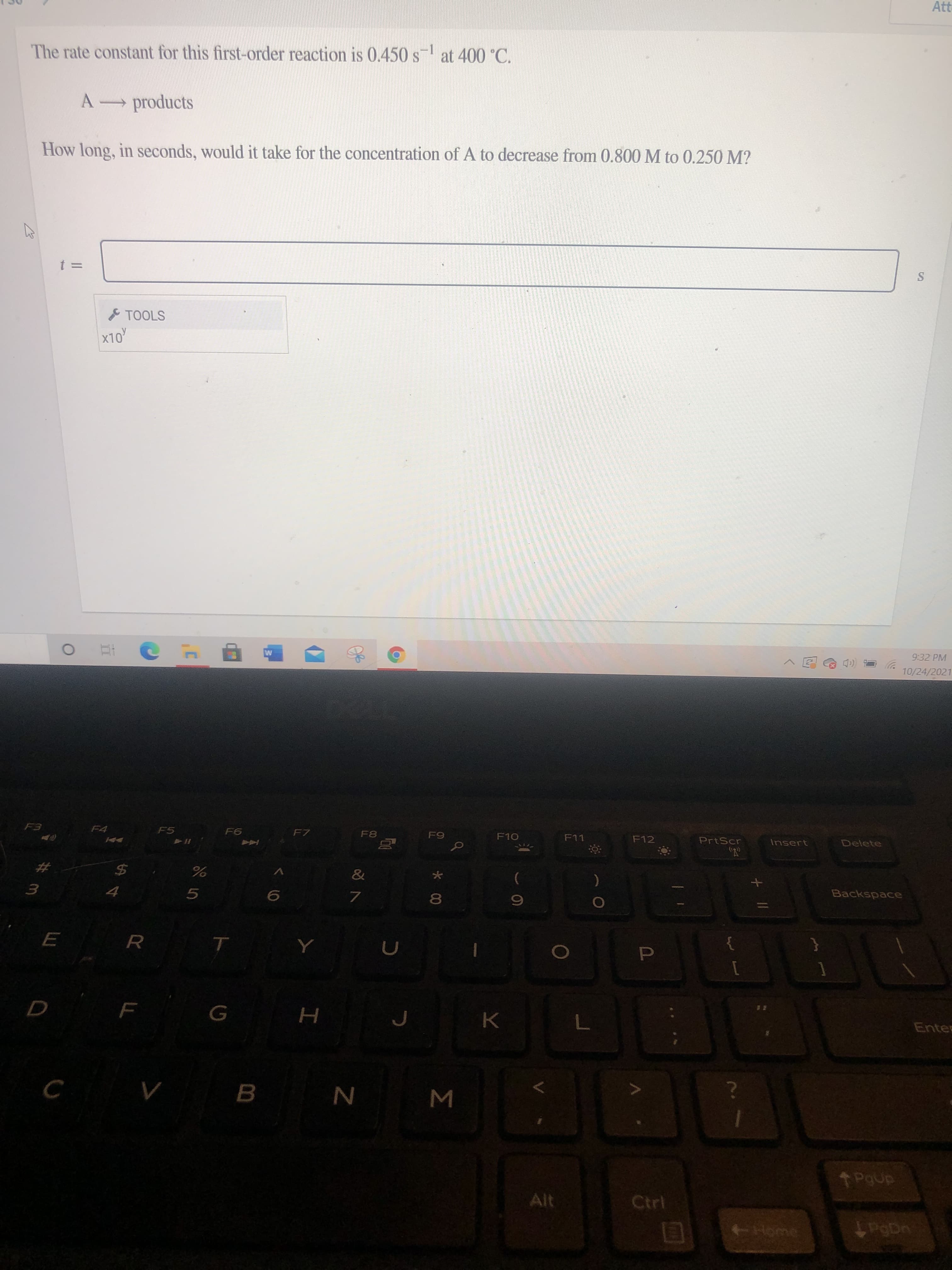
The rate constant for this first-order reaction is 0.450 s at 400 °C.
A products
How long, in seconds, would it take for the concentration of A to decrease from 0.800 M to 0.250 M?
TOOLS
O LX
x10
9:32 PM
10/24/2021
F5
F7
F8
F10
F11
F12
PrtScr
Insert
Delete
of
24
Backspace
5.
Enter
H
B.
Alt
Ctrl
పటరన
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax