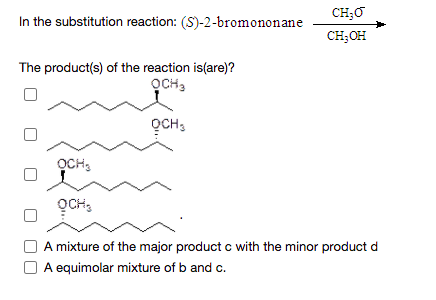
CH;O In the substitution reaction: (S)-2-bromononane CH;OH The product(s) of the reaction is(are)? OCH, OCH, OCH, OCH, A mixture of the major product c with the minor product d O A equimolar mixture of b and c.
Q: The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO-…
A: In bromination reaction, bromine atom is introduced to a compound. Hydrogenation can be done using…
Q: 4 (a) Compound D undergoes a reaction with hydrogen bromide, HBr to produce 2-bromobutane. D exists…
A: Given that Compound D gives 2 - bromobutane on Reaction with Hydrogen Bromide, thus compound D is…
Q: Br2 NaN3 b) IBr (CH3),S=O EtONa, ETOH reflux CH;OH d) cat. H,SO4 ICI e) CH;OH H
A: The major product for all the given reactions is shown below with their appropriate stereochemistry…
Q: Draw the structures for the intermediates and product (left to right) as represented by A to C in…
A: In this question we have to tell the product of the reaction sequence.
Q: Compare the two reactions shown below excess LICH,CH, excess LICH,CH3 Both electrophiles, an ester…
A: Given reaction is addition of organolithium on ester and ketone.
Q: What is (are) the major organic product(s) obtained from the following reaction? 1. OsO4 2. NaHSO3…
A:
Q: Give the major organic product(s) of the following. Include stereochemistry when applicable. 1) Nal…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 3. 'Please provide the products for the following reactions by writing the structures of the…
A: The above given reactions are pericyclic reactions or [2+4] cyclo addition. These reactions were…
Q: Draw the major organic products of the reactions, showing stereochemistry as needed. If no…
A: The details solution for this reaction is provided below in attach image.
Q: The stereochemistry of the products of reduction depends on the reagent used, as you learned in…
A: a.
Q: -CECH + 2Br, → H,C CH3
A:
Q: What is the major substitution product of this reaction? (A) (B) (C) (D) OH Br + Br ye Br + Br HBr…
A:
Q: 6. Complete the product(s) of the following substitution reactions. Include the stereochemistry.…
A: 6. a. OH group is converted into OTs using TsCl and py. b. 2-butanol is protonated first followed…
Q: 0 CH₂ → ?? If the above compound is subjected to combustion, what are the products? O ethanoic acid…
A: The above compound is classified as the ester. We know the reactions of ester ; Combustion :- when…
Q: 4. What are the products formed when each alkene below is treated first with MCPBA in CH2CI2 solvent…
A: Both Product formed in each step are Enantiomers because of the fact that OH- Always attack in anti…
Q: R*R?CHOH-CR®R* R'R?C=CR®R* uld orientation of each substituent in reactant to convert it into…
A: solution- reaction - R1R2CHOH -CR3R4 → R1R2C=CR3R4 assuming, R > R2, R3 >R4R1 and R2 we…
Q: In the reaction of ethylene with H2O in the presence of sulfuric acid,which one adds across the…
A: The reactants given are H2O and ethylene i.e CH2=CH2 in presence of catalyst sulfuric acid i.e…
Q: Determine the product(s) formed when cyclohexene is treated with the following reagents. Br HO. HO…
A: Dear student since it is a multiple type questions with multiple subparts so according to Guidlines…
Q: 1) NaBH4/ether 2) H2O КОСН3 in CH,OН TH. H;O+
A: The reaction mechanism is given below
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. NaOCH3 / CH,OH…
A: The hydrogen present in the alpha carbon of the carbonyl group is acidic due to the resonance…
Q: Q1/Complete the following reactions and mentioning CO₂CH3 NaOCH3 CH3OH + methylacetate O₂Et NaOEt…
A: A chemical reaction involves the reactant molecules reacting with each other either in the presence…
Q: Draw all of the substitution and elimination products formed from thegiven alkyl halide with each…
A: a.
Q: Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral? Is it optically active?…
A:
Q: 2) Draw and name the organic compound found in every reaction. d) Reaction of…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: 7В. (2' Draw the structure of the product, substrate or condition in the following reactions (should…
A: Alkene are unsaturated hydrocarbons. They have free electron density to give. thus it can easily…
Q: CF3 KOET ELOH, A SO,CH3
A:
Q: R'R°CHOH-CR'R* R'R?C=CR*R* What should orientation of each substituent in reactant to convert it…
A: The given reactant is alcohol. The reaction given above is a dehydrating reaction. Here the alcohols…
Q: Draw the structure(s) of the organic product(s) predicted when this compound reacts with NABH4,…
A: NaBH4 acts as a reducing agent in organic reactions. In presence of carbonyl groups, the BH4-…
Q: nost" und nder the one acetaldehyd e "most" under the one which t" under the one which would be le…
A:
Q: cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a…
A: The formation of a double bond between two carbon atoms by the elimination of halogen halide during…
Q: What would you expect to be the major product obtained from the following reaction? AICI, ? еxcess…
A: Given reaction is electrophilic substitution reaction . it is fridel craft`s alkylation reaction. we…
Q: Chemistry Draw the main product of the following reaction. Show stereochemistry in product structure…
A:
Q: Give the major organic product(s) or reagents needed. Show stereochemistry Where approp OH K,CrO7,…
A: In organic reaction we can convert one functional group into other carrying out certain reaction…
Q: HgCOAc)z, water O con. Hq SO4 O NBS, 120OR OH NGOH, A 3 cat. Osu, NMO
A: The reactions given are,
Q: Question 35 of 47 > Draw the product formed by the reaction of potassium 1-butoxide with…
A: In the presence of strong base elimination always prefer E2 elimination E2 elimination occurs as…
Q: Which is the most likely product for the reaction below: CH, CH,CH,CH-C CH,OH CH,
A:
Q: SOC2, py HO:
A: Alcohol reacts with thionyl chloride in the pyridine solvent to give alkyl chloride.
Q: 5. Provide the missing reactants or products for the following reactions. Show stereochemistry. NC.
A: Here we have to determine the reactant and product which are used and formed during cycloaddition…
Q: Draw all of the substitution and elimination products formed from the given alkyl halide with each…
A: The substitution and elimination products formed from the given alkyl halide with given reagent has…
Q: The SN2 reaction ur l-chloro-3-methylbutane with hydroxide OH is relatively slow, but it can be…
A:
Q: NaBH4, EtOH + ОН H H ОН exo is MAJOR product. endo is MINOR product.
A:
Q: What is the product of the following reaction sequence? You can ignore and stereochemistry. The…
A: In the first step the reactant is reacted with ethyl magnesium bromide and then with water to obtain…
Q: 2. faj - What are Gilman reagents?: How is it prepared in the laboratory? S%ggest the mechanism of…
A:
Q: When glucose is treated with NaBH4, reaction occurs to yield sorbitol, a polyalcohol commonly used…
A: Answer is explained below.
Q: When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed.…
A:
Q: Give two syntheses for (CH3)2CH¬O¬CH2CH3, and explain which synthesis is better
A: The given compound is ether. It can be synthesized by Williamson synthesis. The best yield is…
Q: Draw the product formed by the reaction of t‑butoxide with (1R,2S)‑1‑bromo‑2‑methyl‑1‑phenylbutane.…
A:
Q: sctio Supply the missing products, reagents or starting materials as necessary. NABD3CN 9. HOẠC а.…
A: See products and reagents in red boxes.
Q: Which of the following is/are the expected product/s of the reaction of 2-pentanone (CH3COCH2CH,CH3)…
A: Given :- CH3COCH2CH2CH3 + excess Br2 + OH- → To identify :- Products formed in above reaction
Step by step
Solved in 4 steps with 2 images

- M 6 write the principal product in a, c & e and the neccesary reactives for b, d & f in the following reactions :The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?Draw all of the substitution and elimination products formed from thegiven alkyl halide with each reagent: (a) CH3OH; (b) KOH. Indicate thestereochemistry around the stereogenic centers present in the products,as well as the mechanism by which each product is formed.
- cis-4-Bromocyclohexanol and trans-4-bromocyclohexanol form the same elimination product but a different substitution product when they reactwith HO-. a. Why do they form the same elimination product?b. Explain, by showing the mechanisms, why different substitution products are obtained.c. How many stereoisomers does each of the elimination and substitution reactions form?Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.
- What products would ypu obtain from reaction of 2,4-dimethyl-2-pentene with BH3, followed by H2O2, OH-? Show its complete reaction mechanism.Chemistry Q: Kindly explain the striking observation that methylation of the alcohol 1 leads to a methoxy compound in which the stereochemistry of the oxygen has changed. Explain it by providing a mechanism accounting for the transformation. (write legibly pls).There are two isomeric cyclohexa-1,4-diene products when benzoic acid undergoes the Birch reduction (see Problem 25.24). (a) Draw the mechanism that leads tothe formation of the major product. (b) Will the Birchreduction of benzoic acid occur faster or slower than theBirch reduction of benzene itself? Hint: Is –CO2H anelectron-donating or an electron-withdrawing group?
- Draw the organic product(s) of reaction of the alkyne above with H3O+ in the presence of HgSO4 (shown in two different angles)Reaction of 2° alcohol A with HCl forms three alkyl chlorides, all of whichresult from rearrangement of the 2° carbocation initially formed. Draw thestructures of these products and a mechanism that illustrates how eachis formed.

