Q: Explain the meaning of “the reaction goes to less than 100% completion”
A: In all the Reactions the Reactants are not converted to products completely .This Reactions always…
Q: b Dependent on a single reactant's concentration even if there are multipl
A: In Pseudo first order kinetics, molecularity of reaction is more than 1 but order of reaction is…
Q: Define Synthesis or Combination Reactions ?
A: In a chemical reaction, two or more chemical species react with each other and form the desired…
Q: О3, -78 Celcius
A: 1) The reaction with Ozone is basically the ozonolysis reaction in which the double bond is broken…
Q: 1. It is defined as the detailed description, step by step, of the same, which means knowing all the…
A: 1. It is defined as the detailed description, step by step, of the same, which means knowing all the…
Q: Decomposition AB
A: Decomposition means Broken down of substances into a simpler and more than one product .
Q: The process of adding one reactant to another while monitoring the completeness of the reaction is…
A: Titration is the slow addition of one solution of a known concentration where the reaction is…
Q: Classify the reaction: H₂SO4 + KOH -----> K₂SO4 + H₂O
A:
Q: Raising the temperature of a reacting system increases the rate of reaction but does not increase…
A: Rate of a reaction increases with rise in temperature.
Q: Which of the following changes leads to an increase in reaction rate? O A. Increase the product…
A: The reaction rate depends on the concentration of reactants.
Q: Classify each of these reactions.
A: Since you have posted a question with sub-parts , we are entitled to answer the first three…
Q: Look at the picture below. 1. What is a fruitful collision? 2. What is an unfruitful collision?
A: Collision Theory of bimolecular gaseous reaction aims at quantitative calculation of rate of a…
Q: Classify each of these reactions. A Ba(CIO,), → BaCl, + 30,
A:
Q: The presence of a catalyst: lowers activation energy increases activation energy O increases…
A:
Q: Which of the following is/are expected to affect the rate of a chemical reaction? 1. Increasing the…
A: The rate of a chemical reaction depends upon: 1. Temperature: Generally Reactions speeds up with…
Q: Which of the following would NOT increase the rate of a chemical reaction? An increase in the…
A: Rate of a reaction depends on 1. Concentration of reactants 2. Temperature 3. Catalyst
Q: H2O (s) ---> H2O (l) is what type of reaction
A: To define the type of below reaction H2O (s) ---> H2O (l)
Q: . a) changing the nature of the reactants Changing the nature of the reactants might increase or…
A: 1) changing the state of reactant as solid have less kinetic energy while gas have high kinetic…
Q: CH3 CH3 CH, -C-CH,-C=CH2 Lindlar's A H, catalyst B 2HI NANH,
A: Organic reactions are those that involve organic compounds as reactants. The reagent can be organic…
Q: Show the steps, intermediates, and reaction conditions
A:
Q: What happens to the activation energy as the temperature in a reacting system increases? the…
A: *Activation energy is extra amount of energy needed to make the reaction effective . *…
Q: For some medical procedures, doctors cool the patient's body before beginning. Following the…
A: ✓Normal functioning of human body is based on homeostasis. ✓Homeostatis is the process by which the…
Q: Which of the following is most closely associated with relatively slow rates of chemical reaction?…
A: High concentration of reactants , low energy of activation and presence of catalyst will increase…
Q: Which factors will influence the rate of a chemical reaction? O adding a catalyst O changing the…
A: The rate of reaction depends on various factors and can be altered by changing specific parameters.
Q: Outline how and why increasing temperature, concentration, and a catalyst can speed up the rate of a…
A:
Q: To complete the following statement: To speed up the reaction, a catalyst... A) increases the…
A: The main role of catalyst is to bring a short way for any certain reaction to take place. Therefore,…
Q: Which of the following will not affect the reaction rate of a reaction? Increasing the surface area…
A: The rate of a reaction depends upon the following factors
Q: Add all four reactions steps together to get the overall reaction. Remember to cross out anything…
A: For a multistep reaction, the overall reaction is obtained by combining the intermediate steps. All…
Q: I. Reacting Mg with Aqueous HCl Time & Day ofcure 1. Appearance of Mg reac you will complete in th…
A: Reaction 1 : Reactants given are Mg and HCl. Since Mg is a solid metal and HCl is a solution Hence…
Q: A. If a reaction will occur, give the product(s) otherwise write no reaction. Also indicate if the…
A:
Q: Write the overall reaction identify reactants, products, catalyst and reaction intermediate
A: The overall reaction can be written as: O3 (g) + Cl2 (g) → O2 (g) + Cl2O (g)
Q: Describe how increasing the concentration of a solution affects the rate of a chemical reaction.
A:
Q: A is a substance that increases the rate of a reaction without being used up during the reaction. O…
A:
Q: Solutions make it easier to A.handle chemicals safely B.react chemicals with one another…
A: Solution is made up of two components. Solute and solvent. The component which is present in major…
Q: How does a catalyst affect each of the following: (a) reaction rate; (b) ΔH; (c) Ea; (d) K; (e)…
A: The effect of a catalyst on the given products is shown below.
Q: Which of these reactions will not produce hydrogen gas when carried out at room temperature?…
A: The answer is given as follows
Q: Type of Reaction: Description: What is happening in the cartoon? Explanation: How does the cartoon…
A:
Q: Minimum amount of energy needed by a chemical reaction. reaction meeh anism C. rate activation enery…
A: Solution: Chemical reaction is defined as the reaction between one or more reactants to form one…
Q: e Types of reactions
A:
Q: Which of the following statements about a catalyst is true? O Catalysts increase the rate of the…
A:
Q: Why does the rate of most reactions decrease over time? A. A decrease in reactant
A: We have to tell Why does the rate of most reactions decrease over time
Q: The reaction A(aq) + B(aq) C(aq) + D(aq) has a ΔHrxn = -85 kJ/mol. This reaction is…
A:
Q: H2C-O-Ĉ-R H2C-OH HO-CH ОН R-C-о-сн R,-CH2·OH R1-CH2-0 Catalyst 3 H2Ć-OH Нас-о-с-R
A:
Q: For the reaction below: HO H*, catalyst
A: The reaction between catechol and acetone ot give 4,4'-(propane-2,2-diyl)bis(benzene-1,2-diol). The…
Q: Solve the equation NaI+H2O--->
A: In this question we have to solve the given reaction problem. NaI + H2O ------> Here Na is…
Q: In the following reaction mechanism: Cl + O3--> ClO + O2 ClO + O3 --> Cl + 2O2 The intermediate…
A: The reaction mechanism is, Cl + O3--> ClO + O2 ClO + O3 --> Cl + 2O2 Final equation = O3 + O…
Q: _CaBr2 + КОН- A Reaction Type: --> --- S D, SD, DD, or C?
A: First we write the Complete Balanced equation and then see the type of reaction. You can see…
Q: describe 3 ways to increase the rate of a chemical reaction.
A: Rate or speed of the reaction can be increased in ways as discussed in the following step.
Q: O 1. Na/NH3 2. RCO3H 3. H30* O 1. Na/NH3 2. cat. OsO4/NMO O 1. H2/Lindlar's catalyst 2. RCO3H 3.…
A: Alkynes are organic compounds containing triple bonds and alkenes contain double bonds.
Q: Figure 10 CH3 CH3 Lindlar's A CH3-Ć-CH2-c=CH2 H, catalyst B 2HI C D + NANH,
A:
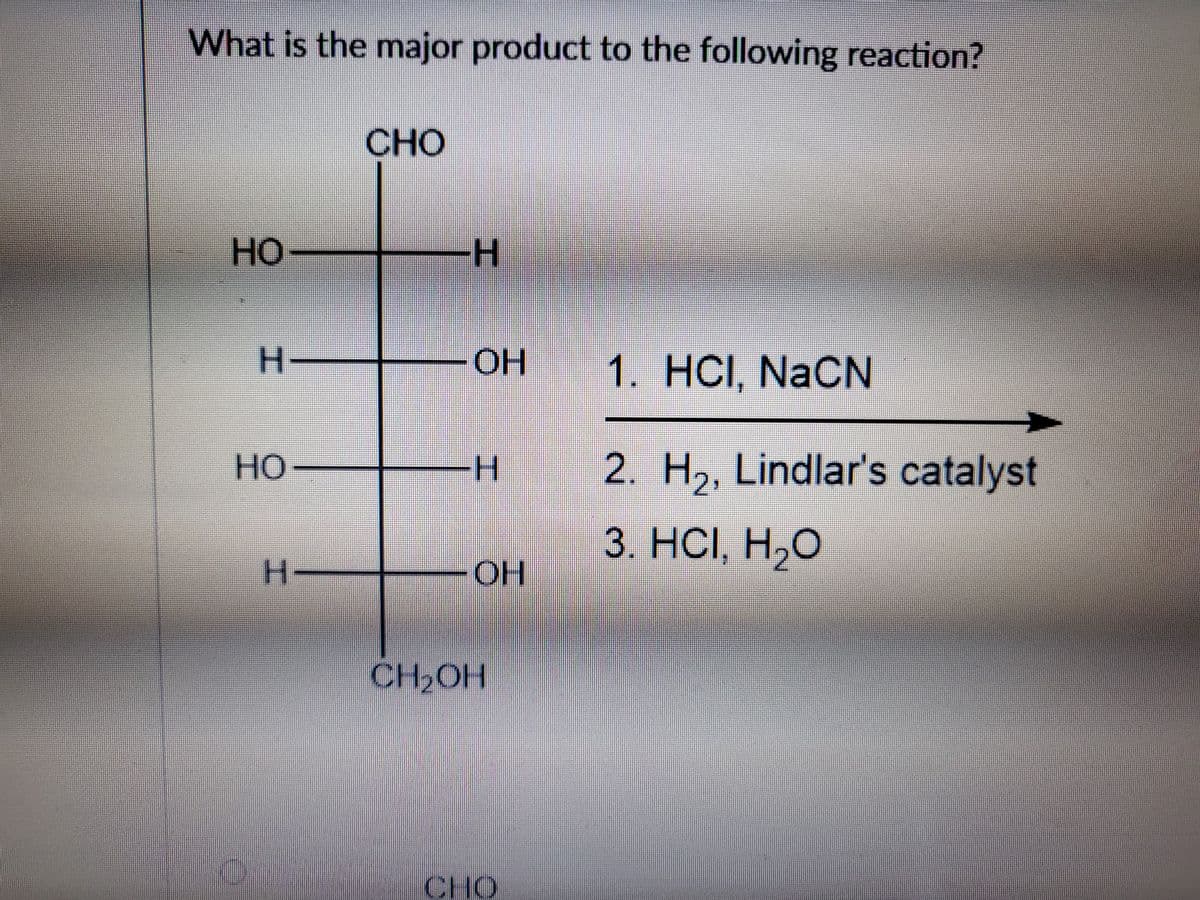
Step by step
Solved in 2 steps with 1 images

- C3H6 + H2O + H2SO4 --> C3H8O + H2SO4 what is the catalyst in the reaction above?(CH3)3CHOH is reacted with CH3-O-C--C6H5 in a transesterification reaction. What is the M+ of the product? || O 102 80 178 None of theseIn the reaction from compound 10 to compound 11, why the C=C bond is retained and is not hydrogenated? f) LiAlH4, Et2O, 08C; g) Ac2O, py, DMAP, CH2Cl2, RT, 77% over 2 steps;
- Which compound is the main product of the reaction in Figure 22? * Compound A Compound B Compound C Compound D None of the above.What is the missing product in the following reaction? ZnCO3--> ____________+ CO2Trimethyllead hydride, (CH3)3PbH, can react with methyl bromide (CH3Br) in a homolytic dissociation reaction. What will the lead-containing product be? (CH3)2Pb PbBr2 (CH3)3PbBr Pb metal
- Ca2+(aq)+C2O42 ( aq) CaC204 (s) kc = 2.3 *10-9 1.Explain the components of the equilibrium mixture based on the value of the equilibrium constant . 2. What can be said about the rate of the reaction of ethanol versus the rate of reaction of the oxidation of ethylene glycol ? 3.Large amounts of ethanol are indicated for the treatment of ethylene glycol poison , using the LeChatelier Principle discuss why this treatment is effective.Chemistry What is the major product of each of the following reactions?2H2S(g) + SO2(g) <--> 3S(s) + 2H2O(g) Would this reaction be favored at a high or low temperature?

