Q: When copper metal is added to nitric acid, the following reaction takes place Cu (s) + 4 HNO₃ (aq) ...
A:
Q: ΔΗν your a
A:
Q: 75mL of a 0.2M solution of a tripeptide Ser-Cys-His is adjusted to pH=8.6. The solution is then titr...
A:
Q: Calculate the free energy change accompanying the transfer by osmosis at 30 °C of one mole of water ...
A:
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of t...
A:
Q: The volume dispensed from the buret amounts to 32 33 32.25 ml O 33.75 mL O 33.70 ml O 32.20 mL
A: Given,
Q: Indicate the electron pair geometry and the molecular geometry for each of the six compounds. Molecu...
A: Electron pair geometry includes both bond pair and lone pair while molecular geometry only consider ...
Q: Arrange the following ions in order of increasing ionic radius: sodium ion, aluminum ion, fluoride i...
A: Explanation - The electron or multi-electron takes into account the number of shielding electrons th...
Q: Estimate the theoretical density for molybdenum, a BCC metal, when the atomic mass and atomic radius...
A:
Q: QUESTION 4 How many milligrams of each NaCl and lidocaine HCl are needed to prepare 75 mL of a 1% so...
A: A hypotonic solution can be made isotonic by adding an adjusting substance, usually sodium chloride;...
Q: The Pauli Exclusion Principle tells us that no two electrons in an atom can have the same four quant...
A: Concept is based on Quantum numbers:
Q: compounds are ionic and which are covalent
A:
Q: What parameters should be considered when choosing between distillation methods? Are there any diffe...
A: Distillation is refining method of refining by alternating evaporation and condensation to remove ...
Q: What is the maximum amount of hydrogen iodide that can be formed? grams What is the FORMTLA for the ...
A:
Q: volume of carbon dioxide gas Es. CS.
A:
Q: 1) Complete the esterification reaction below by providing the structure of the missing compound. Ac...
A:
Q: An Organic compound contains 59.00% by mass C, 7.15% by mass H, 7.65% by mass N, and 26.20% by mass ...
A: Given data is as follows: The elements are given in percent which is out of 100. So, the mass of C =...
Q: concentration
A:
Q: C = C1H CH3 2 NaNHa 31 H+ 000
A: See mechanism below.
Q: Look for materials at home which can be classified as pure substances or mixtures. Identify at least...
A:
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of t...
A:
Q: 13.22 grams of the gas X2 are placed in a sealed container at 35.0°C with 15.99 grams of the gas T2....
A:
Q: The solubility of Ag2CrO4 (Ksp = 1.2 x 10-12) is several times greater than that of AgCl (Ksp = 1.82...
A: Given: Mass of K2CrO4 = 1 mg Ksp Ag2CrO4 = 1.2 x 10-12 Ksp AgCl = 1.82 x 10-10 To determine: The ...
Q: three significant figures.
A: For rounding off to three significant figures, we need to check the fourth number from the left.
Q: weight of Mn ore
A:
Q: Draw the Newman pr0jecti0ns 0f the three p0ssible staggered c0nf0rmati0ns 0f 2,3-dimethylbutane, vie...
A: Newman projection is useful in alkane steriochemistry visualizes the conformation of a chemical bond...
Q: 1.) How many grams of pure phosphoric acid (H3P04) is present in a liter of this 85% by mass solutio...
A: A question based on quantitative analysis, which is to be accomplished.
Q: 1A 8A H 2A 3A 4A 5A 6A 7A He Li Be BCNOF Ne Na Mg 3B 4B 58 6B 7B - 8B - IB 28 AI si PS CI Ar K Ca Sc...
A: Atomic radius increases when we go down in the group(vertical) from top to bottom and atomic radius ...
Q: The great French chemist Antoine Lavoisier discovered the Law of Conservation of Mass in part by doi...
A: 1. Balanced chemical equation Given;
Q: equilibrium constant
A:
Q: Titration of 15.00 mL of a weak, monoprotic acid solution requires 22.84 mL of a 0.09837M standardiz...
A: Given Volume of weak acid ( V1 ) = 15 mL Volume of NaOH solution ( V2 ) = 22.84 mL Molarity o...
Q: In Pure In Mixed Mixed Pure With With Solute Solute
A: The table is given as,
Q: 2. Consider the following hypothetical results for an experiment like this one. Complete the table a...
A: Metal or element which is more reactive than other element displace other element from its salt solu...
Q: Boron trifluoride gas is collected at 8.0 °C in an evacuated flask with a measured volume of 5.0 L. ...
A: Given, Volume of vessel = 5.0 L Temperature, T = 8.00C = 281 K P...
Q: What is the molality of a salt solution prepared by dissolving two moles of salt in 500 grams of wat...
A: What is the molality of a salt solution prepared by dissolving two moles of salt in 500 grams of wat...
Q: 1. LIAIH4 (i) N. H. 2. H30* ОН (G) MnO2 or PCO 1. CHзCH2MgBr В А 2. H20
A: 1) Reaction - mechanism-
Q: :ö: 0-H :ở ö-H :o :ö H-O.
A:
Q: Consider the following reaction of K2S2O3 for neutralizing chlorine in industrial bleaching. K2S2O3 ...
A:
Q: Why add a pinch of NaHCO3 to NaCl sample solution that will be used as the analyte in a titration wh...
A: NaHCO3 is known as sodium Bicarbonate which is a weak base and used as AntaAcid Also .
Q: A titration experiment required 12.50 ml of standard hydrochloric acid solution to reach the phenolp...
A: Volume of HCl required for phenolphthalein endpoint = 12.5 mL Volume of HCl required for methyl oran...
Q: 12a. Complete the Lewis structure of glycine by adding the five (5) hydrogens in the structure, lone...
A: Given :- Molecular formula of glycine = C2H5NO2 To draw :- Its lewis structure Also determine s...
Q: Identify what state/s of matter is/are applicable for the following reagents: Choices: Solid Liqui...
A: Matching of state's of matter.
Q: please balance NH4I + Cl2 → NH4Cl + I2 is it a single displacement reaction
A:
Q: 1) Complete the esterification reaction below by providing the structure of the missing comp Acid ca...
A: This is an esterification reaction in which acid and alcohol in presence of acid form Ester
Q: If cyclopentane were planar and each eclipsing interaction costs 4.0 kJ/mol, how much torsional stra...
A:
Q: (R)-2,2-dimethylcyclopentan-1-ol (а). bicyclo[4.1.0]heptane (b). cis-
A: To solve this problem we have to write the structure of the given compounds .
Q: How much oxygen is needed to produce 105.2 kJ via the following reaction? 2C2H6(g) + 7O2(g) → 4CO2(g...
A: ∆H = -3120 KJ/mol How many moles of O2 will produce 105.2KJ Molar mass of oxygen = 32.0 g/mol
Q: An analytical method that is based on determining the volumes of reagent of known strength that is r...
A: Volumetric titrations involve measuring the volume of a solution of knownconcentration that is neede...
Q: Draw the skeletal (line-bond) structure of (S)-3-bromo-3- iodohexane.
A:
Q: A 0.128 g sample of KHP (HKC8H4O4) required 28.54 mL of NaOH solution to reach a phenolphthalein end...
A:
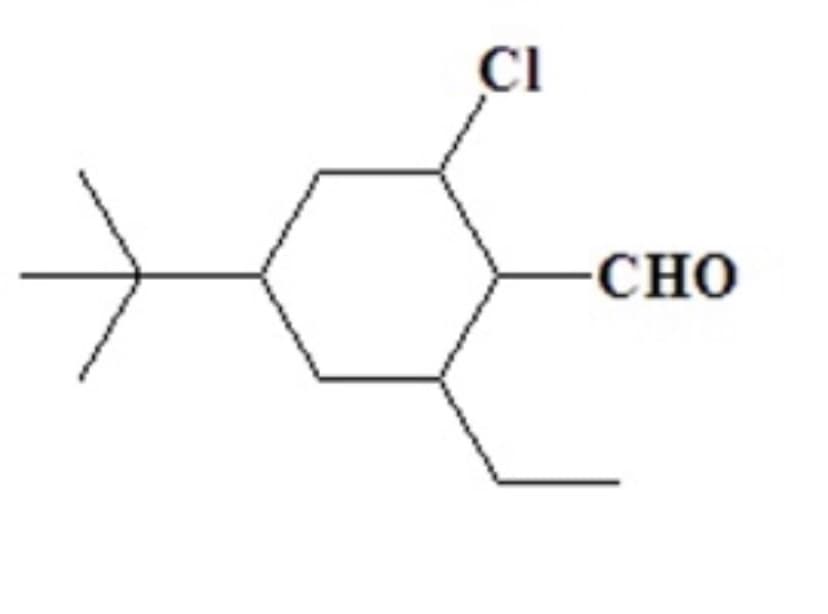
Step by step
Solved in 2 steps with 1 images


