Q: Discuss the reasons for the differences in glass transition temperature for the following pairs of…
A: As we can see that the transition temperature for Poly(vinyl alcohol) (85°C) is greater than…
Q: Determine the alkalinity of a 1M solution of (a) H2CO3 (b) Na2CO3 For this problem, pKa,1 = 6.3 and…
A: The alkalinity of a solution means its pOH value.
Q: Identify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this…
A:
Q: What is the molar solubility of Tl2S in deionized water relative to the molar solubility of Tl2S in…
A:
Q: Benzophenone starting material Relative Transmittance T 0.8 0.6 0.4- 0.2 INFRARED SPECTRUM C-H…
A: IR spectra is used to identify the functional groups of unknown compound based on the characteristic…
Q: 14.5 Assign IUPAC names to the following aldehydes and ketones:
A: There are some basic rules for the naming the organic compounds : Find the longest continuous…
Q: c) Calculate the pH of a mixture formed by adding 50.0ml of 0.020M HCI to 150ml of 0.010M HI?
A: C) pH of a solution is defined as the negative logarithm of hydrogen ion concentration. pH = - log…
Q: . Fermentation is a complex chemical process of making wine by converting glucose into ethanol and…
A: So, this is simple question combined with the concept of gaseous state and mole concept.
Q: A solution is prepared by diluting 37.0 mL of 7.5x10-2 MBa(OH)2 to a volume of 315.5 mL. Express the…
A: In this question, we will determine the pH of the diluted solution of Ba(OH)2 You can see the…
Q: A flat nonstereospecific version of 1-chloro-2-bromo-3-iodocyclohexane is shown in Figure 7-2. Which…
A:
Q: For the aqueous ammonia (NH3) equilibrium, what affect would be observed if concentrated HCl were…
A: The aqueous ammonia equilibrium involves the reaction between ammonia (NH3) and water (H2O) to…
Q: Determine the pH during the titration of 29.7 mL of 0.450 M acetic acid (K₂ = 1.8x10-5) by 0.367 M…
A: “Since you have posted a question with multiple sub-parts, we willprovide the solution only to the…
Q: 4 3 D
A: Axial and Equatorial are types of bonds found in the chair conformation of cyclohexane .The chair…
Q: Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure -] :H-…
A: We have to check if the given Lewis structures are reasonable
Q: actually added to 1 tablet (L) *Concentration (M) of HCI Report to 4 sig figs Expand M out to a…
A:
Q: In the titration of 95.0 mL of 0.400 M HCOOH with 0.150 M LIOH, how many mL of LiOH are required to…
A:
Q: (9 лдви 2. NH4Cl, H₂0 Brz CH ₂ C l ₂ 1 полен V z.en. Nowa Лон ? ?
A: The question is based on the concept of organic reactions. we need to identify the product formed in…
Q: 12. How many grams of MgCl₂ are needed to make 700 mL of a 1.40 M solution? (MgCl₂ MW: 95.211)
A: 12) Given data is Molarityof MgCl2 solution required= 1.40 M Volume of MgCl2 solution= 700 mL…
Q: Balance the chemical equation: Ca(C2H3O2)2 (aq) + Na2Co3 (aq) --> CaCO3 (s) NaC2H3O2 (aq)
A:
Q: 5. What is the final ONPG concentration (in mM) of each of the Reaction tubes? ***Hint: Use the…
A: O-nitrophenyl-beta-D-galactopyranoside (ONPG) test id done to study enzyme kinetics of β…
Q: A helium balloon takes up 4.35 liters of space. How many moles are in the balloon?
A: Since, We know that, Standard volume at STP = 22.4 L then,
Q: Macmillan Learning Automobile airbags contain solid sodium azide, NaN,, that reacts to produce…
A: To calculate the value of work for the system if 16.3 g NaN3 reacts completely at 1.00 atm and 22…
Q: Draw the diene and the dienophile used to make: HN O H H
A: This question belong to Pericyclic reaction that is Diels-Alder reaction. Diels-Alder reaction:…
Q: Using the formula M1V1 = M2V2, how many millimeters of 6.0M sodium hydroxide are required to…
A:
Q: 50.0 mL of 0.100 M HOAc is titrated with 0.100 M NaOH. Calculate the pH at the beginning of the…
A: An acid-base titration is the titration used to determine the unknown concentration of an acid or…
Q: Explain why is HI the strongest acid in this periodic table group.
A: In the given question we have to find the reason that why H-I is strongest acid.
Q: What is the pH of a buffer that has 0.150 mol HC2H3O2 and 0.120 mol NaC2H3O2 in 2.00 L that has…
A:
Q: Determine the mass of Br2 produced from the reaction of 7.00 mol of HBr with excess K₂SO4. K₂SO4 + 2…
A: Answer: Thee quetsions are based on the concept of limiting reagent. Limiting reagent is the…
Q: 2. Predict the major product(s) for each of the following reactions. 1. PhMgBr 2. H₂O OH CrO3 py
A:
Q: Shown below are Newman projection formulas for (R,R)-, (S,S)- and (R,S)-2,3-dicholorobutane (a)…
A:
Q: An unknown weak base with a concentration of 0.170 M has a pH of 9.42. What is the Kb of this base?
A: Given data : Initial Concentration of Base (C) = 0.170 M pH = 9.42 Required : Base Ionization…
Q: -6-0 SO3H dentify the reagents represented by the letters in the above reaction scheme. Enter your…
A: Reaction involved following concepts 1) Fridel crafts alkylation 2) sulphonation 3) oxidation of…
Q: Given the following reactions: 3C ⇒D Kc=7.13× 10-4 D3A+3B Kc=8.54 × 10-² Calculate the value of the…
A: The question is based on equilibrium concepts. we need to calculate equilibrium constant value for…
Q: Using the reaction enthalpy data provided in Q4, predict the relative strength of the H-X bond for…
A: concept used : High bond dissociation energy means high bond strength, low bond energy means low…
Q: Predict the reagents and/or conditions and draw the detailed mechanism for the following reaction.…
A:
Q: Name the following amine by the IUPAC system of nomenclature: NH ₂ CHỊCHỊCH,CH(CH3)
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: The H- ion cannot be solved exactly because it includes more than two distinct particles. True False
A: Answer: Usually Hydrogen is a cation. But some reaction Hydrogen is act as an anion. Hydrogen anion…
Q: Fiдеге 7-1 Row 1 3 3 A д В о 141
A: In chair form of cyclohexane as the number of methyl groups in axial position increases the…
Q: cl CHÍCH 32 THF, A Elimination
A: This is a simple question about the reaction mechanism of organic reaction specially reaction of…
Q: Br CH3 The acetyl group is meta-directing. Sy b Draw the resonance forms for the carbocation…
A:
Q: The acid dissociation constant K of alloxanic acid (HC, H, N₂O₂) is 2.24 × 107. Calculate the pH of…
A:
Q: For the energy level n=4, how many orbitals are possible and what are its m and ml values.
A: This question belong to Atomic Structure. Quantum number. Principal quantum number: n Azimuthal…
Q: Write the IUPAC name of each compound. (a) (b)
A: Rules for naming the compound containing multiple bond are : Find the longest continuous chain of…
Q: 2) Mariana carefully reacts zinc metal with nitric acid in a safety hood to create zinc nitrate,…
A: As per the given reaction Zinc reacts with nitric acid to form zinc nitrate, water and nitrogen…
Q: What do you predict would happen if you added 12 M NaOH to a saturated NaCl solution (how would the…
A: A saturated solution is one that cannot dissolve more solute. It maintains equilibrium between…
Q: Hexane has a density of 0.655 g/mL. Water has a density of 1.000 g/mL. What volume of Water has the…
A:
Q: At normal temperatures and pressures, hydrogen gas is composed of H₂ molecules. An energy diagram…
A: Bond energy is the energy required to completely break the bond . From graph minimum energy in…
Q: Name the following:
A: IUPAC nomenclature refers to the set of rules and conventions used to name organic and inorganic…
Q: First-order perturbation theory corrections to the energy require knowledge of the first-order…
A: First order perturbation theory correction to energy doesn't require first order corrected wave…
Q: Most atoms have multiple term symbols associated with their electronic configuration because…
A:
G.47.
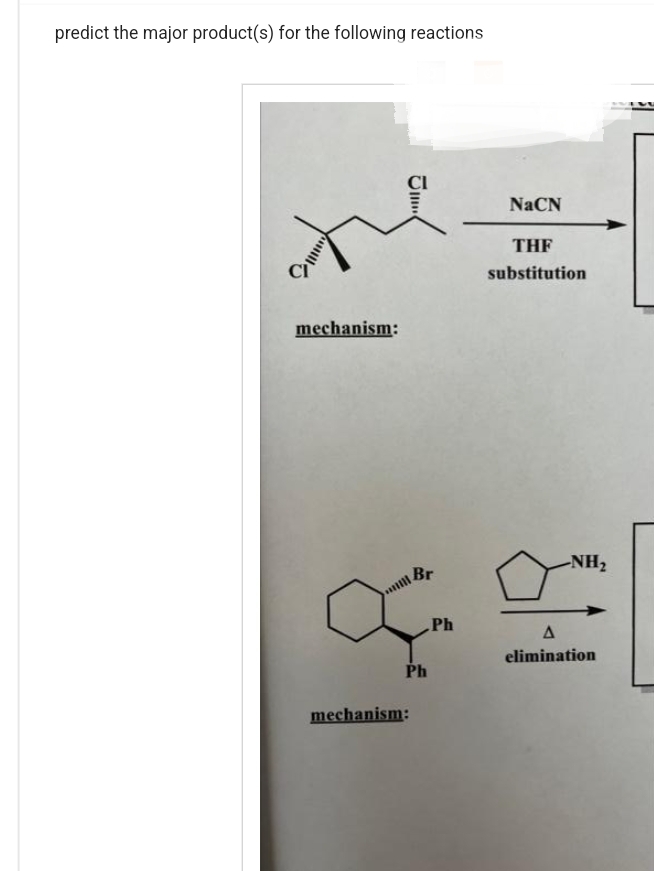
Step by step
Solved in 3 steps with 3 images


