Q: Iodine is prepared both in the laboratory and commercially by adding Cl2(g)to an aqueous solution co...
A: When stoichiometric coefficient results in the equal number of atoms of each element on both the rea...
Q: Two patterns of packing for two different circles of the samesize are shown here. For each structure...
A: Unit cell is the smallest repeating unit in a crystalline solid. If this unit is stacked over and ov...
Q: What is the ratio of urms to ump for a sample of O21g2 at 300 K? Will this ratiochange as the temper...
A: Calculate μrms (root mean square) as follows. In the expression, R is universal gas constant, T is...
Q: Question attached
A: The standard reduction potential of Ni2+/Ni = -0.23 V Whereas that of Pb2+/Pb = -0.13 V Fr...
Q: Suppose you are trying to synthesize the dipeptide Val-Ser. Compare the product that would be obtain...
A: Thionyl chloride is used to convert the carboxylic acid into acid chloride.
Q: Give at least two methods for the synthesis of 2-methylpropane. What happens if it is treated with: ...
A: Preparation:
Q: Draw the products formed when A or B is treated with fo following agent. In some cases, no reaction ...
A:
Q: (a) Give chemical tests to distinguish between the following pairs of compounds :(i) Ethanal and Pro...
A: Hey, since you have asked multiple question, we will solve the first question for you. If you want a...
Q: During Friedel Crafts Acylation , Why does AlCl3 have to be anhydrous??
A:
Q: Please help me with my hw. Please tysm❤️Activity: Answer the following problem. Show complete soluti...
A:
Q: Determine the volume of CO2 obtained from the combustion of 145 gpropane, C3H8 (at 1741 mmHg and 250...
A:
Q: 2 g aluminum sulfate in 200 g of water . density of aluminum sulfate ; density of h20
A: Density can be determined by using the following formula: Density = mass/volume
Q: (AlOOH) ore is the most preferred in aluminum production and hematite in iron production of the ores...
A:
Q: The freezing point of a 1 molal aqueous solution of the nonelectrolyte ethylene glycol (the principa...
A: The cracking of engine is prevented by adding ethylene glycol in the automobile radiator which lower...
Q: What is the effect of a temperature change on a chemical reaction initially at equilibrium?
A: If the reaction is exothermic then liberates heat energy. Consider heat is also one of the product. ...
Q: Write the complete, balanced equations for the following metathesis reaction. Include the notations...
A: The chemical formula of potassium carbonate is K2CO3 and the chemical formula of silver nitrate is A...
Q: Consider the titration of a 35.0-mL sample of 0.175 M HBr with 0.200 M KOH. Determine each quantity....
A: As you have posted question with multiple subparts, we will answer the first three subparts for you....
Q: Today, synthetic detergents like the compound drawn here, not soaps, are used to clean clothes. Expl...
A: Fatty acids are main component of lipids. They are carboxylic acid with long hydrocarbon chains and ...
Q: Activity 1: What is the Mystery Word? Directions: Answer the following questions and combine the ans...
A: The statements are, 1) The symbol of the element in the third period with 5 outermost electrons and ...
Q: 8. Air in a thundercloud expands as it rises. Assume that the initial temperature is 300K and no ene...
A: Solution Lightning is AN electrical discharge; i.e., a fast distribution of electrical charge among ...
Q: The standard reduction potential for the reduction ofRuO4-(aq) to RuO42-(aq) is +0.59 V. Which of th...
A: Given: The standard reduction potential for the reduction of RuO4-(aq) to RuO42-(aq) is +0.59 V
Q: An aromatic imine is formed when 3,5-dihydroxyphenol is treated with ethanenitrile (CH3CN) in HCI in...
A: In the first step, protonation of ethanenitrile with HCl occurs. Protonation makes the ethanenitrile...
Q: Determine electron configuration for each of the atoms or ions. A) Lr6+ B) As3- C) Pt
A: A) The chemical symbol of Lr is Lawrencium. This element is f-block element and is an actinoid. The ...
Q: Where is the force of attraction the strongest on a magnet
A: Magnets have north and south poles. The poles are the regions where the force of attraction is stron...
Q: Iodine is an important reagent in synthetic organic chemistry because bondsbetween carbon and iodine...
A: Given: Iodine is an important reagent in synthetic organic chemistry because bondsbetween carbon and...
Q: Locate the stereogenic centers in attached drug.
A: Those carbons having four different groups or substituents attached are called as stereogenic centre...
Q: How can you use UV spectroscopy to distinguish between the compounds in each of the following pairs?
A: Conjugation on λmax: The wavelength at which the pi to pi star transition occurs increase as the num...
Q: What products are formed by hydrolysis of attached imine or enamine?
A: The hydrolysis of imine gives the corresponding aldehyde or ketone. The reaction proceeds in the fol...
Q: Describe the VSEPR Theory?
A: VSEPR theory or valence shell electron pair repulsion theory is a model that is used to predict the ...
Q: Many lithium salts are hygroscopic (absorb water), whereas the corresponding salts of the other alka...
A:
Q: Oxygen has a volume of 15-liters at a pressure of 760 torr. What isthe pressure that would compress ...
A: It is given that oxygen has a volume of 15 litres when it has a pressure of 760 torrs, to determine ...
Q: P3D.4 The solubility of an ionic solid such as NaCl can be explored by calculating the standard Gibb...
A: For the first reaction, substitute values of absolute entropy and calculate change in entropy (∆S°) ...
Q: Determine whether each of the following properties is more likely to be for an organic or inorganic ...
A: Organic compounds are compounds of carbon. They contain carbon , hydrogen and can also contain othe...
Q: Metallic aluminum reacts with MnO2 at elevated temperatures to form manganese metal and aluminum oxi...
A: The reaction between MnO2 and Al –metal is shown as: Here, MnO2 and Al react in 3:4 ratio. Given th...
Q: PROBLEM 12.6 How would you prepare the following alkyl halides from the corresponding alcohols? (a) ...
A:
Q: Which of the following molecules matches the given data? HDI = 5 IR (cm1 ): 1724 (strong), 1604 (wea...
A: This is compound 2
Q: You and another student were each given an unknown compound. Both samples contained colorless materi...
A: Principle of TLC: Thin layer chromatography is the technique in which there will be a separation of ...
Q: Which of the reactions are exothermic? 2 Mg(s) + O,(g) → 2 MgO(s) + heat O NH, (g) + HCI(g) → NH,Cl(...
A: The reaction that releases heat during the reaction, is called exothermic reaction.
Q: Draw the products of the following acid–base reactions: a.CH3NH3 + H2Ob. HBr + CH3OH c. CH3NH3 + H...
A: Hello. Since you have posted multiple questions and not specified which question needs to be solved,...
Q: What other monosaccharide is reduced only to the alditol obtained from the reduction of1. d-talose? ...
A: Given compounds: d-talose d-glucose d-galactose
Q: Only about 0.16 mg of AgBr(s) will dissolve in 1.0 L of water (this volume of solid is smaller than ...
A:
Q: Question attached
A: A reaction is said to be balanced when mass and charge are same on either side of reaction. Molecula...
Q: From the sequential hydrolysis of ammonia borane in 10 mL aqueous solution at 25 ºC and 1 atm with 2...
A:
Q: Specify the molecular geometry of each of the following molecules using the terms linear, angular, t...
A: Different type of Molecules have different structural shapes and properties. Different type of conc...
Q: Show the cycle on P-v and T-s diagrams.
A: A system present in an initial state (P, V, T) can undergo expansion and compression based on severa...
Q: Show how you would convert(a) oct-3-yne to cis-oct-3-ene.(b) pent-2-yne to trans-pent-2-ene.(c) cis-...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: Write molecular formulas for each bicycloalkane, given its number of carbon atoms. Q.) Hydrindane (9...
A: The alkanes that contain two rings which are shared by two of atoms of carbon is known as bicycloalk...
Q: With all details
A:
Q: Question attached
A: Leaving group is an atom or group of atoms which is fragmented from rest of the molecule, and takes ...
Q: QUESTION 1 etermine the rate law and the value of k for the following reaction using the data provid...
A: From the given data, First, we find the order of Reaction then we calculate the rate constant. Then...
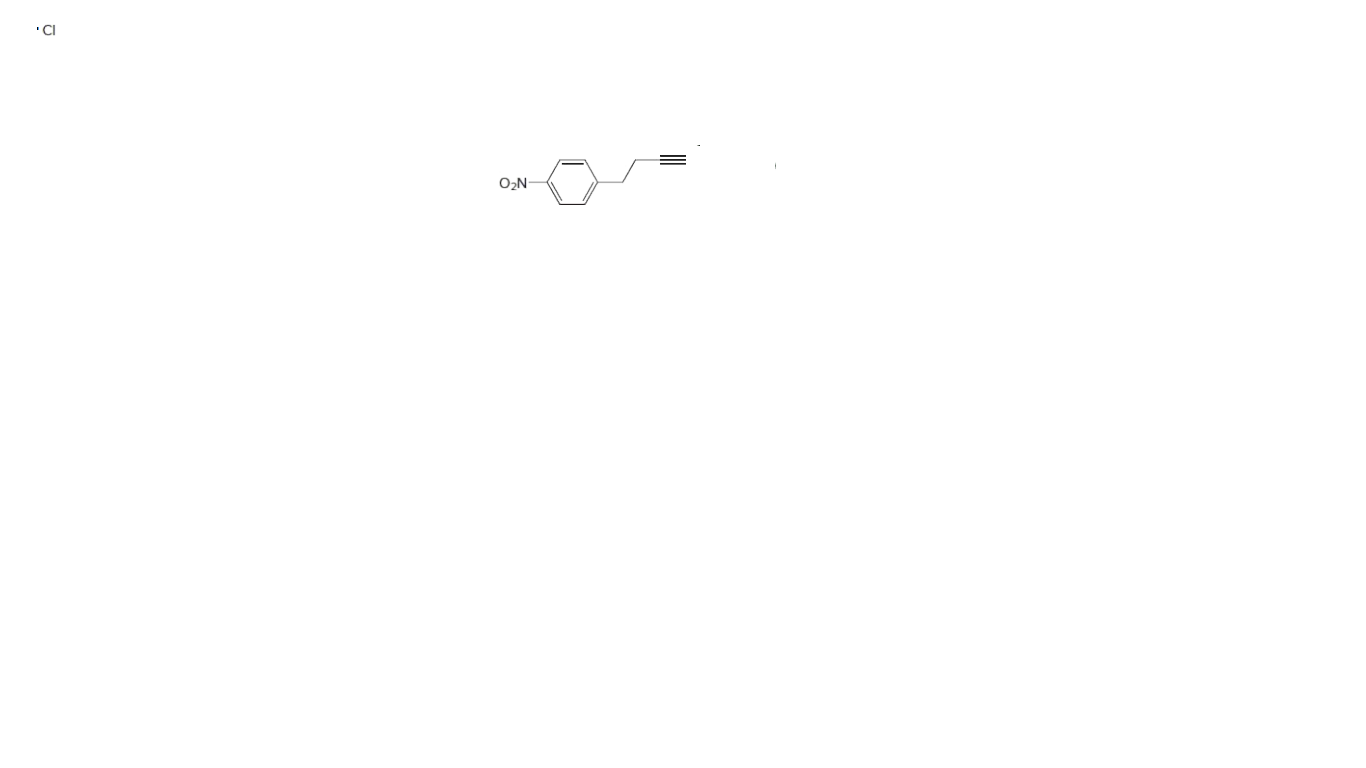
Synthesize each compound.You may use benzene, acetylene (HC≡CH), ethanol, ethylene oxide, and any inorganic reagents.

Step by step
Solved in 2 steps with 2 images


