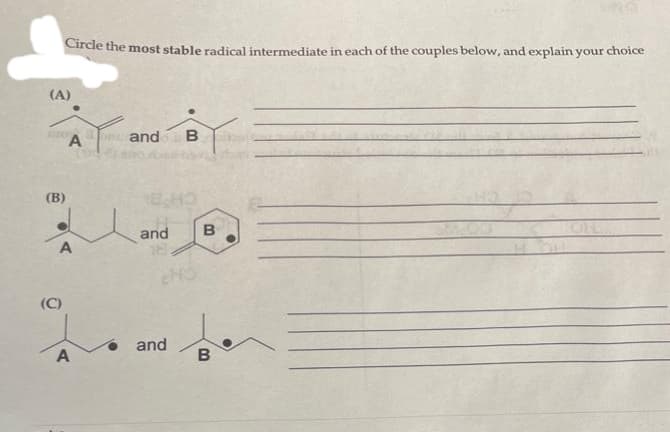
Circle the most stable radical intermediate in each of the couples below, and explain your choice (A) (C) T A (B) 2 A and B 18,40 and and HO B B
Q: (i) (k) (m) OH Хоні X, I PCC CH₂Cl₂ CrO3 OH aq. H₂SO CH₂O K H₂SO4 CH3OH (h) (1) XOR CrO; OH aq. H₂SO…
A:
Q: ition 4 what is the percent composition of a mixture of 5)-2 butanol (elp 13.52 and 04+2 butanol…
A:
Q: The following compound is H Br- H3C H CH3 Br O chiral O achiral but not meso O meso
A:
Q: Identify the substance in each of the following pairs that would have the higher boiling point and…
A: Stronger are the intermolecular forces , more is the boiling point. The strength decreasing order…
Q: UNKNOWN NUMBER: 24 UNKNOWN CATION IDENTITY (with charge shown): M Write molecular, ionic, and…
A: “Since you have posted multiple questions with multiple subparts, we will provide the solution…
Q: How many milliliters of 0.653 M NaOH are needed to neutralize 25.0 mL of a 1.02 M HBr solution? The…
A: A balanced chemical equation contain equal number of atoms of each elements on both sides of the…
Q: Compute the standard electrode potential for the following half-reaction Ni(CN)42- +2e-→ Ni(s) +…
A: Given: E0= -0.25 V Formation constant of Ni(CN)42- = 1.0 x 1022
Q: Predict the product of the following reaction. F H CH₂OH
A: In epoxide ring opening reaction, when acidic medium is used then carbocation is formed and ring…
Q: Give typed explanation A 0.10 M solution of an acid, HA, has a pH of 3.0. What is the Kb of the…
A: We have given, Concentration of acid ,HA= 0.10M pH = 3.0 Kb of conjugate base=?
Q: Ok. I'm still not sure what the chloro benzene is used for?
A: It is a follow up question.
Q: Draw the product that could be formed when 1,3-butadiene reacts with maleic anhydride. Include any…
A: Diels-Alder reaction: It is a [4+2] cycloaddition reaction between a diene and a dienophile to form…
Q: What is the overall order of a reaction with units: M¹/2/s, for the rate constant (k)? 0 O 1/2 01…
A: The unit of rate constant is M1/2/s.
Q: A voltaic cell consists of a Zn/Zn²+ half-cell and a Ni/Ni2+ half-cell at 25 °C. The initial…
A: Given: Initial concentration of Ni+2 = 1.60 M Initial concentration of Zn+2 =0.110 M…
Q: I Which of the following is the correct formula for the ion that would form between iron(III) and…
A:
Q: Although they were formerly called the inert gases, at least the heavier elements of Group 8 do form…
A: To find the theoritical yield and percentage yield of a given chemical reaction.
Q: 1. Draw the following structures. Note the following: i) Use bond-line (skeleton) formulas, with…
A: Meso compound is optically inactive. In spite of having two or more stereocenters, the molecule is…
Q: line is observed at 1.88 X 103 nm in an emission spectrum. This line may be due to a transition from…
A: The Rydberg formula has been used to determine the wavelengths of several spectral series that have…
Q: 3) What is the neutral intermediate in the acid-catalyzed esterification (Fisher esterification) of…
A: Fischer esterification is the formation of ester from reaction between carboxylic acid and alcohol…
Q: 1a. For the following reaction, write how the each of the changes will affect the indicated…
A: Solutions- Here we have to determine the how the each of the changes will affect the indicated…
Q: How many grams of Nacl are needed to prepare 360ml of a 6.50 M solution? Show work.
A:
Q: 1. The reaction of sodium and potassium with the solutions listed in the chart were not tested.…
A: We have to predict the results of the reactivity of sodium and potassium with the solutions listed…
Q: According to valence bond theory, when a double bond is formed: Select True or False.…
A: Valence bond theory (VBT) explain the formation of molecule by chemical bonding and overlapping of…
Q: How many moles of carbon dioxide are in 211 g of Carbon dioxide? 4.8 4.79 (with margin: 0)
A:
Q: Provide a systematic name (correct spelling required) for the following compounds: a) Al2S3 b) CaO…
A: We will discuss the name of Given compounds
Q: The completed Lewis structure of CH4 contains a total of 0 covalent bonds and 0 lone pairs. NOTE: If…
A:
Q: A certain catalyzed reaction is known to have an activation energy E=38.0 kJ/mol. Furthermore, the…
A: This question belongs to Chemical kinetics.
Q: The molar solubility of magnesium carbonate in a water solution is M.
A: The value of Ksp of MgCO3 is 3.5×10-8.
Q: What is the pH of a cleaning solution with a [H3O+] 7.40 × 10-⁹ M H3O+? =
A: Given [H3O+] =7.40×10-9 M pH = ? pH is defined as negative logarithm of hydrogen or hydronium ion…
Q: 10. Draw the Newman projection of all the conformers for 4-aminohexan-3-ol (see structure below).…
A: Newman projection is a 2D representation between two carbons of any 3D molecule. The front carbon is…
Q: As we learned previously, Benadryl, C₁7H21ON, is a weak base. a. The pk, of Benadryl is 5.00 at 25…
A: pKb is defined as the negative logarithm of Kb. It can be given as pKb = - log Kb Given:…
Q: is the following Diamagnetic or paramagnetic: H3BO3 C2H5OH (l)
A: The magnetic form of a substance is determined by its electron configuration. If it possesses…
Q: Advantame is often used as an artificial sweetener. In the structure of advantame shown, highlight…
A: The general formula of some of the functional group is given below:
Q: A researcher is trying to determine the reaction order for the reaction A→B+C by measuring the…
A:
Q: The rate constant for a certain reaction was measured at two different temperatures. At 73.5 °C it…
A:
Q: Zinc reacts with hydrochloric acid according to the reaction equation Zn(s) + 2 HCl(aq) → ZnCl₂(aq)…
A: The balanced equation is Zn(s) + 2HCl(aq) ⇒ ZnCl2(aq) + H2(g)
Q: A diprotic acid, H2A, has values of Ka1 = 1.0×10–5 and Ka2 = 1.0×10–10 at 25 ºC. In a 0.10 M aqueous…
A:
Q: When 18.0 mL of a 5.55x10-4 M iron(III) acetate solution is combined with 15.0 mL of a 7.65x10-4 M…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: which compound does this mass spectrum represent? Please provide only typed answer solution no…
A: A question based on mass spectrum. A mass spectum of an unknown compound is prescribed, using that…
Q: The molar solubility of iron (III) hydroxide in a 0.241 M iron(III) acetate solution is M.
A: Fe(OH)2 (s) <------> Fe2+ (aq) + 2OH- (aq) Ksp = [Fe2+][OH-]2 Ksp = 8.0*10-16 Iron acetate…
Q: Consider the titration of 100.0 mL of 0.200 M acetic acid ( Ka 1.8 × 10-5) by 0.100 M KOH. Calculate…
A: We have to calculate the pH of solution
Q: Does a reaction occur when aqueous solutions of manganese(II) chloride and silver(I) sulfate are…
A: Balanced molecular equation: Balance molecular equation can be define as the reaction in which…
Q: Multiple reagents are missing from the reaction below. Using chemistry from this exam material, what…
A: When alkene reacts with per acid like meta chloro perbenzoic acid (mCPBA) it gives epoxide
Q: A 27.3 mL sample of a 0.454 M aqueous hydrofluoric acid solution is titrated with a 0.493 M aqueous…
A:
Q: D Question 3 NCI 3 How many single bonds are on the nitrogen? How many double bonds are on the…
A: This question belongs to Chemical Bonding.
Q: Calculate the value of the free energy change, AG, for the reaction below at 227.0°C when the…
A: The relation between the equilibrium constant and free energy can be given as ∆G°=-RT ln K where,…
Q: Here are the successive ionization energies for strontium Sr (in kJ mol-1): I1 = 550 I2 = 1064 I3 =…
A: The amount of energy required by an isolated, gaseous atom in the ground electronic state to absorb…
Q: 7. Identify which categories of elements usually bond covalently, and explain why this is the case
A: The question involves the concepts of covalent bonding . we need to identify the type of elements…
Q: lo How much propane (in grams) would have to be combusted to increase the temperature 300mL of water…
A:
Q: Both monomers needed for the synthesis of Nylon 6,6 can be prepared from 1,4- dichlorobutane. How…
A: Alkyl halide reacts with KCN to give alkyl nitrile. Nitrile group can be reduced by Na-ethanol or…
Q: When 18.0 mL of a 4.50x10-5 M ammonium fluoride solution is combined with 12.0 mL of a 7.34x10-4 M…
A:
Give detailed Solution with explanation needed..please explain
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The compound below is treated with N-bromosuccinimide (NBS) in the presence of light. Draw both resonance structures for the radical produced by reaction of the compound with a bromine atom. Assume reaction occurs only at the weakest C-H bond.What is mechanism of ether cleavage Is this cleavage following SN1 or SN2 ? Give your explanation.Of the two molecules below select which molecule would be predicted to react faster via an E2 elimination. (hint: stereoelectronic arguments can help)
- Name the kind of sigmatropic rearrangement that occurs in each of the following reactions. a. Using arrows, show the electron rearrangement that takes place in each reaction.Provide the complete mechanism using Curved Arrow Formalism for the reaction of 1-chloro-1-methylcyclopentane treated with NaOMe in MeOH. If more than one product is formed, which is the major, minor, and very minor product? Is the reaction SN1, SN2, E1, or E2. I appreciate the help on this question.Writing down all the relevant resonant structures and that of the resonance hybrid, propose a mechanism that explains the reason why methyl benzoate is nitrated in the meta position.
- Draw a detailed mechanism for the FeBr3@catalyzed reaction of ethylbenzene with bromine, and show why the sigma complex (and the transition state leading to it) is lower in energy for substitution at the ortho and para positions than it is for substitution at the meta position.Pentene is a most elusive molecule that has never been isolated before. However, the pentalene dianion is well known and very stable. Both compounds are polar. Use all four stipulations of Huckels rule to explain these stabilityIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1- phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Whichproduct predominates—the product of inversion or the product of retention of conguration? (c) Suggest an explanation for this phenomenon.
- 3c)Referring to the intermediates you drew in problem below explain in detail why no meta product is obtained in the Friedel-Crafts alkylation of chlorobenzene. Draw all pertinent resonance structures to support your argument.please answer these questiones and explain it Hand-draw a single-headed (fish-hook) curly arrow mechanism for steps 1 to 3. Explain why the free radical formed in step 1 is relatively stable. thank youPlease answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer.

