Q: a. .14. Predict the major product of the following reaction sequence. منا. Br OH 1. CH3OH, H3O+ 2.…
A: The objective of the question is identify the major product.
Q: 2) Consider the character table for ammonia (C3v). E 2C3 (2) 30v linear, rotations quadratic A1 1 1…
A: The total number of irreducible representations for a molecule shows the symmetries of vibrations,…
Q: In a titration of 28.0 mL of a 0.425 M solution of a diprotic acid H₂C₄H₄O₆ (tartaric acid) with…
A: The objective of this question is to calculate the volume of base (LiOH) required to reach the first…
Q: predict the product. make sure to draw the products
A: Given are organic reactions. The given reactions are condensation reactions. The starting compounds…
Q: The saponification of ethyl acetate with sodium hydroxide proceeds with second- order kinetics. At…
A: The problem describes a second-order reaction for the saponification of ethyl acetate with sodium…
Q: 4.36 Give the IUPAC name for each compound. a. C. b. d.
A: Note:- “Since you have posted a question with multiple sub parts, we willprovide the solution only…
Q: 4) List at least four reduction reactions that have different functional groups.
A: reduction reactionsReduction of Aldehydes to Primary Alcohols:RCHO (aldehyde) + NaBH₄ → RCH₂OH…
Q: Do not use chatgpt.
A: 1. Type of inhibition:Based on the Km and Vmax values obtained, the inhibitor A is a Non-competitive…
Q: Consider the following reactions: A ⇌ B, K₁=6.31 A ⇌ C, K₂=2.00 What is K for the reaction C ⇌ B?
A: The objective of the question is to find the equilibrium constant (K) for the reaction C ⇌ B using…
Q: What two sets of reagents (each consisting of a carbonyl compound and phosphonium ylide) can be used…
A: Wittig reaction is the substitution of C=C for a C=O bond.It is olefination reaction of aldehyde and…
Q: Draw structural formulas for an aldehyde or ketone and alkyl (or aryl) bromide that could be used in…
A: Retrosynthesis is a technique used in organic chemistry to plan the synthesis of complex molecules…
Q: What quantity in moles of CH₃NH₃Cl need to be added to 200.0 mL of a 0.500 M solution of CH₃NH₂ (Kb…
A: The objective of this question is to find out the quantity in moles of CH3NH3Cl that needs to be…
Q: 2A. Provide a mechanism for the following reaction. 2B. Fill in the bubbles consistent with the…
A: The objective of the question is to provide a mechanism for the given reaction, fill in the bubbles…
Q: Using the table of average bond Enthalpies below, calculate the AH for the following gas-phase…
A: Required formula to calculate the change in bond enthalpy,Here, B.E. is the bond energy and ai and…
Q: Do not use chat gpt
A: The solution contains several steps.Explanation:Step 1: Step 2: Step 3: Step 4:
Q: A solution of Na2CO3 is added dropwise to a solution that contains 1.19x10-2 M Fe3+ and 1.59x10-2 M…
A: Given: Concentration of Fe2+ ions = 1.19 X 10-2 MAnd concentration of Cd2+ ions = 1.59 X 10-2 M
Q: The ionic radii of element E and a different metallic element, M, are shown in the following table:…
A: The objective of the question is to determine whether the lattice energy of MO would be less than,…
Q: 16-138 Draw the structural formula for each of the following thioesters. a. Methyl thiopropanoate b.…
A: Given are organic compounds. Given compounds are thioesters.Thioesters are sulfur analogues of…
Q: Use bond energies (see table) to predict AH for the formation of HCI gas. H2(g) + Cl2(9) → 2 HCl(g)…
A: The objective of the question is to calculate the enthalpy change (ΔH) for the formation of HCl gas…
Q: mechanisms for the following reactions. (a) (b) (d) (f) ropose mechanisms for the following…
A:
Q: Identify the correct name or abbreviation for the given nucleoside or nucleotide. deoxyguanosine…
A:
Q: Write and balance the chemical equation where KOH and HCL reacts.
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: By titration, 15.0 mL of 0.1008 M sodium hydroxide is needed to neutralize a 0.1331 g sample of a…
A: An empirical formula represents the simplest whole-number ratio of various atoms present in a…
Q: At 27 °C, Kp = 1.5 × 1018 for the reaction 3NO(g) N₂O(g) + NO2(g) If 0.030 mol of NO were placed in…
A: The objective of the question is to find the equilibrium concentrations of NO, N₂O, and NO2 for the…
Q: Question 32 For the compound below determine which of the circled protons (H atoms) is more acidic…
A: Acidity is defined as the ability of a molecule to release protons.If a compound easily donates…
Q: A student placed 0.410 mol of N2O4(g) into a 1.9 L container at 21.1 °C. At equilibrium it was found…
A:
Q: Predict chemical shifts for all of the protons in the molecule below. Cl -Cl нь OH
A: Proton-NMR spectroscopy is mainly used for identification of complete structure of organic…
Q: Predict the net direction of the following reaction (Complete the reaction by writing the conjugate…
A: A. The net direction is to the rightExplanation:Let's analyze the reaction and identify the…
Q: Reaction of p-nitroaniline with sodium nitrite and hydrochloric acid at 0°C, followed by treatment…
A: The objective of teh question is to predict the product formed in the following reaction given.
Q: Identify how many N (NITROGEN) atoms have sp² hybridization for the compound below: A B 1 (one) sp²…
A: sp hybridization is observed when one s and one p orbital in the same main shell of an atom mix to…
Q: Determination of the Enthalpy
A: The objective of this question seems to be to determine the enthalpy change associated with the…
Q: What's the IUPAC name for this structure?
A:
Q: Enthalpy calculations for alcohol Initial mass burner (g) Initial temperature H2O(°C) Final…
A: The objective of the question is to calculate the-Moles of the fuel burnedEnthalpy change per…
Q: Question 4 Determine the position of equilibrium for the ACID-BASE reaction below: H ? A: cannot…
A: Whether equilibrium lies on the right side or left side depends upon the acidity of the reactant and…
Q: Using this data, 2NO(g) + Cl2(g)=2NOCI(g) KC = 2.39 × 10-3 2NO2(g) 2NO(g) + O2(g) KC = 11.2…
A: If the equation (having equilibrium constant K) is multiplied by 2, the equilibrium constant for the…
Q: Mechanism 2. Provide the complete mechanism for the reactions below. You must include appropriate…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Aluminum Chloride + Sulfuric Acid ?
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Consider the titration of a 60.0 mL of 0.291 M weak acid HA (Ka = 4.2 x 106) with 0.400 M KOH. After…
A: The titration reaction taking place can be written as,=> HA + KOH ------> KA + H2OGiven :…
Q: What is the oxidation product of m-methylbenzaldehyde with K2Cr2O7?
A: The objective of the question is to determine the product of the oxidation reaction of…
Q: using this CNMR and HNMR how can it be confirmed that aspirin has been formed?
A: Aspirin is a drug that is widely used for the treatment of fever, and to reduce body pain. Aspirin…
Q: I and II are: I FL constitutional isomers. O enantiomers. O identical. O diastereomers. O not…
A: Isomers are compounds that have same molecular formula but different structural formula.…
Q: Calculate the concentration of all species (H3PO4, H2PO4-) and the pH of a 0.200 M solution of…
A: 1. The concentration of H3PO4 is 0.165 M.2. The concentration of H2PO4− is 0.035 M.3. The pH of the…
Q: A sample of 7.63 g of Mg(OH)2 is added to 24.0 mL of 0.175 M HNO3. Part A Enter the chemical…
A: Information about the question
Q: Provide the structure of the major organic product in the reaction below. H w 1. CHI (excess) CH3 2.…
A: Synthesis of quaternary amineHoffmann elimination
Q: Draw a skeletal ("line") structure of this molecule: O CH3 CH3-C-N-CH2-C-CH3 Click and drag to start…
A: Given,The molecule is:
Q: A 1.00 liter solution contains 0.44 moles hydrofluoric acid and 0.34 moles sodium fluoride. If 0.17…
A: Mixture contains weak acid and corresponding salt then mixture is buffer solution.Henderson equation…
Q: : :G: G-Pn-G: :G: Determine its VSEPR shape. [Select] Determine its molecular shape. [Select]…
A: The objective of the question is to determine the VSEPR shape, molecular shape, hybridization on the…
Q: Draw the organic product you would expect to isolate from the nucleophilic substitution reaction…
A: Given reaction is an example of nucleophilic substitution reaction.The substitution reaction is a…
Q: Only typed solution
A: The objective of this question is to calculate the pH at the equivalence point of a titration…
Q: Reaction of N,N-diethyl-p-diaminobenzene with sodium nitrite and hydrochloric acid at 0°C, followed…
A: Formation of azo-dye Solution are here in the given picture Thank you so much Explanation:Step 1:…
Which rxn would be fastest? Which would be slowest?
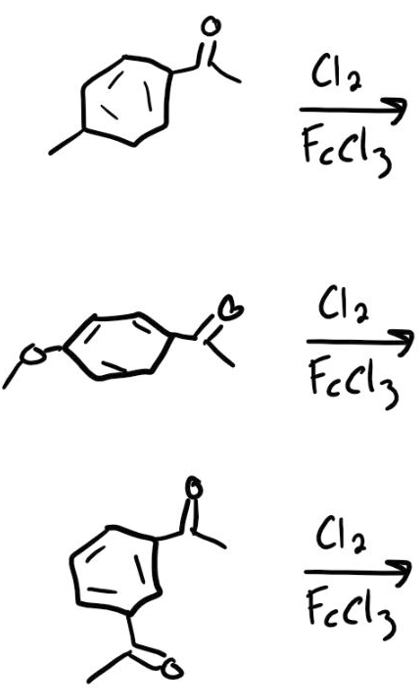
Step by step
Solved in 1 steps




