Chapter12: Unsaturated Hydrocarbons
Section: Chapter Questions
Problem 12.56E
Related questions
Question
help me with no. 6 and 7 please
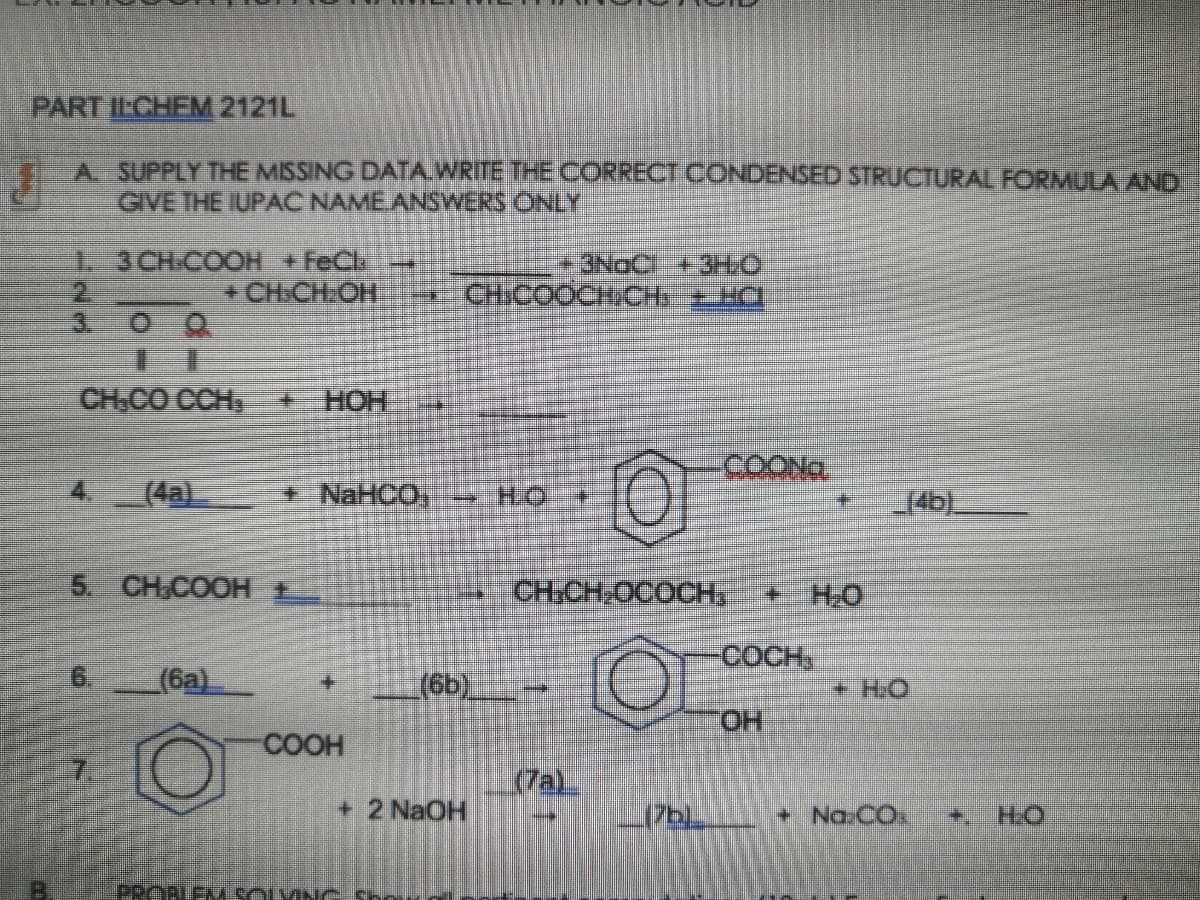
Transcribed Image Text:PART ILCHEM 2121L
A SUPPLY THE MISSING DATA.WRITE THE CORRECT CONDENSED STRUCTURAL FORMULA AND
GIVE THE IUPAC NAMEANSWERS ONLY
1-3CH-COOH +FeCl
+CH.CH:OH
3NOC + 3HO
* CHICOOCH/CH+ HC
3.
CH.CO CCH
HOH +
COONG
(4a)
NaHCO, HO
14b)
5. CH,COOH E
CH:CH,OCOCH,
+H.O
COCH,
(6a)
(6b).
+HO
ОН
COOH
(7a)
+ 2 NaOH
+ Na.CO.
+.
H.O
PROBLEMSOLVING Shou
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

