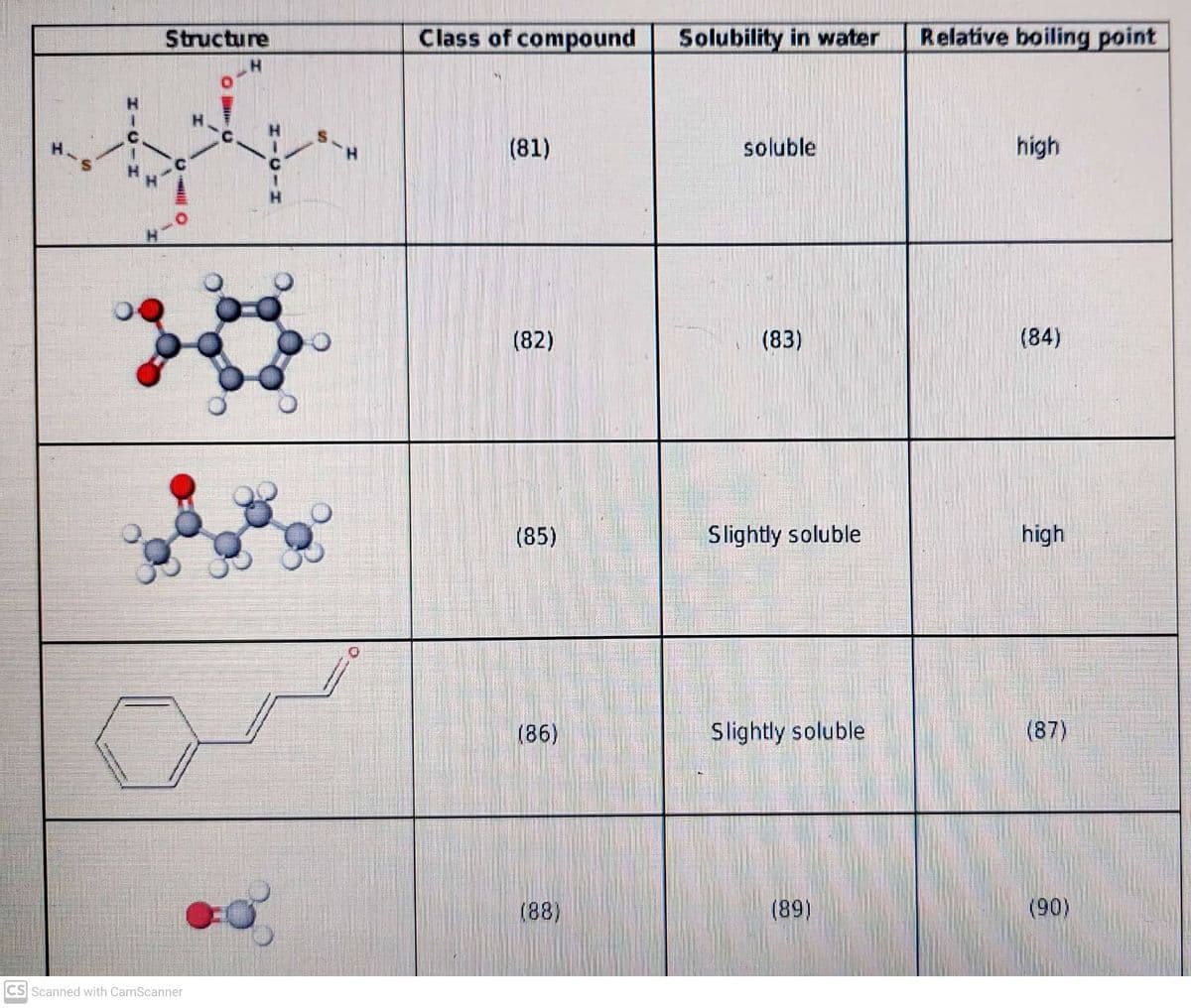
Complete the table below with the following choices. Answer those with numbers 81 to 90. A. Aldehyde B. Ketone C. Carboxylic Acid D. Thiol E. None of these
Q: //Ethane (CnH2n+2 ) is burned with atmospheric air. The analysis of the products on a dry basis is…
A: Given in following question ethene is burned with atmospheric air given 1.CO2 =9.7% ,O2=1.7%…
Q: 49. Which of the following statements is true about phenols? A. Phenols has a lower boiling point…
A:
Q: Choose the correct shape, weak or strong field, and number of unpaired electrons for…
A:
Q: 44. What is the correct order of reactivity of CH3CHO, C₂H5COCH3 and CH3COCH 3? A. CH3CHO >CH3COCH3…
A: Hi! Thank you for the question As per the honor code, We’ll answer the first question since the…
Q: + CO2(g) is heated from 25C to 500°c. Calculate the molar enthalpy of formation for as a real gas,…
A:
Q: The KSP of Agl is 8.50x1017. Find the reduction potential of a saturated Agl solution to Ag(0). Ered…
A:
Q: Consider this reaction Fe + MnO4 - → FeO4 -2 + Mn +4 What is the correct reduction…
A:
Q: [According to Dalton: Atoms of a given element are identical O True O False
A: According to the Dalton atomic theory, the atom is the smallest particle of a substance and it…
Q: Draw 3-D Lewis structure of PO-3
A: We follow the below steps to draw Lewis diagram: Step-1: Find the total valence electrons present in…
Q: 6 Give the reaction mechanism (showing curly arrows) for the bromination of methane in the presence…
A:
Q: 23. A 0.0035 M aqueous solution of a particular compound has a pH = 2.46. The compound is: Select…
A:
Q: The following half-reactions were combined to construct a new galvanic cell. What is the standard…
A: Given-> [AuBr4]-(aq) + 3e- --------> Au(s) + 4Br-(aq). E°=0.854 V Au3+(aq) + 3e- ------->…
Q: What is the pH of an aqueous solution at 25.0°C containing hydronium ions H+? Select one: a. 7.04…
A: To answer this question we have to know the pKw of water at 25.0 oC. The pKw at this temperature is…
Q: 1. Give a mechanism for the below reaction Na CO₂Me Me3SICI CO₂Me OSiMe3 OSiMe3
A: Since, this question have multiple questions. So, I will solved first question ( 1 ) for you. You…
Q: What is the pH of a 0.015 M aqueous solution of barium hydroxide? Select one: a. 1.52 b. 12.25 c.…
A: We are given: Concentration of Ba(OH)2 = 0.015 M
Q: Draw structural for n-octane.
A:
Q: Find the number of molecules in 7 mols of NaBr
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: 75
A:
Q: State whether the following analysis is quantitative or qualitative analysis or both. i)…
A: In qualitative chemical analysis, we identify whether a chemical substance of interest is present in…
Q: Organic chemistry refer to.1 A. Study carbon compounds. O B. Study carbon and hydrogen compounds C.…
A: Please refer to below answer.
Q: 2.Organic compounds are divide to OA - Aromatic hydrocarbons. OB- Aliphatic hydrocarbons. OC -…
A: Given, organic compound are divide to, Aromatic hydrocarbons Aliphatic hydrocarbons Aromatic and…
Q: Which of the following compounds is expected to have the lowest K₁ (complex dissociation constant)?…
A: ✓ A spectrochemical series is the arrangement of common ligands in the increasing order of their…
Q: Observe the following reaction and answer items 73 to 80. H 6= 0=$=0 он ? Ho 73. What is the main…
A: Organic reaction mechanisms: As per our guideline we have to answer first question only.
Q: Consider this reaction Fe + MnO4 - → FeO4 -2 + Mn +4 What is the correct reduction…
A:
Q: 4- General formula of aromatic compounds are. OA - CnH2n. O B-CnHn+2. OC - CnH2n-2.
A: 4- General formula of aromatic compounds is :
Q: 52. Ethers are commonly used as a solvent in chemical reactions due to? A. Ethers are susceptible to…
A: Ether is commonly used as solvent in chemical reactions due to ethers are succeptible to chemical…
Q: Draw all the possible stereoisomers for [Pd(CN)2(en)2].
A:
Q: * Calculate the Molarity of solution have 20 g of Glucose sugar C12H22012 Dissolved in 125 g of…
A:
Q: Give me handwritten answer
A:
Q: An aromatic ring should satisfy Huckel’s rule, wherein the number of electrons participating in the…
A:
Q: Considering the standard reduction potential for Tin (see below), what is the Eored for the…
A: Generally we will consider reduction potential values at 298K and for 1M concentration are standard…
Q: Can you relate it to activated carbon? When activated carbon is acid treated with sulfuric acid.…
A: Sulfonation: The chemical reaction in which the sulfonic acid, sulfonyl chlorides and its…
Q: 67. Ethanol is heated with an acidified potassium manganate (VII) solution. What would be the…
A: Option A is correct. Because acidified pottasium permanganate oxidizes primary alcohol to aldehyde…
Q: Br2 H₂O
A:
Q: What is the main functional group of the organic molecule? Carbonyl functional group Aldehyde…
A: Here molecule is CH3CH(OH) CH2CH3. It is a organic molecular which contain a funtional group.
Q: I. Each -OH group of an alcohol can stabilize four to five carbon atoms. II. The boiling point of…
A: The stability of alcohols is guided by the stability of the carbon skeleton and the polarity of C-O…
Q: Given the reaction: Cu + Ag+ → Cu+ + Ag The reaction is assumed to be that (Cu) = (Ag1+) = 1.0M…
A:
Q: What is the reaction rate constant given the plot of the reaction of reactant A with respect to time…
A: here we are required to find the rate constant for the first order reaction
Q: Rank the following compounds in order of increasing basicity: NH₂ NH₂ NH₂ NH₂ A) B) D) OCH3 NO₂ 2
A:
Q: A flask is charged with 0.124 mol of A and allowed to react to form B according to the reaction A(g)…
A: A(g) ....> B(g) Moles of A at 10s = 0.110 Moles of B at 20s = 0.088
Q: 5. The overall order for a reaction is 2. The units of the rate constant for the reaction will be:…
A: The sum of the exponent term in the rate law for a chemical reaction is known as the order of the…
Q: What is the correct name for the following compound? H.C. Select one: O a. m-ethylacetophenone Ob.…
A: We have name the given compound.
Q: What is the correct name for the following compound? H.C. Select one: a. m-ethylacetophenone b.…
A: Benzene ring substituted with -COCH3 is named acetophenone. In case of doubly substituted benzene…
Q: 2.Organic compounds are divide to OA - Aromatic hydrocarbons. OB-Aliphatic hydrocarbons. OC -…
A: Organic compounds are mainly divided into two catagory - cyclic compounds and acyclic or open chain…
Q: Consider the complexes d bis(ethylenediamine) nickel(II), [Ni(en)₂]+², versus tetracyanidoferrate…
A: Paramagnetic substances have unpaired electrons and diamagnetic substances have paired elections.
Q: 1.Organic chemistry refer to O A. Study carbon compounds. OB. Study carbon and hydrogen compounds…
A:
Q: Intermolecular means Hydrogen bonds can occur among molecules True False
A:
Q: 1. The solubility of monocarboxylic acid in water increases as the alkyl chain increases in length.…
A: Here we have to predict whether the given statements are true or false.
Q: 32. The most common reaction involving the carboxylic acids and its derivatives is related to the…
A: The structure of a carboxylic acid functional group is as follows: In the carboxylic acid…
Q: If the only bonds broken during glucose oxidation were the carbon-carbon bonds, how much energy on…
A: (1) In a 6 carbon sugar, there are 5 carbon-carbon bonds. 673 kcal/mole divided by 5 equals…
Complete the table below with the following choices. Answer those with numbers 81 to 90.
A.
B.
C. Carboxylic Acid
D. Thiol
E. None of these
F. Solube
G. Insoluble
H. High
I. Low
Step by step
Solved in 2 steps with 1 images

- There are a number of terms used in conformational analysis. Match each term below with the appropriate definition.Can facial and meridional isomers be chiral? justifyI had wrote constitutional isomers, since it seemed to me that they were connected differeently, yet my teacher said they were diastereomers. How?
- D-Xylose in aqueous solution exists in the pyranose form as a mixture of α and β anomers. Furanose and acyclic forms are present in negligible amounts and can be ignored. A sample of pure α-D-xylopyranose is dissolved in pure neutral water. The optical rotation is then measured immediately using a polarimeter. The specific rotation [??]?? is +93.6°. The optical rotation is then observed to decrease with the passage of time, and the solution is allowed to reach equilibrium. Eventually the specific rotation stabilises at [??]?? = +18.8°. The anomeric ratio at equilibrium a : β is 37 : 63. A sample of pure β-D-xylopyranose is not available. Using this information, predict the specific rotation of pure β-D-xylopyranose. Show how the a and β anomers equilibrate using a mechanism, assuming the presence of a trace amount of base as catalyst.give a name of any compound which is isostructural with XeF2Indicate (no more than 2) for the following compound:HomotopicHeterotopicEnantiotopicDiastereotopic
- An unknown sugar is known to equilibrate between two forms, I and II, in aqueous solution. Thisequilibration can be followed by measuring the optical rotation of aqueous solutions of isolatedsamples of form I and II. When species I was dissolved in pure water and the optical rotation was measured, it gave aspecific rotation of [?] = +42.0°. Over time, this value decreased to a constant value of [?]= +0.2°. • When species II was dissolved in pure water and the optical rotation was measured, it gave aspecific rotation of [?] = -54.0°. Over time, this value decreased to constant value of [?]= +0.2°. • In each case, both forms I and II were shown to be present in the mixture at the end of theexperiment. Calculate the ratio of the two forms I and II present at equilibrium and comment on anyassumptions made in making that calculation.Pls explain chair conformationsConsidering the following structures (I~V), answer the questions A-J. There may be more than one answer for a given question
- Given the information listed below, identify the compound. Physical description: This substance is a colorless but fragrant liquid that is oily in appearance. Chemical information: Observed Melting point: -78.5 °C Observed Boiling point: 138-142 °C Calculated MW: 130 g/mol Density: 0.876 g/cm3 Solubility in water: Minimally soluble, Other: Flash Point: 77 °F Spectroscopic information: IR bands indicate this compound contains C-H functionalities and the appearance of a strong stretch near 1700 cm-1 as well as several strong bands between 1500 and 1000 cm-1. The C-13 NMR spectrum for this compound indicates 6 unique carbon atoms and no cyclic structure. The proton NMR data indicate 5 sets of unique protons. C-13 NMR data of : PPM: 171.08, 63.13, 37.48, 25.18, 22.51, 20.96 Int: 222, 887, 645, 635, 1000, 473 Assign: 1, 2, 3, 4, 5, 6 H-1 NMR Assign: A, B, C, D, E Shift: 4.095, 2.037, 1.693, 1.521, 0.926Fill the empty boxes with compatible structure. (Give answer in clear handwritten)pls show the the isomers of butane-2,3-diol. Which pairs are enantiomers? diastereomers? mesomers? pls explain why for each

