Q: 3. Write the formula for the following hydrates. a nickel(II) chloride hexahydrate b. cobalt(II)…
A: 3.a [Ni(H2O)6]Cl2
Q: Which of the following represents the hydroxide ion? O H20 O H+ O OH O H30*
A:
Q: Write the correct formula for each of the following:a. manganese(II) oxideb. manganese(III) oxide
A: The molecular formula for manganese(II) oxide is MnO.
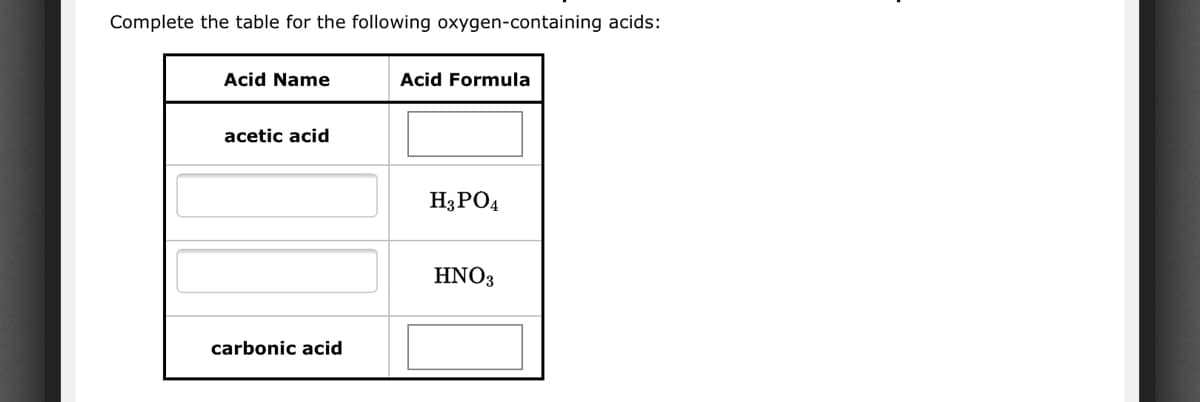
Q: Give the name and formula for the acid derived from each of the following anions: (a) hydrogen…
A: The name and formula for the acid derived from given anions has to be given.
Q: Write the formula for each of the following compounds containing a semimetal. (a) boron tribromide…
A: The formula for the given compounds containing a semimetal has to be given.
Q: How many valence electrons do each of the following molecules contain? Bromomethane (CH3B1) Sulfate…
A: Interpretation: We have to calculate the electrons present in the given organic molecules.
Q: Provide the formula for each of the following binary ionic compounds. (a) cuprous phosphide (b)…
A: The formula for the given compound has to be given,
Q: Give the name and formula for the acid derived from each of the following anions: (a) perchlorate…
A: Hi, since you have posted questions with multiple subparts. We will answer the first three subparts.…
Q: NaHCO3 Mg(HCO,)2 4 Magnesium phosphate 1 Sodium 2 phosphate 1. Sodium carbonate; 2. Magnesium…
A:
Q: Write the name / formula for the given compounds: a) AlH3 ________________________
A: To find: The name of AlH3.
Q: Match the acid formula with its correct name HNO3 1. hydrochloric acid HCIO2 2. nitrous acid HNO2 3.…
A: The formula of different acids given, will be follows:
Q: If provided the name, then you must write the formula. If provided the formula, then you must write…
A: IUPAC naming is world wide acceptable method for naming of organic and Inorganic compound , there…
Q: What is the correct formula for sodium phosphate? (a) Na3PO4(b) Na3P(c) NaPO4(d) Na(PO4)2
A: The correct formula for sodium phosphate has to be given
Q: Consider the newly discovered polyatomic ion Hypobuffite: BfO3-. What is the name of the acid…
A: We have to name the acid H3BfO4 by considering the newly discovered polyatomic ion Hypobuffite:…
Q: Fill in the blanks to complete the following table of chemical formulas for…
A: The formula for an ionic compound that contains Ax+ cation and By- anion, can be written as:…
Q: Table D2: Names of Ionic Compounds, Give the IUPAC (official) name of each of the following ionic…
A: To give the IUPAC name to the given compund , to have to the see the number of different atoms and…
Q: Write a chemical formula for each of the following substances: (Write the answer ONLY) (a)…
A: Since you have posted multiple subparts, the answer for first three subparts are given below. Kindly…
Q: Give the IUPAC name of each ionic compound. Formula Name CaO Al(OH)3 FeCl3 Na3N
A: The question is based on naming of the compounds. we have to systematically write the name of the…
Q: Table 2: Identify the following as ionic or covalent (molecular) compounds. Provide the correct…
A: Compounds can be covalent ionic it's depends on the cation and anion
Q: A What is the correct formula that would result from the combination of the two ionic species? NH₂¹+…
A: According to the question we have two ions which are NH4+ and OH- NH4+ is known as ammonium ion -…
Q: A formula unit of ammonium sulfate consists of ________ ammonium ions
A: Write the formula of ammonium sulfate by using the ionic symbols of NH4+ ammonium ion and SO42-…
Q: Write the formula for the lonic compound formed from the following pairs of ions: (a) Iron(II) ion…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: If the gas HLe(g) is given the name "hydrogen leilide," what is the name of the binary acid HLe(aq)…
A:
Q: 1. In your home life and your chosen careers, you will encounter many ionic compounds. Some of these…
A: Drug for cough and asthma:
Q: Which of the following bonds has the most covalent property? C-Cl S-Cl H-Cl Mg-Cl Na-Cl
A: Covalent bond is formed between two atoms by mutual contribution and sharing of one or electrons.…
Q: Complete the table for the following covalent compounds: Formula Name CO N2F4 disulfur decafluoride…
A: The name of the compound is given by the number of atoms present in the compound. According to the…
Q: Directions: ONLY HANDWRITE YOUR ANSWERS IN THE SPACE PROVIDED and upload all pages as ONE unit as a…
A:
Q: order each list of elements in the table below, if necessary, so that the elements are listed in…
A: Electronegativity is defined as the ability of an atom to attract shared pair of electron in a…
Q: Which of the following naming systems would you use to name. H2 SO4? O a) OUS ACID O b) HYDRO IC…
A: Given Formula of Acid = H2SO4 Name = ?
Q: Predict the chemical formula for the ionic compound formed by Au³⁺ and HSO₃⁻
A: While writing the formula of ionic compound, number of positive charge should be equal to the number…
Q: Draw the structural formula of the following compounds . Element element, ion or ionic group…
A: When a compound is formed if it covalent then octet is completer and when ionic electrons are…
Q: Compare the following: Acid 1: acetic acid , CH3COOH Acid 2: hydrogen carbonate ion , HCO3 Acid 3:…
A: Acids are those which are capable of donating (proton) H+ ion. Strong acids get completely ionized…
Q: Write the chemical formula of the following. A step by step. A. Calcium chloride B. Calcium chlorate
A: A chemical formula represents the symbols of the elements involved. The chemical formula gives…
Q: The correct name for P2O5 is Group of answer choices diphosphorus oxide phosphorus(V) oxide…
A:
Q: Which acids have names ending in “-ic"? Acid# Formula HNO2 H2CO3 3 HI 4 HC1O A Acids 1 and 4 B Acids…
A: HNO2 = Nitrous acid H2CO3 = Carbonic acid HI = Hydroiodic acid HClO = Hypochlorous acid
Q: Compare the following: Acid 1: hydrofluoric acid , HF Acid 2: hydrosulfuric acid , H2S Acid 3:…
A: Acid is a substance which donate H+ ion and form well stabilised conjugate base
Q: Classify each of the following compounds as ionic (1), molecular (M), binary acid (BA), or oxyacid…
A: In binary acids, a hydrogen atom is connected to a non-metal whereas, in oxyacids, a hydrogen atom…
Q: Write the correct formula for each of the following:a. copper(II) oxide b. iron(III) oxide
A: (a) copper (II) oxide In this case, the (II) represents the oxidation state of copper i.e. +2 in…
Q: : No charge indicated in the formula : No prefix on the first or second element : Positively charged…
A: Ionic compounds are neutral compounds made up of positively charged ions called cations and…
Q: Which of the following compound will have only covalent bonds? sodium carbonate O Magnesium oxide O…
A: In covalent compound bond are formed by sharing of electron while in ionic compounds metal…
Q: Arrange the following sets of compounds based on the property given in increasing order. Type in the…
A:
Q: Determine the chemical formula for each of the compounds. sodium sulfite: Na2SO3 potassium…
A: Sodium sulfite - Na2SO3 Potassium phosphate - K3PO4
Q: Which of the following is an example of a compound having both ionic and covalent bonds? sodium…
A: Ionic bond: The ionic bond is formed between the metal and the non-metal. The electronegativity…
Q: The formula for sodium perchlorate is NaClO4 and the formula for calcium chromate is CaCrO4. Write…
A: Given : Formulas of sodium perchlorate and calcium chromate is given. To find: Formula of given ions…
Q: Predict the chemical formula for the ionic compound formed by AU3+ and HSO3-
A: Chemical formula of an ionic compound can be formed by criss- crossing the valencies of cation and…
Q: Complete the table for the following ionic compounds: Name Formula sodium fluoride silver oxide…
A: Ionic compounds are formed by ions that are held together by attractive forces.
Q: Name acid acid or base if the formula is given, or write the formula of the acid or base if the name…
A: We are having the names or formula for acid or base we have to write the for formula for name and…
Q: Choose the correct pair of formula-name. O HP3 -Phosphorus trihydride O PH3 -Phosphorus trihydride O…
A: Every compound is named according to their formation ability i.e. the valency of the element . Here…
Q: 16. Write the formulas for the following molecular covalent compounds. (2 points each) a.…
A: Answer:- This question is answered by using the simple concept of writing the molecular formula of…
Q: What are the chemical equations of the following: CuSO4 + HCl CoCl2 +HCl Ni(NO3)2 +HCl
A:
Step by step
Solved in 2 steps

- Indicate whether each of the following statements is true or false. For each statement that is false, correct the statement to make it true. The strongest acid known is HF because fluorine is the most electronegative element.Which of the following is not an acid A) NH3 B) HC2H302 C) HF D) HCIO Complete the table. pH [H+] [OH-] pOH 7.0 7.3 7.4 7.5
- Given equal concentrations of the following acids, which exhibits the greatest amount of ionization in water?A. Ascorbic acidB. Chlorous acid C. Hydroiodic acidD. citric acidE. Nitrous acidWrite a balanced chemical equation for the following. (Assume the following are strong acids) HI + NaOH H2SO4 + NaOH H3PO4 + NaOHwhy most of acids are liquids and most of bases are solids?
- Which of the statements below would correctly explain why hydrochloric acid is a stronger acid that hydrofluoric acid? a. Fluorine is higher on the periodic table than chlorine. b. Fluorine has a smaller atomic radius than chlorine. c. Fluorine has fewer electrons than chlorine. Selected:d. Fluorine has a larger electronegativity than chlorine.write the chemical formula of the base: Cl-(aq)+HSO4-(aq) HCI(aq)+S042-(aq)Polyproptic Acid Question - Chemistry
- classify each of the following compound as either Ionic,Molecular, binary acid, oxyacid Please answer ASAPWhich of the following is a polypeotic acid? a)perchloric acid b)nitric acid c)acetic acid d) sulfurous acid Thanks for your help!Indicate whether each of the following statements is true or false. For each statement that is false, correct the statement to make it true. Hydrotelluric acid 1H2Te2 is a stronger acid than H2S because Te is more electronegative than S.



