Concentration sodium thiosulfate solution used: 0.056 mol L-¹ ACCURATE TITRATION VOLUME OF SODIUM THIOSULFATE SOLUTION USED Volume titration 1, (mL) Volume titration 2, (mL) Your Group 15.80 15.75 Group 1 16.05 15.70 Group 2 16.30 16.25 ANALYSIS Average volume used, (mL) Standard deviation Number of moles sodium thiosulfate used, (mol) Number of moles calcium iodate in solution, (mol) Volume of calcium iodate used, (ml) Concentration of saturated calcium iodate solution (mol L-¹) Unrounded value 15.9750 ✓ 0.26220 0.00089400 ✔ ✔ 0.00036700 A 6 Rounded value 15.97 0.262 8.94 x 104 10.0
Concentration sodium thiosulfate solution used: 0.056 mol L-¹ ACCURATE TITRATION VOLUME OF SODIUM THIOSULFATE SOLUTION USED Volume titration 1, (mL) Volume titration 2, (mL) Your Group 15.80 15.75 Group 1 16.05 15.70 Group 2 16.30 16.25 ANALYSIS Average volume used, (mL) Standard deviation Number of moles sodium thiosulfate used, (mol) Number of moles calcium iodate in solution, (mol) Volume of calcium iodate used, (ml) Concentration of saturated calcium iodate solution (mol L-¹) Unrounded value 15.9750 ✓ 0.26220 0.00089400 ✔ ✔ 0.00036700 A 6 Rounded value 15.97 0.262 8.94 x 104 10.0
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.11QAP
Related questions
Question
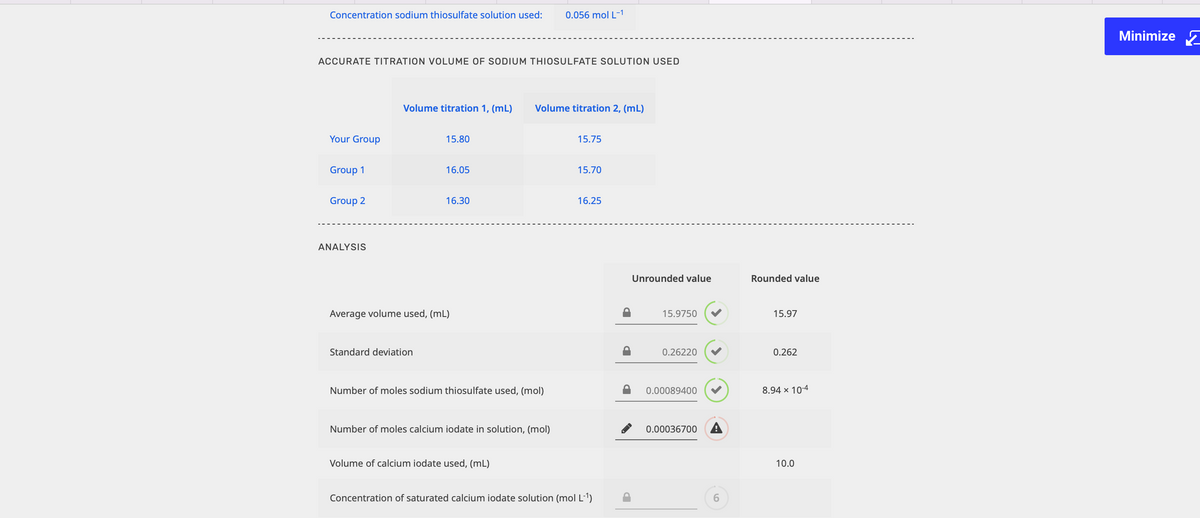
Transcribed Image Text:Concentration sodium thiosulfate solution used:
0.056 mol L-1
ACCURATE TITRATION VOLUME OF SODIUM THIOSULFATE SOLUTION USED
Volume titration 1, (mL)
Volume titration 2, (mL)
Your Group
15.80
15.75
Group 1
16.05
15.70
Group 2
16.30
16.25
ANALYSIS
Average volume used, (ml)
Standard deviation
Number of moles sodium thiosulfate used, (mol)
Number of moles calcium iodate in solution, (mol)
Volume of calcium iodate used, (mL)
Concentration of saturated calcium iodate solution (mol L-¹)
Unrounded value
15.9750
0.26220
0.00089400
0.00036700 A
Rounded value
15.97
0.262
8.94 x 10-4
10.0
Minimize
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning