Consider an element that reaches its first excited state by absorption of 480.4 nm light. Determine the energy difference in kilojoules per mole between the ground state and the first excited state. AE = 248.2 kJ/mol If the degeneracies of the two states for the element are g*/go = 3., determine N'/No at 2070 K. N° 1.63 x10-6 No Incorrect By what percentage does N'INo change if the temperature is raised by 20 K? percentage: Incorrect What is N*/No at 5620 K? N° No ancorrect
Consider an element that reaches its first excited state by absorption of 480.4 nm light. Determine the energy difference in kilojoules per mole between the ground state and the first excited state. AE = 248.2 kJ/mol If the degeneracies of the two states for the element are g*/go = 3., determine N'/No at 2070 K. N° 1.63 x10-6 No Incorrect By what percentage does N'INo change if the temperature is raised by 20 K? percentage: Incorrect What is N*/No at 5620 K? N° No ancorrect
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter8: An Introduction To Optical Atomic Spectrometry
Section: Chapter Questions
Problem 8.8QAP: The Doppler effect is one of the sources of the line broadening in atomic absorption spectroscopy....
Related questions
Question
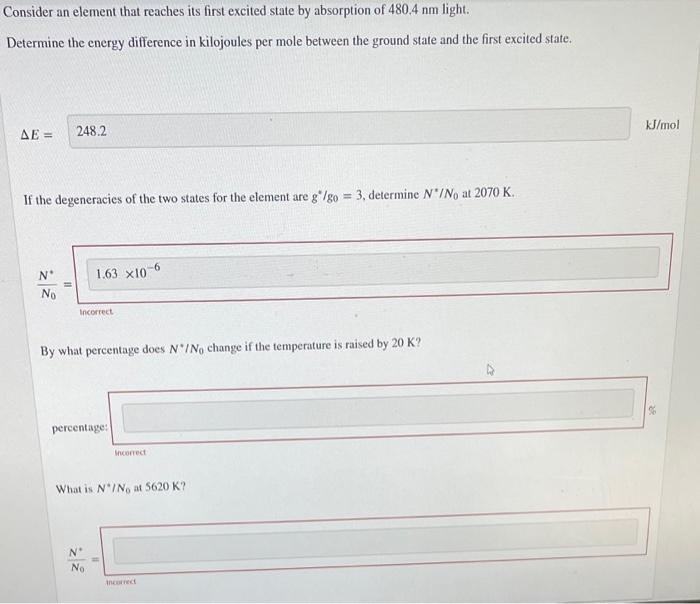
Transcribed Image Text:Consider an element that reaches its first excited state by absorption of 480.4 nm light.
Determine the energy difference in kilojoules per mole between the ground state and the first excited state.
AE =
248.2
kJ/mol
If the degeneracies of the two states for the element are g*/go = 3., determine N'/No at 2070 K.
N°
1.63 x10-6
No
Incorrect
By what percentage does N'INo change if the temperature is raised by 20 K?
percentage:
Incorrect
What is N*/No at 5620 K?
N°
No
ancorrect
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning