Consider the following data for osmium: 190.23 mol atomic mass electronegativity 2.20 kJ 106.1 mol electron affinity kJ 840. mol ionization energy kJ 31. mol heat of fusion You may find additional useful data in the ALEKS Data tab. Does the following reaction absorb or release energy? release absorb (1) Os (g) Os(g) + e Can't be decided with the data given. yes Is it possible to calculate the amount of energy absorbed or released by reaction (1) using only the data above? no If you answered yes to the previous question, enter the amount of energy absorbed or released by reaction (1): |kJ/mol
Consider the following data for osmium: 190.23 mol atomic mass electronegativity 2.20 kJ 106.1 mol electron affinity kJ 840. mol ionization energy kJ 31. mol heat of fusion You may find additional useful data in the ALEKS Data tab. Does the following reaction absorb or release energy? release absorb (1) Os (g) Os(g) + e Can't be decided with the data given. yes Is it possible to calculate the amount of energy absorbed or released by reaction (1) using only the data above? no If you answered yes to the previous question, enter the amount of energy absorbed or released by reaction (1): |kJ/mol
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter7: Chemical Bonding And Molecular Structure
Section: Chapter Questions
Problem 7.86PAE: 7.86 Nitrogen triiodide, NI3(s) , is unstable and will spontaneously detonate to form a bright...
Related questions
Question
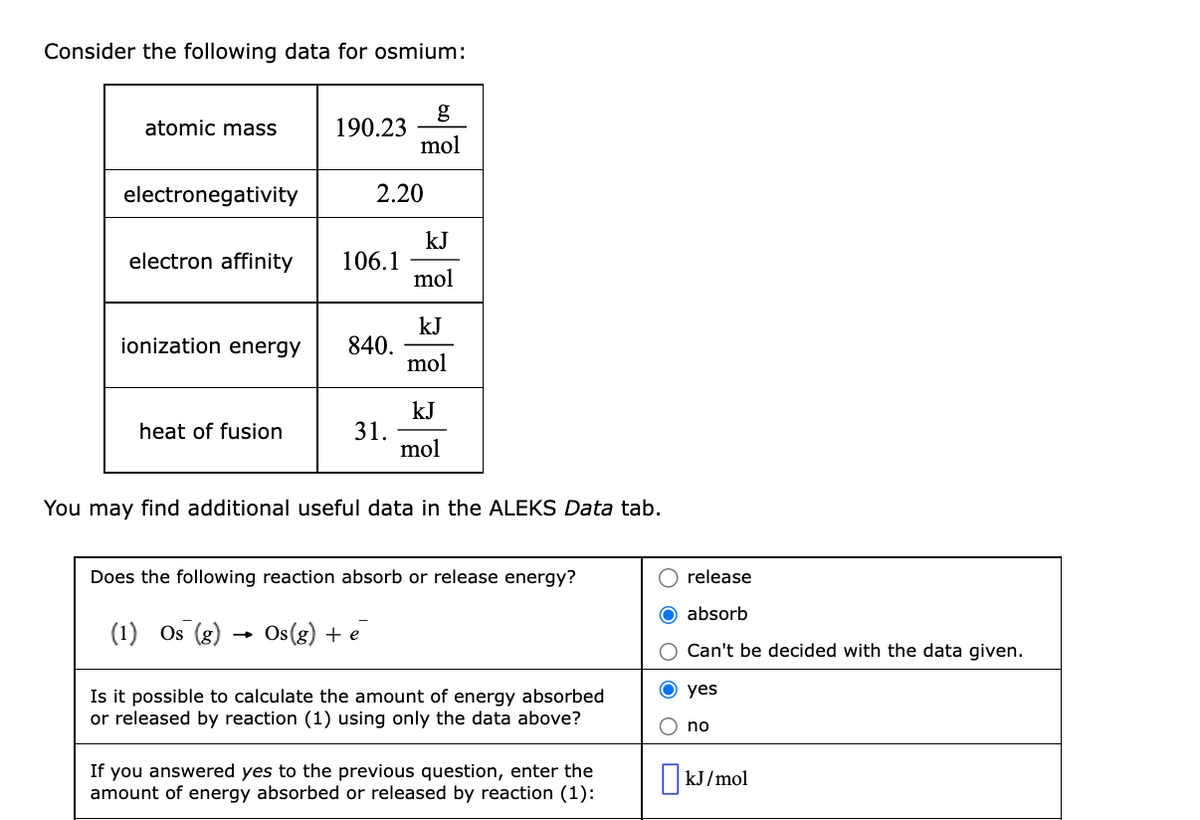
Transcribed Image Text:Consider the following data for osmium:
g
190.23
mol
atomic mass
electronegativity
2.20
kJ
106.1
mol
electron affinity
kJ
840.
mol
jonization energy
kJ
31.
mol
heat of fusion
You may find additional useful data in the ALEKS Data tab.
Does the following reaction absorb or release energy?
release
O absorb
(1) Os (g)
Os (g) + e
Can't be decided with the data given.
yes
Is it possible to calculate the amount of energy absorbed
or released by reaction (1) using only the data above?
O no
If you answered yes to the previous question, enter the
amount of energy absorbed or released by reaction (1):
|kJ/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning