Q: Describe principle of a glass membrane pH electrode.
A: A glass membrane pH electrode is a type of sensor used to measure the acidity or alkalinity (pH) of…
Q: O H P !!!( O
A: To calculate the formal charge of HPO3 2-, we need to first determine the valence electrons of each…
Q: In the laboratory you are given the task of separating Ba2+ and Fe²+ ions in aqueous solution. For…
A: HBr Can be used to separate the ions: Y Formula of the precipitate: BaBr2 (barium bromide) Ba2+…
Q: Describe the difference in structure between the male hormone testosterone and the female hormone he…
A: The main hormones that regulate male and female sexual characteristics are testosterone and…
Q: Calculate the volume of 0.830-M NaOH solution needed to completely neutralize 30.7 mL of a 0.890-M…
A: Answer: This question is based on stochiometric calculation where by using stochiometric…
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 1.03kg sample…
A: We have to calculate the specific heat capacity of the substance
Q: A model of a cycloalkane appears in the window below. 00 ball & stick 0 CH₂ Which of the following…
A: Answer: When two compounds have same molecular formula but different structural formulas the such…
Q: How many moles of ibuprofen contain 1.62 x 10^23 atoms of C?
A: To find the number of moles of ibuprofen that has the given number of carbon atoms.
Q: A portion of the citric acid cycle is shown below. What are the identities of A and B? citrate →…
A: Citric acid cycle consists of a series of closed loop in which the given portion is Citrate…
Q: Calculate the pH for each case in the titration of 50.0 mL of 0.220 M HClO(aq)0.220 M HClO(aq) with…
A: In the multisubpart questions,we will solve only the first three questions as per our Q&A…
Q: A 22.6 mL sample of 0.356 M ethylamine, C₂H5NH₂, is titrated with 0.279 M hydroiodic acid. After…
A: Given that : The volume of ethylamine = 22.6 mL The molarity of ethylamine = 0.356 M The molarity of…
Q: How much energy is needed to raise the temperature of 40.0 g of argon from 25°C to 40°C? The…
A:
Q: Question 25 of 42 As the concentration of reactants decrease, the rate of the reaction generally…
A: Rate of a reaction depends upon various factors like Concentration Temperature Nature of reactant…
Q: why is the closest whole number 5?
A: It is hit and trial method At the end we are trying to get whole number. Let 2.4 is multiply with 2…
Q: The reaction of a metal, M, with a halogen, X2, proceeds by an endothermic reaction as indicated by…
A: Answer: In the given question metal atom M and non-metal molecule X2 are reacting together to form…
Q: [Review Topics] [References] Use the References to access important values if needed for th A sample…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: OH NBS NaHCO3 H -Br
A: This is a simple organic reaction of addition reaction followed by intramolecular substitution…
Q: 1. How many MOLES of nitrogen trifluoride are present in 3.44 grams of this compound? for this…
A:
Q: I'm trying to follow the formula: Eocell= Eocathode-Eoanode, does this formula apply to any of…
A: 2 ClO2(g) + 2 e- → 2 ClO2-(aq) Eº = 0.95 V eq 1 It is oxidation process at anode Sn4+(aq) + 2e-…
Q: The mechanism proposed for the oxidation of iodide ion, I, by the hypochlorite ion, CIO, in aqueous…
A: Answer: Slowest step of the reaction is the rate determining step and according to law of mass…
Q: Select all that apply. What amino acids are synthesized from a-ketoglutarate alanine isoleucine…
A: amino acids are the organic compounds that containing both amino and the carboxylic acid .
Q: A given aqueous solution has a density of x g/mL and y% solute by mass. Derive the mathematical…
A: Given that the density of aqueous solution is x g/mL. The mass percent of solute in the aqueous…
Q: A 1.00 liter solution contains 0.44 M ammonia and 0.57 M ammonium bromide. If 0.140 moles of calcium…
A:
Q: Determine the name for N₂O5.
A: Given compound N2O5
Q: The reaction below is a decomposition reaction of HI. 2HI → H₂ + 1₂ In this reaction, the following…
A: The rate of a chemical reaction is typically expressed as the change in concentration of a reactant…
Q: What is the volume (in liters) of a 0.78 Molar solution made from 0.29 moles of potassium bromide?…
A: Molarity is defined as number of moles of solute divided by volume of solution in liter .
Q: Check the box next to each molecule on the right that has the shape of the model molecule on the…
A: Molecular geometry is the three-dimensional arrangement of the atoms that constitute a molecule. It…
Q: Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only to the…
Q: The key enantioselectivity determining step in the synthesis is given below. Provide a mechanism. Br…
A:
Q: Give detailed Solution with explanation needed
A: Benzene shows an electrophilic substitution reaction such as halogenation, nitration, sulfonation…
Q: When the central atom of a molecule has a steric number (i.e., number of electron regions) of 5 and…
A: Steric number is defined as sum of number of sigma bond plus lone pair around central atom .
Q: Are the two structures shown below resonance forms? Why? ? → :A=B-A: B=Ä=Ä: A: O Yes, because they…
A: Resonance involves the different arrangement of electrons. In resonance, arrangement of atoms…
Q: The sodium D lines from a sodium lamp consist primarily of a doublet at 589.5 and 589.0 nm. Suppose…
A:
Q: portant values if needed for this question. 1. How many GRAMS of boron are present in 2.07 moles of…
A: Solving only the first question as per guidelines. if u want all of them answered kindly post them…
Q: Why does this produce a bright-line emission spectrum (rather than a continuous spectrum)? In terms…
A: Q1 An ion is an atom or molecule that has an electrical charge due to the gain or loss of one or…
Q: For Acetone, the normal boiling point is 45.6 ° C and the enthalpy of vaporization is 36.7 kJ/mol.…
A: The Clausius-Clapeyron equation that relates the pressure and temperature is lnP2P1=-∆HvapR1T2-1T1…
Q: Consider the chlorotrioxyl (CIO) cation. What is the central atom? Enter its chemical symbol. How…
A: We have ClO3+ cation here central atom is chlorine
Q: Multiple Choice Which equation best represents the measured electric potential (Emeasure) of a redox…
A: The correct answer is 3. Emeasure=1/2(E0Fe3++E0Ce4+)-Ereference.
Q: D Question 3 NCI 3 How many single bonds are on the nitrogen? How many double bonds are on the…
A: This question belongs to Chemical Bonding.
Q: When 18.0 mL of a 4.50x10-5 M ammonium fluoride solution is combined with 12.0 mL of a 7.34x10-4 M…
A:
Q: 19.11 Deduce the formula of each of the following transi- tion metal complexes: (a)…
A: Naming of transition metal complex : 1. Cation should be named first followed by the name of…
Q: A solution consists of 1.50 M Ni(NO3)2 and 1.25 M Fe(NO3)2. Which species will precipitate first as…
A: Precipitation depends on the Ksp ( solubility product value). If the Ksp is lower in value, that…
Q: When does a molecule's molecular geometry differ from its electron geometry? When the terminal atoms…
A: According to VSEPR Theory , Molecular geometry : Molecular geometry of any molecules is determined…
Q: A 50.0-g sample of liquid water at 25.0°C is mixed with 29.0 g of water at 45.0°C. The final…
A:
Q: 1.Give the Correct Chemical Formula of the Following Chemical Names Example: Sodium Phosphate=…
A: Check the cation and anion for given formula and use correct method to write formula for given…
Q: What is the reason why chemical rxns undergo elementary/non-elementary rates?
A: Elementary reactions are single-step reactions that occur in a single event, where the reactants…
Q: 1. Write the Chemical Equations to correspond to each of the following description. 2.a When Zinc is…
A:
Q: The molar solubility of iron (III) hydroxide in a 0.241 M iron(III) acetate solution is M.
A: Fe(OH)2 (s) <------> Fe2+ (aq) + 2OH- (aq) Ksp = [Fe2+][OH-]2 Ksp = 8.0*10-16 Iron acetate…
Q: The molar solubility of barium phosphate in a 0.122 M potassium phosphate solution is
A: Ba3(PO4)2(s) <---------> 3Ba2+(aq)+2 PO43-(aq) The solubility product constant, Ksp is the…
Q: Acetylene C2H2 gas is often used in welding torches because of the very high heat produced when it…
A: Acetylene gas undergoes combustion when treated with oxygen, carbon dioxide gas and water vapor are…
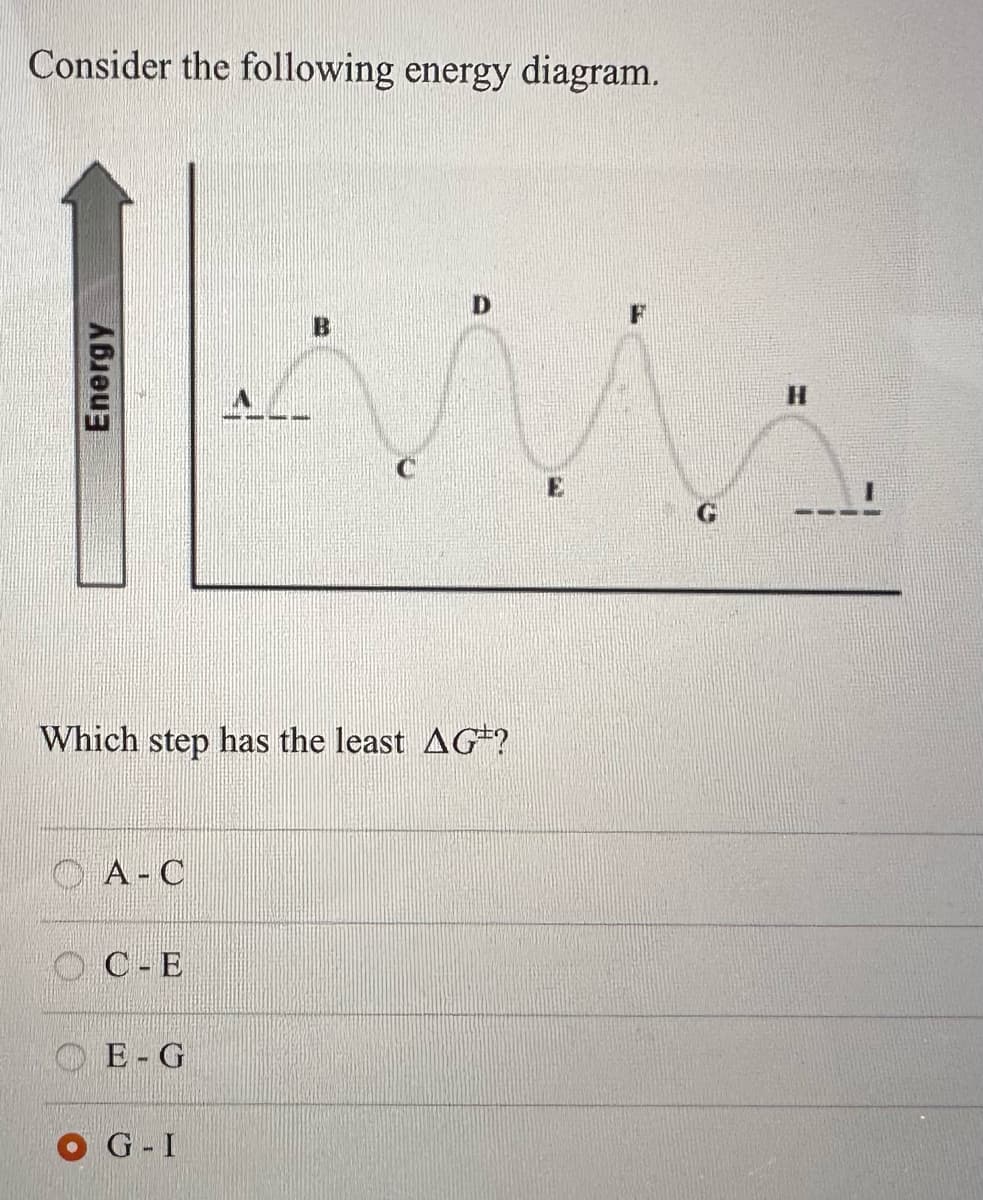
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- At equilibrium, the free energy of the reactants is equal to the free energy of the products. true or falsefor part b, besides drawing the reaciton coordiate diagram, can you give. a quite detailed explanation about how do we know which curve corrresponds to A or B. i.e. how do we know A or B energy is higher ; whether theres energy differnce between reactant and products. thanks help with part b pleaseIf the free energy change DG for a reaction is -46.11 kJ/mol, the reaction is: A) at equilibrium B) endergonic C) endothermic D) exergonic 5. E) exothermic
- What is the overall reaction called? Which step in the backward direction releases the most free energy?In each of the following three reaction coordinate diagrams, state: (a) Whether the reaction is exothermic or endothermic. (b) Whether the reaction is the slowest, the fastest, or intermediate in rate. (c) If all three reactions have the same entropy change between the reactant and product, which reaction has the largest favorable G0.Draw the organic products K and L and classify the chemical transformations reactions
- A chemical interaction in which the products contain less free energy than the reactants is called a(n) ____ reaction.Which side of this equilibrium is favored? Reactants because they are more stable Reactants because they are less stable Products because they are more stable Products because they are less stableAnswer true or false to the following statements about energy diagrams and reactions. Q.) A reaction coordinate diagram is used to visualize the change in the internal energy of chemical structures that occurs during chemical reactions
- Rank the following from highest entropy to lowest. O2 O O3Calculate the net energy for the following reaction: 4 (C-H) + O=0 ----> 2 (C=O) + 4 (0-H) 1772 kJ 948 kJ O-1772 kJ O-948 kJ -1308 kJ GCalculate the dHo of the synthesis reaction using the values from the table provided. H2(g) + O2 (g) ---> H2O (g)



