Consider the following reaction: PCI{(g) PC13(g)+ Cl2(g) If 1.00×10-3 moles of PCI5, 0.302 moles of PC13, and 0.366 moles of Cl, are at equilibrium in a 17.0 L container at 730 K, the value of the equilibrium constant, Kp. is
Consider the following reaction: PCI{(g) PC13(g)+ Cl2(g) If 1.00×10-3 moles of PCI5, 0.302 moles of PC13, and 0.366 moles of Cl, are at equilibrium in a 17.0 L container at 730 K, the value of the equilibrium constant, Kp. is
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter15: Principles Of Chemical Reactivity: Equilibria
Section: Chapter Questions
Problem 5PS: A mixture of SO2, O2, and SO3 at 1000 K contains the gases at the following concentrations: [SO2] =...
Related questions
Question
Stuck on this

Transcribed Image Text:Consider the following reaction:
PCI{(g) PC13(g)+ Cl2(g)
If 1.00×10-3 moles of PCI5, 0.302 moles of PC13, and 0.366 moles of Cl, are at equilibrium in a 17.0 L
container at 730 K, the value of the equilibrium constant, Kp. is
Expert Solution
Introduction
Equilibrium constants mainly get measured in concentration terms as well as pressure terms. The equilibrium constant evaluated in pressure terms is symbolized "Kp" whereas equilibrium constant evaluated in concentration terms is symbolized "Kc".

Solution
The given reaction is as follows,

The formula for the calculation of the equilibrium concentration/molarity is shown below.
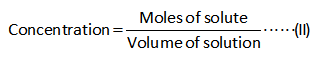
Solution
Substitute the known values in the equation (II) to calculate the concentrations.
The concentration of PCl5:
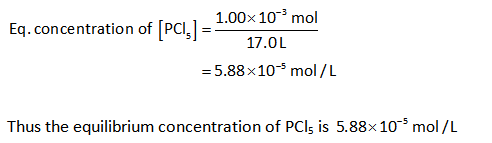
The concentration of PCl3:
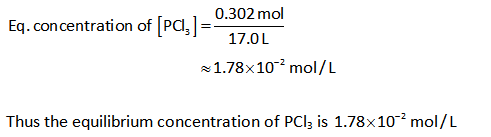
Solution
The concentration of Cl2:
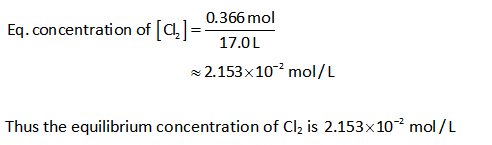
Substitute the known values in the equation (I).
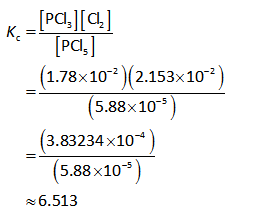
Thus the value of equilibrium constant (Kc) is 6.513.
Step by step
Solved in 7 steps with 12 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER