Consider the image of a mercury manometer below. Based on the image, if the pressure of the atmosphere is 720.0 mmHg, what is the pressure of the gas inside the manometer, in mmHg?
Consider the image of a mercury manometer below. Based on the image, if the pressure of the atmosphere is 720.0 mmHg, what is the pressure of the gas inside the manometer, in mmHg?
Introductory Chemistry: A Foundation
8th Edition
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter13: Gases
Section: Chapter Questions
Problem 137AP
Related questions
Question
Consider the image of a mercury manometer below. Based on the image, if the pressure of the atmosphere is 720.0 mmHg, what is the pressure of the gas inside the manometer, in mmHg?
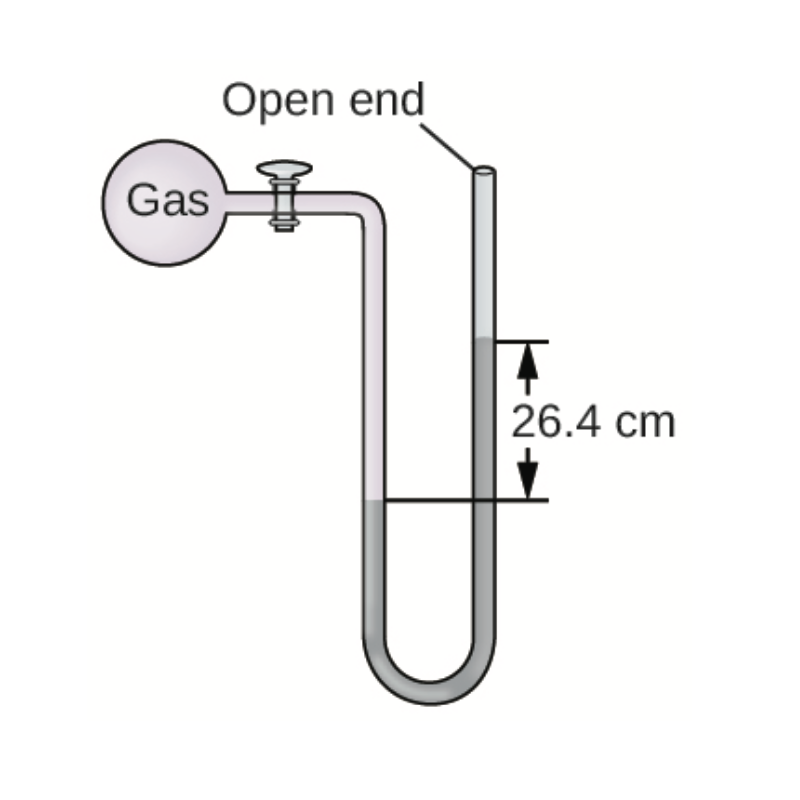
Transcribed Image Text:Open end
Gas
26.4 cm
Expert Solution
Step 1
Given that:
Atmospheric pressure = 720.0 mm Hg
Height of the mercury column = 26.4 cm Hg
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning