Q: Write the chemical formula for diarsenic trioxide
A: The chemical formula of diarsenic trioxide is
Q: Chemically and physically describe the combustion of methane CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H20…
A: Combustion of Methane : Combustion of Methane is a chemical reaction in which methane gas burns,…
Q: Write a balanced equation for the combustion of liquid propanol (C3H7OH).
A: Given chemical compound is: propanol C3H7OH. Write a balanced equation for the combustion of liquid…
Q: What mass of water will be produced from the combustion of 1 gallon of octane (C3H18) given that the…
A: 2C8H18 + 25O2 → 16CO2 + 18H2O Mass of octane = Volume x density Mass of C8H18 = (3.8×1000)ml…
Q: Hydrocarbons are compounds that contain only C and H atoms. When a hydrocarbon reacts with O 2, CO 2…
A: The given hydrocarbons are a) benzene b) toluene c) isooctane To find: Balanced equation for the…
Q: Write a balanced chemical equation, including states of matter, for the combustion of ethylene gas,…
A: Combustion reaction is a reaction in which carbon compounds react with oxygen to form carbon…
Q: How many kilograms of CO2 are produced by the complete combustion of 3.8 kg of n-octane?
A: The balanced chemical reaction for combustion of n-octane is shown as follows:
Q: crude oil
A: Crude oil is defined as a mixture of hydrocarbons that occurs in liquid form in natural underlying…
Q: What mass of methane is required to produce 1.130 kg of carbon dioxide during combustion? (Assume…
A:
Q: What is the total number of moles of O2(g) required for the complete combustion of 1.5 moles of…
A:
Q: Write a chemical equation for the combustionof each of the following substances. If a…
A: Combustion, also known as combustion, is a high-temperature exothermic redox chemical reaction that…
Q: Write a balanced chemical equation, including states of matter, for the combustion of gaseous…
A: A balanced chemical equation or reaction is the one in which the total number of atoms for each…
Q: Identify A–J:
A:
Q: Write a balanced chemical equation for the hypothetical reaction of liquid dichloromethane, CH2CI2,…
A: Given that : We have to write a balanced chemical equation of liquid dichloromethane, CH2Cl2, with…
Q: Classify the following compounds as primary (1o), secondary (2o), or tertiary (3o) alcohol
A: Primary (1°) carbon :- A carbon atom directly bonded to only one other carbon is known as primary…
Q: How could an industrial plant control the proportions of methane and chlorine to favor the…
A: In a reaction mechanism, all steps must be written with the intermediates formed during the…
Q: Why are carbon and its compounds used as fuels for most applications?
A: Carbon and its compounds are used as fuels for most applications because of the following reasons-…
Q: They are all part of one question. Ethanol, C2H6O, is most often blended with gasoline - usually as…
A: Since heat of combustion is the amount of energy released when 1 mole of reactant is com-busting…
Q: 4. Write and balance the combustion reaction of 1-naphtol, C1oHsO
A:
Q: How many grams of oxygen are needed for the complete combustion of 52.0 g of acetylene?
A: Stoichiometry furnishes the relation of reacting molecules with produced molecules. This technique…
Q: The compound shown here is classified as what type of organic compound? HỌC-CH, COCH,CH A) ether B)…
A: Different functional groups in organic chemistry are, Note: Here, R or R' is a alkyl group. Aldehyde…
Q: What is the balanced molecular equation for the complete combustion (in the presence of excess…
A: Combustion is a reaction in which the compound reacts with the oxygen gas to produce carbon dioxide…
Q: What mass of water is produced by the complete combustion of 225.0 g of butane (C4H10)? The balanced…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: A. Complete and balance the following combustion reactions. Assume that each hydrocarbon is…
A: Combustion reactions are those in which a hydrocarbon burns in the presence of air ( specially…
Q: How many carbon monoxide molecules (CO) are needed to react with 12 hydrogen molecules (H2) to…
A: Mole Ratio: is a conversion factor between compounds in a chemical reaction, that is derived from…
Q: Write the balanced equation for the combustion of ethane, C2H6
A:
Q: which of the following are the general products of a combustion reaction? (A) CO2 and H2 (B)CO2…
A:
Q: NaH, ? HO
A: NaBH4 is a reducing agent. Aldehyde or ketone is reduced to give corresponding primary or secondary…
Q: biodiesel is considered what type of organic reaction?
A: Biodiesel is a type of diesel fuel extracted from animals or plants and composed of straight chain…
Q: Write a balanced equation for the combustion of isooctane, C8H18, a component of gasoline.
A: The balanced equation for the combustion of isooctane has to be given.
Q: 4. Write and balance the combustion reaction of methyl sorbate, C-H10O2.
A: In balanced reaction number of each atom on reactant side is equal to number of each atom on product…
Q: Calculate the total mol amount of gaseous products that form from the combustion of the 20.0 mL of…
A: The combustion reaction of benzene is given as follows: C7H8(g)+9O2(g)→∆7CO2(g)+4H2O(g) The volume…
Q: Chemistry The organic product formed from the following reaction is 1) BH, THE 2) H,O, OH
A:
Q: Why isn't kerosene used as engine fuel?
A: Answer - Generally, kerosene, although a lighter blend of diesel, makes a poor fuel for spark…
Q: An impure sample of hydrocarbon having a total mass of 50.8g undergoes combustion analysis to…
A: Since the compound is given as hydrocarbon. Hence the compound consist of hydrogen and carbon only.…
Q: cigarette lighters burn butane, C4H10. write a balanced equation, assuming complete combustion, that…
A: Combustion reaction is a type of reaction when a particular substance is burnt in presence of oxygen…
Q: All of the following are key characteristics of a combustion reaction except formation of heat O…
A: 1) formation of heat combustion reactions is exothermic reactions and heat is released in the…
Q: Write a balanced chemical equation, including states of matter, for the combustion of gaseous…
A: A combustion reaction (commonly known as burning ) is an exothermic reaction in which something…
Q: Write the major products of the reaction of ethylene oxide with each of the following: Indicate…
A: We have given that Write the major products of the reaction of ethylene oxide with each of the…
Q: Complete reaction
A: Jones reagent consists of an oxidizing agent, an acid, water and the acetone. Jones reagent is used…
Q: During combustion, if nitrogen is present in the original fuel, it is converted to N2, not to a…
A:
Q: How many kilograms of CO2 does the complete combustion of3.8 kg of n-octane produce?
A: The balanced chemical reaction for combustion of n-octane is shown as follows:
Q: a) What mass of carbon dioxide is produced from the complete combustion of 9.00 x 10-3 g of methane?
A:
Q: Why do we usually not balance equations for organic reactions?
A: We usually not balance the equations for organic reactions but we always balance the inorganic…
Q: How many g of CO2 will be produced by the combustion of 4 mol of ethanol?
A: The Molecular reaction equation for combustion of ethanol C2H5OH + 3 O2 → 2 CO2 + 3 H2O + ∆ From…
Q: Complete and balance the complete combustion of octane (C8H18)
A: Octane is an alkane with the molecular formula C8.H18, which has 8 carbons and 18 hydrogen…
Q: What is the reaction that represents the combustion of C6H12O2
A:
Q: Methane is oxidized in the atmosphere by photochemical reaction. Write the chemical equation of this…
A: Since the methane is getting oxidised. Hence the addition of oxygen will take place on methane and…
Q: This refers to the residue left from the complete combustion of the sample in the open air at 700°C±…
A: During the analysis of coal generally we use two methods of analysis ; Ultimate analysis Proximate…
Complete and balance the following combustion reaction . Assume that each hydrocarbon is converted completely to carbon dioxide and water.
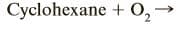
Step by step
Solved in 2 steps

- Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.Draw the structural formula for each (i) 3-methyl-5-(2,2-dimethylpropyl) nonane(ii) 1,2-dichloro-3-methylcyclohexaneof the following compoundsThe sex attractant of the female tiger moth is an alkane of molecular formula C 18H 38. Is this molecule an acyclic alkane or a cycloalkane?

