Q: A solution of AgNO3 boils at 101.5 °C. At what temperature does the solutions freeze? Use the…
A: ΔTb = Kb x m ------(1)Where, ΔTb = Boiling point elevation, Kb = molal elevation constant and m =…
Q: The number that relates to the main energy level that an electron is found in is called spin…
A: The correct answer is :----primary quantum number
Q: Cyclohexane is expected to be most soluble in: HCl water None of the above pentane HI
A: There is a saying, "Like dissolve like".Polar solutes dissolve in polar solvents and non-polar…
Q: A chemistry graduate student is studying the rate of this reaction: →2H3PO4aq+P2O5aq3H2Oaq She fills…
A: The rate law of the reaction gives the relation between the rate of reaction, rate constant, and…
Q: What reagents are required to convert the alkene 1-propene to the alcohol 2- propanol? A) H₂ B) H.O,…
A:
Q: 2 moles CH₂Cl₂ AICI 3
A: In Friedel-Crafts alkylation in benzene ring, any alkyl group is attached with the benzene ring via…
Q: Draw the structure of cis 1,3 dimethyl cyclohexane and trans 1,3 dimethyl cyclohexane and explain…
A: 1. Chair conformation 2. E2 Elimination reaction
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains…
A: Given,Mass of the water in water bath = 5.20 KgInitial temperature of water bath = 33.7 0CSpecific…
Q: AICI3 CI
A:
Q: O A © O B O A D OE Provide the structure of the major mononitration product of the compound below.…
A: Mononitration product
Q: Calculate the energy required to heat 0.90 kg of aluminum from -1.4 °℃ to 9.6 °c. Assume the…
A: It is based on the concept of specific heat capacity.Heat is form of energy.Here we are required to…
Q: or this:
A: The process of retrosynthesis follows a "backwards" approach, starting from the desired product and…
Q: In the laboratory, a student dilutes 16.8 mL of a 6.40 M hydrochloric acid solution to a total…
A: • Before dilution,Concentration of HCl solution (M1) = 6.40 MVolume of HCl solution (V1) = 16.8 mL•…
Q: What is the pH of an aqueous solution made by mixing 300 mL of 0.10M hydrofluoric acid (pKa= 3.18)…
A:
Q: or this: H
A: We have been given 2 reaction schemes.In the first scheme, we need to show the synthetic pathway…
Q: A buffer solution contains 0.314 M hydrocyanic acid and 0.307 M potassium cyanide. If 0.0366 moles…
A: Given,(i) Molarity of hydrocyanic acid =0.314 MMolarity of potassium cyanide =0.307 Mvolume of the…
Q: How many grams of NaF ( molar mass = 41.99 g/mol) would be needed to be added to 2.00 L of 0.100 M…
A: Molar mass of NaF = 41.99 g/molVolume of HF = 2.00 LMolarity of HF = 0.100 MpKa = 3.18 pH = 4.00
Q: Consider a titration where 100mL of 0.100 M acetic acid is titrated with 0.050 M sodium hydroxide.…
A: The balanced equation is CH3COOH (aq) + NaOH (aq) ------> CH3COONa (aq) + H2O (l)Number of moles…
Q: 28.9 g per day of a certain industrial waste chemical P arrives at a treatment plant settling pond…
A: Mass = 28.9 g per day Volume = 1000 m3Half-life = 33 hr
Q: Which of the following is the weakest intermolecular force? ionic bonding dipole-dipole…
A:
Q: Test I. PROBLEM SOLVING ( 4. Find the degree of unsaturation NH₂ Organic Chemistry Test I. PROBLEM…
A: We have to calculate the degree of unsaturation of the given compounds.
Q: A sample of glass, which has a specific heat capacity of 0.670 J-gc, is put into a calorimeter (see…
A: Details
Q: Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right).…
A:
Q: 1. The percent yield of this reaction is 69.0%. 2Al(s)+3 F2(g)⟶2AlF3(s) Determine the mass, in…
A: 2Al(s) + 3F2(g) ⟶ 2AlF3(s) Percent yield = 69.0 %mass of aluminium (Al) = 126.0 gmass of product…
Q: How much energy is absorbed by a 3.5g substance that has a specific heat capacity of 0.38J/g^2C and…
A:
Q: 3Si(s) + 2N2(g) yields Si3N4(s) In this synthesis equation for production of trisilicon…
A: Mass of silicon = 75.0 gMolar mass of silicon = 28.085 g/molMolar mass of trisilicon tetranitride =…
Q: Which of the following forms when a strong acid reacts with sodium bicarbonate? A. Carbonic acid B.…
A: Sodium carbonate is a salt of a strong base, sodium hydroxide, and weak acid, carbonic acid. Sodium…
Q: A constant current is passed through an electrolytic cell containing molten MgCl2 for 13.0 h. If…
A: Given,Mass of Cl2 = 5.80 x 105 gTime = 13.0 h
Q: Which of the following does not have an electron configuration ending with 2S^2 2P^6? chlorine ion…
A: we have to select the ion that does not have an electron configuration that ends as
Q: CaCO3(s)→CaO(s)+CO2(g) Express your answer as a chemical equation. Identify all of the phases in…
A: Concept of writing the balanced chemical equation in which number of atoms of each elements in…
Q: A compound such as CH3CH₂CH₂OCH₂CH3 is classified as what type of organic compound? 4 A) alcohol B)…
A:
Q: Test tube A Test tube B Test tube C Reactants Methanol + salicyclic acid and H+ Pentan-1-ol + acetic…
A: Esterification : Reaction of carboxylic with alcohol under acid condition gives an ester.
Q: Consider the neutralization reaction 2 HNO3(aq) + Ba(OH)₂ (aq) → 2H₂O(1) + Ba(NO3)₂(aq) A 0.110 L…
A:
Q: What is the major organic product obtained from the following reaction? (Ignore the bent…
A:
Q: Describe the Bohr model for the atom. Match the items in the left column to the appropriate blanks…
A: We have to fill the blank with the help of matches given .
Q: A certain reaction is second order in N₂ and first order in H₂. Use this information to complete the…
A: [N2] [H2] initial rate of reaction0.851 M1.62 M73.0 M/s0.851 M0.467 M?2.98 M0.463 M?Order of…
Q: Draw the skeletal (line-bond) structure of 4-nitrobutanoyl chloride.
A: The structure of an organic compound is written in the following steps:Determine the number of…
Q: Is the following statement True or False? Volume can be measured directly by taking the dimensions…
A: Volume can be measured directly by taking the dimensions of an object, while mass can be determined…
Q: 4. H₂SO4 reacts with NaOH to form water and Na₂SO4. In a simple calorimeter, a 100.00 mL sample of…
A: Given,Molarity of H2SO4 = 0.355 mol/LVolume of H2SO4 = 100.00 mLVolume of NaOH = 50.00 mLThe rise…
Q: Which of the following affects the melting point of a substance? London dispersion forces…
A: Intermolecular forces are the forces of attraction between molecules that are interacting with each…
Q: Calculate the mass of urea that should be dissolved in 201 g of water at 35°C to produce a solution…
A:
Q: Draw both resonance structures of the most stable carbocation intermediate in the reaction shown.
A: A carbocation is an electron deficient species with carbon having positive charge. The stability of…
Q: State the relationship between the following pairs of molecules selecting from the following options…
A: •In the first case , the molecule must be converted in fisher projection formula ..by which we can…
Q: An aqueous solution has a hydroxide ion concentration of 1.0 × 10-¹1 M. What is the hydronium ion…
A:
Q: Examine the curved arrow mechanism below, and answer the question in the box. If the mechanism is…
A: During mechanism flow of electrons occur from higher electron density to lower electron densityA…
Q: Consider the dissolution of calcium chloride in water. Which of the changes listed below will…
A: Lechatelier Principle is used to explain the change during an equilibrium reaction so that overal…
Q: Consider the reaction that describes the disassociation of the following weak acid. H3A (aq) + H₂O…
A:
Q: ¹₂(g) + ICl(g) → HI(g) + HCl(g) HI(g)+ ICI(g) 1₂(g) + HCl(g) Suppose also k₁«k₂. That is, the first…
A:
Q: need to complete the retrosynthesis for this: or this: H O
A: Retrosynthesis is a hypothetical way to cleave a target compound into simpler fragments by which the…
Q: You are playing the role of a high school chemistry teacher. Provide an analogy to explain the…
A: The state of reaction at which the contraction of the reactants and products does not change with…
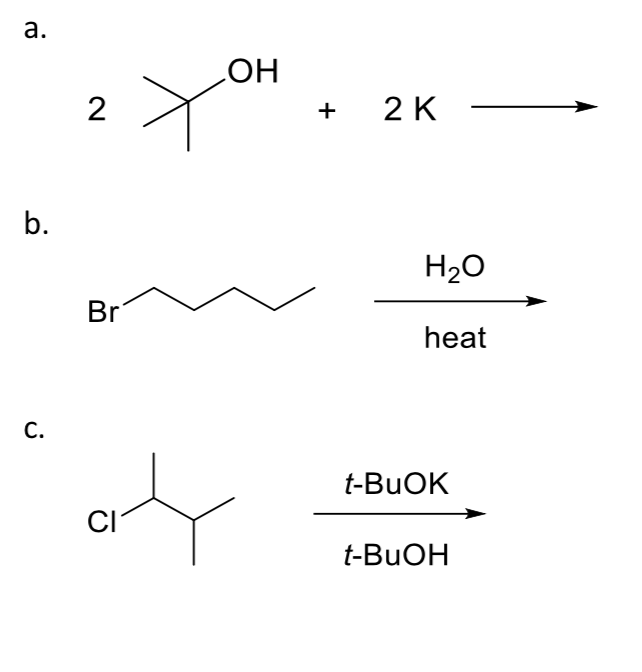
Predict the major product(s) for each of the following reactions. If no reaction is expected,
write NR.
Step by step
Solved in 3 steps with 2 images


