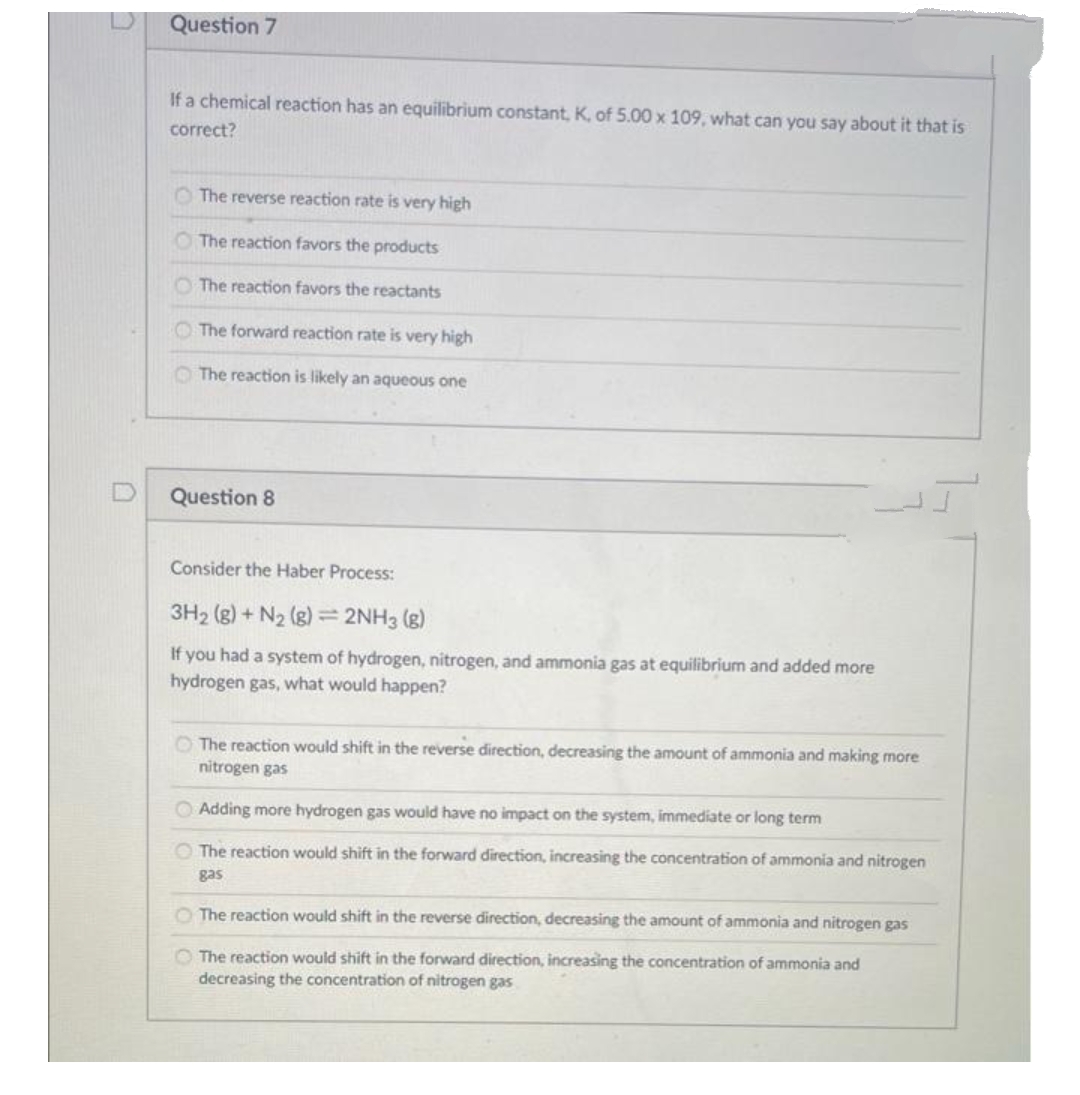
D Question 7 If a chemical reaction has an equilibrium constant, K, of 5.00 x 109, what can you say about it that is correct? The reverse reaction rate is very high The reaction favors the products The reaction favors the reactants The forward reaction rate is very high The reaction is likely an aqueous one Question 8 Consider the Haber Process: 3H₂(g) + N₂ (g) = 2NH3(g) If you had a system of hydrogen, nitrogen, and ammonia gas at equilibrium and added more hydrogen gas, what would happen? The reaction would shift in the reverse direction, decreasing the amount of ammonia and making more nitrogen gas Adding more hydrogen gas would have no impact on the system, immediate or long term O The reaction would shift in the forward direction, increasing the concentration of ammonia and nitrogen gas O The reaction would shift in the reverse direction, decreasing the amount of ammonia and nitrogen gas The reaction would shift in the forward direction, increasing the concentration of ammonia and decreasing the concentration of nitrogen gas
D Question 7 If a chemical reaction has an equilibrium constant, K, of 5.00 x 109, what can you say about it that is correct? The reverse reaction rate is very high The reaction favors the products The reaction favors the reactants The forward reaction rate is very high The reaction is likely an aqueous one Question 8 Consider the Haber Process: 3H₂(g) + N₂ (g) = 2NH3(g) If you had a system of hydrogen, nitrogen, and ammonia gas at equilibrium and added more hydrogen gas, what would happen? The reaction would shift in the reverse direction, decreasing the amount of ammonia and making more nitrogen gas Adding more hydrogen gas would have no impact on the system, immediate or long term O The reaction would shift in the forward direction, increasing the concentration of ammonia and nitrogen gas O The reaction would shift in the reverse direction, decreasing the amount of ammonia and nitrogen gas The reaction would shift in the forward direction, increasing the concentration of ammonia and decreasing the concentration of nitrogen gas
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 9RQ: What is Le Chteliers principle? Consider the reaction 2NOCI(g)2NO(g)+Cl2(g) If this reaction is at...
Related questions
Question
Transcribed Image Text:D
Question 7
If a chemical reaction has an equilibrium constant, K, of 5.00 x 109, what can you say about it that is
correct?
The reverse reaction rate is very high
The reaction favors the products
The reaction favors the reactants
The forward reaction rate is very high
The reaction is likely an aqueous one
Question 8
Consider the Haber Process:
3H₂(g) + N₂ (g) = 2NH3(g)
If you had a system of hydrogen, nitrogen, and ammonia gas at equilibrium and added more
hydrogen gas, what would happen?
The reaction would shift in the reverse direction, decreasing the amount of ammonia and making more
nitrogen gas
Adding more hydrogen gas would have no impact on the system, immediate or long term
O The reaction would shift in the forward direction, increasing the concentration of ammonia and nitrogen
gas
O The reaction would shift in the reverse direction, decreasing the amount of ammonia and nitrogen gas
The reaction would shift in the forward direction, increasing the concentration of ammonia and
decreasing the concentration of nitrogen gas
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning