Data Table 1: Reaction Data Sample Moles of vinegar (mol) Moles of baking soda (mol) Small, bigger Relative size of bag Was excess vinegar present? Color with indicator? Reacts with additional baking soda? Moles of gas produced (mol) Watch the limiting reagent! #1 0.042 #2 yes yellow 0.042 0.014 чем Smat Small 0.028 #3 0.042 0.042 #4 0.042 0.071 #5 big yes yes Yellow yellow Geen 0.042 0.88 more bigger bigger NO No Blue Calculations 1. Use Table 1 to determine which reactant was limiting in each trial. Use the amount of limiting reactant to calculate the moles of carbon dioxide gas that were produced. These values should be reported in the Moles of Gas Produced row in the table.
Data Table 1: Reaction Data Sample Moles of vinegar (mol) Moles of baking soda (mol) Small, bigger Relative size of bag Was excess vinegar present? Color with indicator? Reacts with additional baking soda? Moles of gas produced (mol) Watch the limiting reagent! #1 0.042 #2 yes yellow 0.042 0.014 чем Smat Small 0.028 #3 0.042 0.042 #4 0.042 0.071 #5 big yes yes Yellow yellow Geen 0.042 0.88 more bigger bigger NO No Blue Calculations 1. Use Table 1 to determine which reactant was limiting in each trial. Use the amount of limiting reactant to calculate the moles of carbon dioxide gas that were produced. These values should be reported in the Moles of Gas Produced row in the table.
Chapter20: Applications Of Oxidation/reduction Titrations
Section: Chapter Questions
Problem 20.6QAP
Related questions
Question
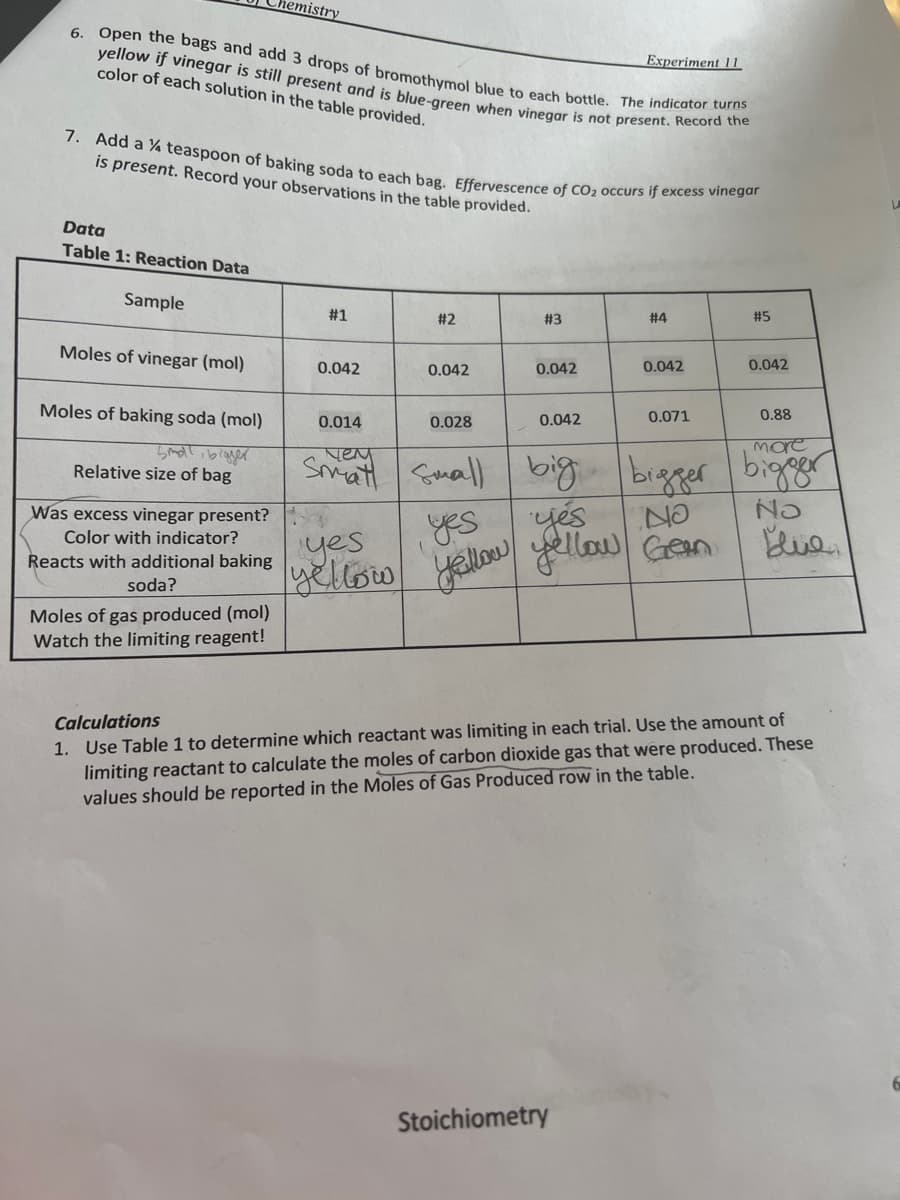
Transcribed Image Text:6. Open the bags and add 3 drops of bromothymol blue to each bottle. The indicator turns
yellow if vinegar is still present and is blue-green when vinegar is not present. Record the
color of each solution in the table provided.
Data
Table 1: Reaction Data
Sample
7. Add a 4 teaspoon of baking soda to each bag. Effervescence of CO₂ occurs if excess vinegar
is present. Record your observations in the table provided.
Moles of vinegar (mol)
Moles of baking soda (mol)
mistry
Relative size of bag
Was excess vinegar present?
Color with indicator?
Reacts with additional baking
soda?
Moles of gas produced (mol)
Watch the limiting reagent!
#1
0.042
0.014
чем
yes
yellow
#2
0.042
0.028
#3
yes
yellow
0.042
0.042
Experiment 11
yes
yellow
#4
Stoichiometry
0.042
0.88
more
small big bigger bigger
No
0.071
#5
NO
Geen
0.042
Blue
Calculations
1. Use Table 1 to determine which reactant was limiting in each trial. Use the amount of
limiting reactant to calculate the moles of carbon dioxide gas that were produced. These
values should be reported in the Moles of Gas Produced row in the table.
6
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

