Determine how the following changes affect the given equilblirium reaction that is held in a closed container. = -0.500 kJ/mol AH°rxn 2 A(g) + B()= C(g) + 2 D(s) Removing some A(g) Removing some D(s) Removing some B(I) Increasing the temperature Increasing the volume of the container A Moving to another question will save this response. MacBook Air 80 DII F1 F2 F3 F4 F5 F6 F7 F8 2$ % & * 1 2 4 くO # 3
Determine how the following changes affect the given equilblirium reaction that is held in a closed container. = -0.500 kJ/mol AH°rxn 2 A(g) + B()= C(g) + 2 D(s) Removing some A(g) Removing some D(s) Removing some B(I) Increasing the temperature Increasing the volume of the container A Moving to another question will save this response. MacBook Air 80 DII F1 F2 F3 F4 F5 F6 F7 F8 2$ % & * 1 2 4 くO # 3
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter5: Principles Of Chemical Reactivity: Energy And Chemical Reactions
Section: Chapter Questions
Problem 70GQ: You have a large balloon containing 1.0 mol of gaseous water vapor at 80 C. How will each step...
Related questions
Question
100%
20
The answer options for each of the blanks are: 1)No effect, 2) Shift equilibrium to the right, 3) Shift equilibrium to the left.
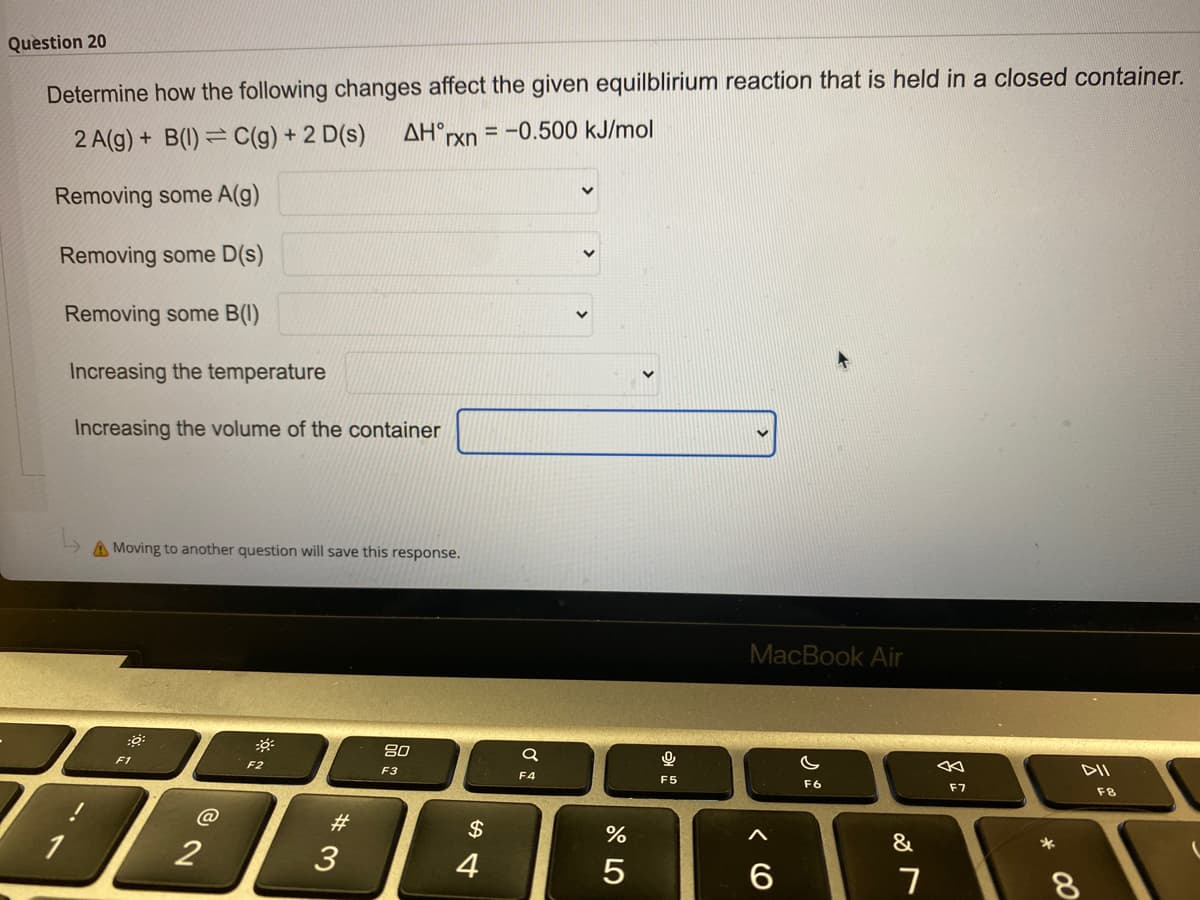
Transcribed Image Text:Question 20
Determine how the following changes affect the given equilblirium reaction that is held in a closed container.
= -0.500 kJ/mol
AH°rxn
2 A(g) + B(1) = C(g) + 2 D(s)
Removing some A(g)
Removing some D(s)
Removing some B(I)
Increasing the temperature
Increasing the volume of the container
A Moving to another question will save this response.
MacBook Air
80
DII
F1
F2
F3
F4
F5
F6
F7
F8
#3
$
&
*
2
3
4
5
6.
8.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
