Determine theoretical oxygen demand (mg/L) for 250 mg/L of methanol (CH3OH). CH,0H + 02→ C0, + H,0 Equation is ot balanced.
Determine theoretical oxygen demand (mg/L) for 250 mg/L of methanol (CH3OH). CH,0H + 02→ C0, + H,0 Equation is ot balanced.
Sustainable Energy
2nd Edition
ISBN:9781337551663
Author:DUNLAP, Richard A.
Publisher:DUNLAP, Richard A.
Chapter14: Ocean Thermal Energy Conversion And Ocean Salinity Gradient Energy
Section: Chapter Questions
Problem 5P
Related questions
Question
I need the answer as soon as possible
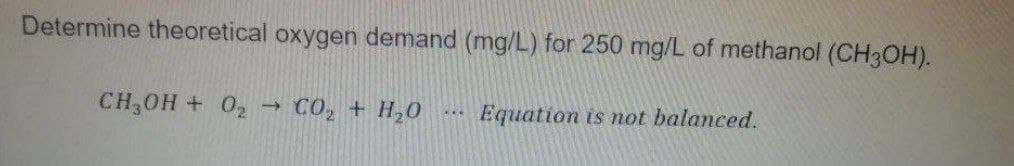
Transcribed Image Text:Determine theoretical oxygen demand (mg/L) for 250 mg/L of methanol (CH3OH).
CH,0H + 02 → C0, + H,0
Equation is mot balanced.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, civil-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

