Q: The following isomerization reaction, drawn using D-glucose as starting material, occurs with all…
A: The first step is the abstraction of proton from the carbon atom of aldehyde group to form enolate.…
Q: Draw both pyranose anomers of each aldohexose using a three-dimensional representation with a chair…
A: The following rules should be obeyed to convert Fischer projection to Haworth projection- Draw a…
Q: Draw a stepwise mechanism for the reaction of an alcohol with an isocyanate to form a urethane.
A: The given reaction is,
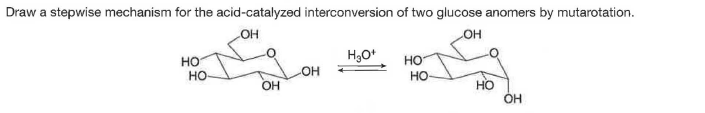
Q: The following isomerization reaction, drawn using D-glucose as starting material, occurs with all…
A: The stepwise formation of products is given below
Q: Most Activating 2 Least Activating GOH3 OH Oph CH Pheno? anisure 13r |-naphthol Diphenyl etler…
A: This question is checking the basic concept of nature of Benzylic lone pairs. We have to tackle…
Q: 1.Explain what is anomeric effect and why it happens?
A: As there are two questions ,I am solving the first question.
Q: Draw a mechanism for the conversion of glucose from its aldehyde form to its hemiacetal form and…
A: Glucose from its aldehyde form converts to its hemiacetal form and then to its methyl glycoside…
Q: Draw the mechanism for how a-pinene is biosynthesized from geranyl pyrophosphate. a-Pinene
A: Alpha –Pinene (α-pinene) is an organic compound of the terpene class, one of two isomers of pinene.…
Q: What acetylide anion and epoxide are needed to synthesize attachedcompound?
A: Given:
Q: The product of the following reaction is НО CH2OH Н ОН Н O ОН Н Н OH H-C-OH HO-C-H an uronic acid an…
A:
Q: CH3NH2 H30* e) j) NaOEt Aldol Condensation
A: Given reactions,
Q: All are true or false a)In solution, glucose is in the cyclic acetal form only. b)The driving…
A: Glucose is one of the monosaccharides which is highly soluble in water at room temperature. The…
Q: Draw the organic products formed in the following reaction.
A: The first step is the reaction of given mixture of enantiomers of amino acid with acetic anhydride…
Q: 2 CH3COOCH,CH3 C2H5O°K"/C2HSOH ---> CH3COCH2COOCH2CH3 +] Hofmann elimination Clemmensen reduction…
A: The above given reaction is a claisen condensation. Claisen condensation is a organic coupling…
Q: Nole, formatión of the enolate is carried out under irreversible conditions. 1. LDA, THF, -78 °C…
A:
Q: pyranose under acidic conditions. You do NOT have to show the mechanism for the reverse equilibrium…
A: In this question, we will convert Five member ring to six member ring by using arrow pushing…
Q: HO Ph. Ph HO H
A: This is an example of sharpless asymmetric epoxidation. It is enantioselective in nature. Diethyl…
Q: Complete the following esterification reactions: i Br H₂C ONB DMF CH₂CH₂ONa CH₂CH₂OH
A: Detail mechanistic pathway is given below to find out the product
Q: Draw the mechanism for the reduction of Camphor to Isoborneol and Borneo CH3 CH3 (ехо) exo attack…
A: Endo mechanism is most favourable because of stearic hindrance In exo mechanism H- will bear a…
Q: Draw both pyranose anomers of each aldohexose using a three-dimensional representation with a chair…
A: The structure of D-sugar is given by, The steps for the conversion of Fischer projection of D-sugar…
Q: Draw the following sugar derivatives (I) methyl B-D-glucopyranoside
A: Please find your solution below : methyl B-D-glucopyranoside has the molecular formula C7H14O6 Its…
Q: Draw a stepwise mechanism for the following reaction, the last step in afive-step industrial…
A:
Q: Draw each of the following
A: a. Structure of beta-D-talopyranose: b. Structure of alpha-D-idopyranose:
Q: The following isomerization reaction, drawn using D-glucose as starting material, occurs with all…
A: First, abstraction of proton from alpha carbon present at -CHO group occurs and results to form…
Q: Draw the product for the tollowing hydrolysis Ht + H2O -C-0- ren Free
A: Ester hydrolysis: When an ester is hydrolyzed in presence of an acid, an alcohol and a carboxylic…
Q: HO CH3 HO HO 1L но HO OMe 2. HO Proton Transfer но HO. HO. HO CH3OH, H* HO curved-arrow mechanism…
A: Hemiacetal is one of the important functional group and it is susceptible for acids. When…
Q: The following isomerization reaction, drawn using D-glucose as starting material, occurs with all…
A: The first step is the abstraction of proton from the carbon atom of aldehyde group to form enolate.…
Q: Draw the expected product of the reaction of the following sugars with excess methyliodide and…
A: The –OH group present in given sugar reacts with alkylating agents like CH3I and Ag2O and results in…
Q: Draw the product formed when (CH3CH2CH2CH2)2CuLi is treated withattached compound. In some cases, no…
A: The general formula of Gilman reagent is expressed as R2CuLi. Here, R is an alkyl or aryl group.…
Q: Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some…
A: Organometallic reagents are very useful in the field of chemistry as they can make a variety of…
Q: During the metabolism of glucose, glyceraldehyde 3-phosphate is converted to dihydroxyacetone…
A: Keto –enol tautomerizations means the chemical equilibrium between enol form and keto form. Keto…
Q: below? CHO -OH 1. NH₂OH 2. Ac₂0, 100°C HNO3 -OH 3. HG, HO A HO -H H -OH CH₂OH ect one: a. The…
A: In this question, we will draw the final product structure and identify it is optically active or…
Q: Draw a stepwise mechanism for the conversion of geranyl diphosphate to α-terpinene.
A: In the first step, the removal of OPP group from geranyl diphosphate which results in the formation…
Q: 3) Draw the product of the following reactions: a. H3CO- CH3 b. Draw the product of the following…
A:
Q: 3-Fucosyllactose is another HMO found in breast milk. (a) Locate any acetal and hemiacetal. (b) What…
A: The Human Milk Oligosaccharide (HMO) 3-FL (3-Fucosyllactose)the most abundant fucosylated HMOs, and…
Q: ArSO,H Draw curved arrows to show the movement of electrons in the following step of an…
A:
Q: Draw the products formed when D-arabinose is treated with each reagent: (a) Ag2O, NH4OH; (b) Br2,…
A: (a) Ag2O, NH4OH Here the given reagents will helps in the oxidation of aldehyde to carboxylic acid…
Q: : 20:28 : 20. Draw the full acetal formation scheme on your paper, and then draw in the curved…
A: Acetals are formed when excess alcohol( 2 equivalent) react with acetaldehyde. The complete…
Q: 0-H D-Glucose, cyclic (D-Glucopyranose) STEP 1 0-H D-Glucopyranosyloxy ania STEP 2 Ring-opening 0-H…
A:
Q: True or False ____________7] Salivary amylase can be use to hydrolyze 1,4 glycosidic bonds of…
A: Given : We have to tell whether statement is true or false.
Q: Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With…
A: Given compounds: Phenylacetic acid (C6H5CH2COOH)
Q: a) reduction reaction that produces an aldaric acid. b) oxidation reaction which results in an…
A: A multiple choice question based on organic reaction type, which is to be accomplished.
Q: Choose one answer from each parenthesis. Th
A: The two reactions that are taking place are: 1. Epoxidation of alkene 2. Acidic hydration of alkene
Q: Draw the two aldohexoses and one ketohexose that can be derived from the enol shown below. HC HO- C…
A: A monosaccharide compound containing six carbon atoms is called Hexose. Now, if Hexose contains…
Q: Give a concise svnthesis for the molecules if they start from a benzene. a. F- -Br b. Но. OH C.
A:
Q: Draw both pyranose anomers of each aldohexose using a threedimensional representation with a chair…
A: The given carbohydrates (monosaccharides) are the aldoses (containing aldehydic group). Generally,…
Q: сно но- "C=N H: H;0 HCI Pd/ Baso, H- CH,OH whats the product for WOHL degradation? for this
A: Wohl degradation is used for the formation of compound having one less carbon chain than the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the product formed when attached compound is treated with two equivalents of CH3CH2CH2CH2MgBr followed by H2O.Which D-aldopentose is oxidized to an optically active aldaric acid and undergoes the Wohl degradation to yield a D-aldotetrose that is oxidized to an optically active aldaric acid?Draw a stepwise mechanism for the following transformation, using curved arrows to show the movement of electrons at each step. The reaction is xylan to D-xylose with added HCl, H2O, and heat.
- If anisole is allowed to sit in D2O that contains a small amount of D2SO4, what products are formed?Choose one answer from each parenthesis. The identity of Structure A is represented by the structure labelled (SAT / MAT / CAT / BAT / WAT / RAT / NAT) while that of Structure B is labelled (SAT / MAT / CAT / BAT / WAT / RAT / NAT). The missing reagent in the first reaction is (dilute H2SO4 / H2O / MCPBA / Br2 and water).Draw the mechanism for the dehydration step in the Wohl degredation.
- Draw a mechanism for the conversion of glucose from its aldehyde form to its hemiacetal form and then to its methyl glycoside (acetal) form under acidic conditions. Show all bond breaking and bond forming steps; show all intermediates.Draw the products formed when β-D-galactose is treated with each reagent. a. Ag2O + CH3I b. NaH + C6H5CH2Cl c. The product in (b), then H3O+ d. Ac2O + pyridine e. C6H5COCl + pyridine f. The product in (c), then C6H5COCl + pyridineAll are true or false a)In solution, glucose is in the cyclic acetal form only. b)The driving force for the Wittig reaction is the elimination of triphenylphosphine oxide c)1H NMR spectroscopy, you can tell the difference between an aldehyde and a ketone because an aldehyde has a proton signal between 9–10 ppm. d)When an aldehyde is reacted with excess ethanol with an acid as a catalyst it is called hemiacetal e) You can't use acidic conditions (such as aqueous hydrochloric acid) for the addition of a Grignard reagent to a ketone because the ketone will be protonated and thus unreactive

