Q: Click the "draw structure" button to launch the drawing utility. Draw the structure of the acyclic…
A: Carbonyl compound (aldehyde and ketone) reacts with alcohol to form hemiacetal in the presence of an…
Q: Sketch the MO diagram of Trans- [Co(en)2 C12]CI and Cis-[Co(en)2C12]CI
A: Since the given coordination complexes contain cobalt(III) ions with two ethylenediamine (en)…
Q: 6) A standard solution of 0.134 M NaOH is used to titrate a 20.00 mL sample of 0.233 M HCI. What is…
A: The volume of the sample = The concentration of the sample = The concentration of the solution =…
Q: A 20.00ml aliquot of a 0.75M solution of a weak acid at {:1.00x10^(-5) at 25deg C) is titrated with…
A: Volume of weak acid (HA) = 20.00 mLConverting to liters:Molarity of weak acid = 0.75 MKa = Molarity…
Q: Q18. Predict the major product from the reaction shown below and draw the curved arrow mechanism for…
A: The objective of the question is to find the major product by curved arrow mechanism for the given…
Q: Question 3. a) Using an arrow, show the formation of the radical product that will result from the…
A: (a) The formation of the radical product is shown. Free radicals are generated by the homolytic…
Q: draw the condensed structural formula for 5- ethylhexane, what is wrong with this name? What would…
A: Wrong name = 5-ethylhexaneStructure of the compound,
Q: Draw the correct organic product for the oxidation reaction: Na2Cr2O7, acetone OH H2SO4 Select
A: Sodium dichromate NaCr2O7 in the presence of H2SO4 acts as an oxidising agent. it oxidises -1o…
Q: What multiplicities are observed for the signals in the off-resonance decoupled 13C spectrum of…
A:
Q: Draw both resonance structures of the most stable carbocation intermediate in the reaction shown +…
A: “Since you have posted a question with multiple questions, we will provide the solution only to the…
Q: 2. Provide the structure of the intermediates and products for the following reactions. (a) (b) CH3…
A: Hydroboration-oxidation of alkenes:Alkenes on hydroboration with boron hydride gives organoboranes,…
Q: complete and balance each of the following equations for acid base reactions HCl (aq) + Ba(OH)2 (aq)…
A: The objective of the question is to balance the given acid-base reaction. In this case, the reaction…
Q: [Review Topics] [References] Draw the major organic product of the reaction shown below. OH H2SO4 •…
A: Please check the solution in explanation part Explanation:
Q: Q1. Draw a detailed curved arrow mechanism for the following Friedel-Crafts acylation. 1. OH MeO OMe…
A: This is an example of Friedel-Crafts acylation reaction
Q: What is the major organic product obtained from the following reaction? 1. NaOH. H₂O OCHS 2. H₂O*…
A: This is an example of ester hydrolysis under alkaline condition
Q: For each pair of substituents, select the higher priority substituent according to the…
A: The Cahn-Ingold-Prelog rules or CIP rules help in assigning priorities to the substituents. Based on…
Q: Using the Haber-Bosch process to fix nitrogen and manufacture ammonia was a major industrial…
A: Equilibrium constant is defined as the ratio of partial pressure of products to the partial pressure…
Q: e o or the reaction. is shown below. cohol, The reaction, which requires two moles of the NIS…
A: The objective of the question is to understand the stoichiometry of the given chemical reaction…
Q: A solution is prepared by adding 750.0 mL of 4.00*10-3 M Ce(NO3)3 to 300.0 mL of 2.00*10-2 M KIO3.…
A: When ionic product is greater than solubility product then precipitate will form.
Q: atomic radius is 0.1430 nm calculate the planer density of (211) plane at BCC structure
A: The objective of the question is to calculate the planar density of the (211) plane in a…
Q: Balace each chemical equation HI (aq) + O2 (g) ----> H2O(1) + I2(s)
A: Answer:The Law of conservation of mass states that mass cannot be created nor destroyed. That's why…
Q: 8) Using benzene as one of the starting reagents, how could you prepare the compound listed below…
A: In the given question we have to predict the mechanism of the reaction.The benzene undergoes an…
Q: Give the chemical mechanisims for these organic reactions KCH, H₂NNH hy Brz, h Br
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: You find a very dilute solution of a very weak acid. Which of the following is true of the [H+]…
A: The question is asking about the concentration of hydrogen ions [H+] in a very dilute solution of a…
Q: Macmillan Learning H&C. CH2 H&C CH HC Consider alkene 2. CH3 ΤΟ =CH2 H2 H₂C 생 Consider alkene 3. H₂C…
A: The objective of the question is to correct the names of the given alkenes. The names of organic…
Q: 1. LDA 2. H-
A: Given that, the reaction is:
Q: Draw the mechanism for the reaction you completed in lab starting with the Grignard reagent as one…
A: please see the photo for mechanism.Explanation:Step 1: Step 2: Step 3: Step 4:
Q: In the molecular editor below, draw the molecule that would be formed from the side with the red…
A: SMILES stands for Simplified Molecular Input Line Entry System. It is a code for the structure of a…
Q: Use the following atomic weights and quantities to calculate the overall % yield of benzopinacol.…
A: 1. Yeild = 71% 2. option a and f is rightExplanation:1. 2.
Q: Which of the following statements accurately describes the Nernst equation shown below?…
A: The Nernst equation is a fundamental equation in electrochemistry, named after Walther Nernst, a…
Q: 1. List the relationship between each pair of structures as one of the following: same, structural…
A: To find out the relation between the two given structure of the substituted cyclohexane, we will…
Q: Please don't provide handwritten solution ...
A: The objective of the question is to understand the formation of sp2 hybrid orbitals and their role…
Q: The following data were obtained for the reaction: FeO (s) + CO (g) → Fe (s) + CO2 (g) Use the data…
A: A plot of ln K (equilibrium constant) vs 1/T (temperature) is called van't Hoff plot. The straight…
Q: Balance each chemical equation Na2S (aq) + Zn(NO3)2 (aq) ----> NaNO3 (aq) + ZnS(s)
A: The objective of this question is to balance the given chemical equation. Balancing a chemical…
Q: Consider the titration of 100.0 mL of 0.010 0 M Ce4+ in 1 M HClO4 with 0.040 0 M Cu+ to give Ce3+…
A: The objective of this question is to calculate the potential of the indicator electrode after adding…
Q: At 1000 K, Kp =2.1 x 10° and AH = -101.7 kJ for the reaction H2 (g) + Br2 (g)=2HBr (g). Part A A…
A: Given ,
Q: Malic acid, (HO₂C)CH,CH(OH)(CO-H) has two acidic protons. What is the pk, for the following…
A: The pKa value is a measure of acidic strength of a solution. The higher the pKa value, the lower the…
Q: Please help Please don't provide handwritten solution ...
A: The objective of the question is to understand the reason behind washing colored iron pills before…
Q: -OH SOCI₂ pyridine
A: An alcohol molecule reacts with SOCl2 to form an alkyl halideThis reaction usually takes place via…
Q: Write the common (not systematic) name of each organic molecule. structure name N trimethylamine N
A:
Q: The byproduct of the photo dimerization of benzophenone to benzopinacol is? hv OH Ph .Ph OH Ph' Ph…
A: 1. The byproduct of the photodimerization of benzophenone to benzopinacol is a. propanone. 2. The…
Q: Question 2. While internal cysteine amino acid sidechains in proteins undergo oxidation to produce…
A: In conclusion, Ellman's reagent effectively detects free thiols in proteins by reacting with them to…
Q: Polymers may be composed of thousands of monomers. Draw two repeat units of styrene. Assume there…
A: Answer:A polymer is a large molecule that is formed by the repetition of small units called a…
Q: Use your calculator to find the log of the following numbers.(a) 10−9(b) 1 × 10−11(c) 7.4 × 103(d)…
A: (a). −9 (b). −11 (c). 2+log10(74) Or approximately 3.86922 (d). 5 (e). 0 Explanation:
Q: 5. An infinite flat plate of a solid has a surface potential of 50 mV in an infinite bath of 10-2…
A: 5. Utilize flat plate with a surface potential of 50 mV in a 10-2 molL-1 NaCl(aq) solution at Cl -…
Q: Draw a curvy arrow (or arrows) to show the initial bond forming reaction between any of these…
A: -> Sodium alkoxide has nucleophilic character. Hence, it can give nucleophilic substitution…
Q: 2) The product of the reaction shown is which of the following? a. An alkane b. A cis alkene c. A…
A: Product of reaction is a. An alkane Explanation:Step 1: Step 2:When hydrogen gas (H2) and a…
Q: When 3.00 mol of A and 1.00 mol of B are mixed in a 1.00 L vessel, the following reaction takes…
A: 0.12Explanation:
Q: Draw the simplest mechanism possible for the reaction below. You may need to re-draw structures to…
A: The simplest possible mechanism for the reaction can be drawn as follows:Explanation:The reaction is…
Q: The [H3O+] of a solution with pH = 9.7 is what?
A: The objective of the question is to find the concentration of hydronium ions (H3O+) in a solution…
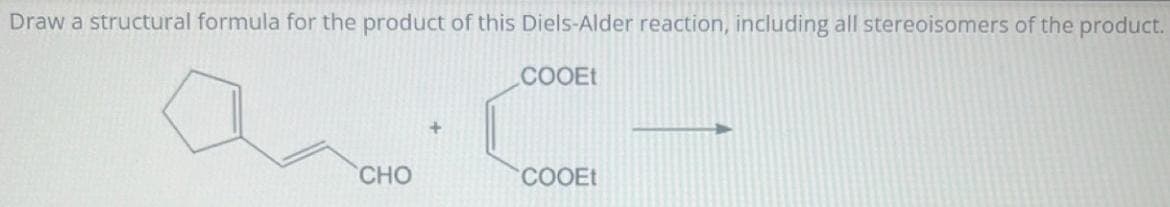
Step by step
Solved in 1 steps with 1 images

- Identify the major products for the following Diels-Alder reactionUnder certain conditions, 1,3-butadiene can function as both a diene and a dienophile. Draw a structural formula for the Diels-Alder adduct formed by reaction of 1,3-butadiene with itself.Complete the Diels-Alder reactions by drawing structures for the products in each question.
- Can someone draw the arrows for this reaction so I can understand how the product was formed? I'm having trouble understanding that aspect of the Diels-Alder reaction.The Diels-Alder reaction is not limited to making six-membered rings with only carbon atoms. Predict the products of the following reaction that produce rings with atoms other than carbon in them.What product(s) would be obtained from the Diels-Alder reaction of transCH3CH=CHCO2Et with EACH of the following dienes:(a) 1.3- Butadiene(b) Isoprene(c) Cyclopentadiene________________________________
- Predict products of each Diels-Alder reaction (including stereochemistry)Explain the stereochemistry of the product of the Diels-Alder reaction shown below:Is the following true or false? To catalyse a Diels-Alder reaction a common approach involves the use of a Lewis acid capable of lowering the LUMO of the dienophile.



