Q: The figure below shows titration of a weak monoprotic acid with a NaOH solution (titrant). Would the…
A:
Q: Q3. Describe the preparation of (a) 500 mL of 4.75% (w/v) aqueous ethanol (C₂H,OH, 46.1 g/mol). (b)…
A: According to the question, w/v% for the ethanol solution is given by = 4.75 %w/w% for the ethanol…
Q: On a day when atmospheric pressure is 76 cmHg, the pressure gauge on a tank reads the pressure…
A: Ideal gas equation is the equation which obeys gas laws under all conditions of temperature and…
Q: COMPOUND ZnCl2 K3PO4 Al2(SO4)3 MgCrO4 CaC4H406 NaCl K2Cr2O7 H₂SO4 Cu(OH)2 MgSO4-5 H2O FORMULA MASS
A: Introduction The formula mass of a compound can also be determined by multiplying the atomic mass of…
Q: Question 18 Draw of the structure of the products for the following reaction. Pay attention to…
A: The given incomplete reaction is The products of the above reaction are
Q: For the following reaction, predict the major product and draw a mechanism for its formation. 26c…
A:
Q: 27.9 g of a solute raises the boiling point of benzene to 83.74 °C. The mass of the benzene solvent…
A: Given data
Q: Percent of N was with Perot NHL produced 300 38.PS BA% 12 16 20 Ma 300 Total pressure (atm en M.P 04…
A: Answer: In Haber process N2 and H2 are made to react in presence of a catalyst to form NH3 and we…
Q: When working with molality, and no amount of solvent is specified, it may be convenient to assume…
A: Definition of molality- Molality is the number of moles of solute dissolve in one kilogram of the…
Q: When 48.0 JJ of heat is added to 13.4 gg of a liquid, its temperature rises by 1.71 ∘C∘C.
A:
Q: 3. Assuming that all the following compounds are planar, circle those that are aromatic, underline…
A: For a structure to be aromatic it must follow the following conditions:-…
Q: The activation energy, Ea, for a particular reaction is 13.6 kJ/mol. If the rate constant at 475 K…
A:
Q: H₂ `N Br CH3 sion: DMSO 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20 10 f1 (ppm) 60000…
A: given commpound is P-bromoacetanilide sites of unsaturation in given compound is 5 detail…
Q: Analyze the following reaction by looking at the electron configurations given below each box. Put a…
A: Answer: For an element its atomic number is always equal to the number of protons present in its…
Q: One quick follow up...this works well with adding 13 moles. Do you use the same math if fewer or…
A: The equilibrium reaction is A3+(aq) +B-(aq) ⇔AB2+(aq) K =2
Q: Based on the data in the figure below, knowing that the nominal volumes of the flask, pipette and…
A: Explanation: In order to determine the accuracy of the three instruments, we need to consider error…
Q: For the following reaction, 0.139 moles of sulfur are mixed with 0.467 moles of carbon monoxide.…
A: It is based on the stoichiometry. Here we are required to find the maximum amount of sulphur…
Q: Determine the equilibrium constant, Kgoal, for the reaction 4PC15 (g) P4(s) +10C12 (g), Kgoal = ? by…
A: Answer: In this question we have to establish the relation between Kgoal, K1 and K2 for the given…
Q: Write the mechanism of imine formation through aminoalcohol reacting butanone with aniline (C6H5NH2)…
A: Imine bond is -C=NH Amino alcohol intermediate is -C(OH)-NH- The lone pair on N atom of Aniline…
Q: Jimmy is doing a reaction. 3 A + B →C+2D that can be modeled as follows: What is the average rate…
A: Given -> Volume= 384 ml = 0.384 L (1L = 1000ml) Initial moles of A = 0.55 moles Final moles of…
Q: Consider the compound. What is the IUPAC name for the compound shown?
A: Given:-
Q: For each bond, show the direction of polarity by selecting the correct partial charges. Si-S CI-S…
A: Introduction Chemical polarity is a term used to describe the uneven distribution of electrons…
Q: KMnO4 H*/H₂O (A) COOH (B) -COOH (C) COOH -COOH (D) -COOH
A: KMnO4 is strong oxidizing agent in the presence of acid and this reagent oxidized to all benzylic…
Q: 2. Identify the general trends of factors affecting solubility in the following table: Condition…
A:
Q: Erwin Schrödinger Orbitals which result from solving the Schrödinger wave equation can be…
A: In physics and chemistry, an orbital is a mathematical function that describes the behavior of an…
Q: Use the molar bond enthalpy data in the table to estimate the value of AHin for the equation - CC14…
A: Given:-
Q: A 8.17 g sample of an unknown salt (MM = 116.82 g/mol) is dissolved in 150.00 g water in a coffee…
A:
Q: Now draw out the products you would expect when this mechanism occurs. H H H H
A: According to the Lewis acid-base theory, the compound that donates electrons acts as a base while…
Q: Classify each of the types of spectra we observed in the following chart: Emission or Absorption…
A: Solutions- We are classify the types of spectra of given substance.
Q: Identify the following compound(s) havingthe lowest boiling point? ОТ || IV O III and IV Н I H HHH…
A: The organic molecules have certain interactions in them which are responsible for their boiling…
Q: Please give example and explanations of each words: Volume Percent Weak Electrolyte Weight Percent
A: An electrolyte is a substance that, when dissolved in a solution or melted, dissociates into ions,…
Q: How many unpaired electrons are there in a nitrogen atom and its ground state
A: Nitrogen is a group 15 , and second period atom.
Q: Name the following aromatic compound using the IUPAC system. NO₂ Cl OH
A:
Q: Lone pairs on nitrogen and oxygen are often the electron source (Lewis base). Circle the electron…
A:
Q: You want to prepare an aqueous solution of ethylene glycol, in which the mole fraction of solute is…
A:
Q: LEFT RIGHT silver (Ag) silver copper (Cu) zinc (Zn) VOLTAGE +0.43 +1.53 MACROSCOPIC OBSERVATIONS…
A: This is a example of redox reaction , where oxidation and reduction process occurs simultaneously.
Q: If a Sodium (Na) atom has 13 neutrons, what is its mass number?
A: The mass number of an atom is the total number of protons and neutrons in the nucleus of the atom.…
Q: Which of the following statements is FALSE? The rate of vaporization for simple molecules with…
A: Vaporization: The conversion of a substance from its liquid state to gaseous state is considered as…
Q: A 15.12 gram sample of iron is heated in the presence of excess sulfur. A metal sulfide is formed…
A: We have to calculate the empirical formula of the compound
Q: For Ca2+ and Mg2+ determinations in a tap water sample, two 50 mL aliquots were pipetted and placed…
A: Volume of water samples = 50 mL Concentration of EDTA4- = 5.07*10-3 mol/L Volume of EDTA4- used…
Q: An ice cube with a mass of 54.2 g at 0.0 °C is added to a glass containing 352 g of water at 45.0…
A: we have to calculate the final temperature of the system
Q: 4.22L of F2 @STP has how many molecules ? round answer to 2 decimal places.
A: Given Volume of gas (V) = 4.22L The volume of gas at STP occupy V = 22.4L…
Q: The specific heat of iron is 0.116 cal/g °C. Assuming that no heat is lost during the experiment,…
A: Given data specific heat of iron = CFe = 0.116 Cal/g-°C Mass of iron. MFe = 30.0 g Initial…
Q: H₂(g) + F₂ (g) 2 H₂(g) + O₂(g) calculate the value of AHxn for 2 HF(g) → 2 H₂O(1) AHrxn 2 F₂ (g) + 2…
A: According to Hess's law change of enthalpy in a chemical reaction is same regardless of whether the…
Q: Atoms of which of the following elements are largest? CI OP OS O None, they are all the same size
A: This is a periodic table topic question.
Q: For the following reaction NO₂(g) + SO₂(g) ⇒ NO(g) + SO3(g) K = 4.15 at a particular temperature. If…
A: Well, first we have to write the equilibrium expression- NO2(g)+SO2(g)⇔NO(g)+SO3(g)now we can write…
Q: Complete the following oxidation-reduction reactions and calculate the Gibbs free energy per mole of…
A: To Complete the following oxidation-reduction reactions and calculate the Gibbs free energy per mole…
Q: Convert a concentration of 6.0x1014 molecules cm-3 of NO2 to (a) ppm and (b) molarity. Assume 25 ⁰C…
A:
Q: Which would have a larger AHvap? A) CH3F B) CH₂O
A: Answer:- This question is answered by using the simple concept of enthalpy of vaporization which…
Q: Which simple ion would the following element be expected to form? sodium, Z = 11 Na+ O Na Na²+ Na³+
A: The electronic configuration of an element refers to the arrangement of electrons in the orbitals of…

Given compound:-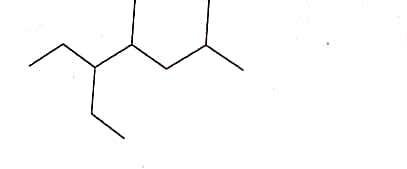
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Name these organic compounds: structure CH3- O O || C OH OH CH3 CH3 | | II C- CH CH₂ - C - OH 1 CH₂-CH- CH3 name acetic acid 0 2-methylpropanoic acidWrite the systematic name of each organic molecule: structure CI CI-CH2 CH-CH2 OH HO || -C-CH2- - CH3 name Х OH CH3-CH2 CH ― CH -C-OH ☐ OHH H For the molecule below, what is the molecular formula and is it a structural, condensed or line formula? H エー H-C- H C -C-C-O-H |H CH3 H
- stion 4 of 20 > bo Draw the expanded, or complete, structural formula for the hydrocarbon represented by the line-angle, or skeletal, structure shown. Macmillan Learning 80 S Draw the complete structural formula. Include all hydrogen atoms. Select 000 000 Draw |||||| C H F5 Templates MacBook Air F6 F7 More DII F8 F9 7 F10 F11 EraseWorksheet 1: IUPAC Nomenclature Use the IUPAC Nomenclature to correctly name the following organic molecules. Name Name H H H H H H FC C C 4 H2C- -CH3 H H. H. H Name Name ннн нн H H H 1 C 21 3 4 C C -H H-C C=C-C-C--H CH3 H H нн Name Name H CH3 H. H Н—С — Н H H H- с —Н CH3 H. H. H H Name Name C2H5 H H H нннн нн 1 2 3 4 6. H- -H H-C-C-CFC,-C-C-H H H H CH3 H. H H H Н-С-н H.Write the systematic name of each organic molecule: structure O H-C-CH-CH3 OH || CH3–CH2–CH2–CH-C-H CH3 O OH 0=0 HO–CH2–CH2–CH2CH-C-H name 0 | 0
- CH,-CH-CH-CH, a-ÇH-CH,-CH, CH, CH,-CH-CH,-Ca CH,Write the systematic name of each organic molecule: (10 HO—C—CH–CH2–CH–CH2–CH2−C CH3 I CH3 structure || HO—C—CH—CH,—OH CH3 CH3 CI-CH₂-CH-CH₂-C-OH name 0Question-1 Answer the required items for the following compounds 1- C,H&F2 2- Trans-Dichloro -2-butene 3- CH;COOH 4- (CH3)2СHCCCH 5- СНЗСН2CНО [Name, Structural formula ] [ Skeletal, Condensed formula] [ Name, Functional group] [Name, Functional group] [Name, Functional group] 6- C5H100 [ Name, Condensed formula , Functional group] 7-2,3,4-Tribromo cyclopentene [Skeletal formula] [ Skeletal formula ] [ Name Condensed 8- Methylcyclobutane 9- C,H,COOCH3 10- CH3CH2CH,CH2CH2CHCH2 [ Name, Skeletal formula , Functional group ] Add file
- Write the systematic name of each organic molecule: CH3 HỌC—CH2=CH-CH2−C O || HO CI Equ CH3-CH-CH₂-CH-CH-C-OH CH3 CH3 structure 0=0 O 010 C—CH–CH2–CH2CH3 CH3 name 0 10 0 Xffset%3Dnext&assignmentProblemID=D170497300 I Re Part A What is the IUPAC name for the following compound? CH3-CH2-CH2-CH2-CH2-CH2-CH2-CH2 -CH2-CH3 Enter the name of the molecule. > View Available Hint(s) Submit Part BQ2\ A\ Give the nomenclature for the following compounds by IUPAC system? || -С-ОН C3H1-O H3C-O- -CH3 H-C-H C5H,Cl

