Q: CH3CH2CH2CH2CHO) is treated with each reagent.
A:
Q: Draw the products formed when each compound is treated with CH;CH,COCI, AICI3. CH(CH3)2 N(CH)2 Br…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: Draw the major stereoisomer formed when attached compound is treatedwith NaOH.
A: The stereo chemical requirement follows the E2 elimination reaction that is leaving the group and…
Q: Draw the products formed when A and B are treated with each of the following reagents: (a) Br2,…
A: (a) .Br2, FeBr3:
Q: Draw the product formed when each compound is treated with NaNO2 and HCl.
A: Primary and secondary amines on reaction with NaNO2 in the presence of HCl yield different products.…
Q: What product is formed when each compound is treated with K 2Cr 2O 7? With some compounds, no…
A: Potassium dichromate (K2Cr2O7) is an inorganic reagent and most commonly used as an oxidizing agent…
Q: Draw the organic products formed when CH,CH,C=C Na reacts with BRCH,CH,CH,CH,OH. Higher mass product…
A: The reactants given are and .
Q: Draw the products formed when CH3CH2C=C Na+ reacts with each compound. a. CH3CH2CH2Br…
A: a). In the reaction, butylate anion behaves as a strong nucleophile which undergoes nucleophilic…
Q: Draw the products formed when each compound is treated with HNO3 and H2SO4.
A: EXPLANATION: a) part -OCH3 is ortho, para director whereas -COOCH3 is a meta director.Because -OCH3…
Q: t-BUOK -> A is the reacich SN1,SN2 ET or E2 and wnat is the Br majar product. er EtONa
A:
Q: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous OH?
A: a). The cyclic product is given as
Q: Draw the products formed when CH3CH,C=CCH,CH3 is treated with each reagent: (a) Brz (2 equiv); (b)…
A: To find: The products formed when 3-hexyne is treated with each of the given reagent.
Q: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous OH?
A: a) The cyclic product is given as The given 1,5-dicarbonyl compound (A) undergoes self condensation…
Q: 6. Draw the products formed from nitration of each compound OH OH b. а. CH3 ČHa
A:
Q: 3) Which aa has the strongest acid side chain? b) H2N-CH-ċ-OH H2N- -сн—с—он CH2 CH2 CH2 CH2 =0 O:…
A: Solution: We know the carboxylic acids are most acidic than other organic compounds like alcohol,…
Q: OH) is treated
A: When phenol reacts with concentrated H2SO4, it produces a mixture of o- and p- products, with the o-…
Q: Draw the product formed when phenylacetaldehyde (CsH;CH,CHO) is treated with each reagent.…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Give the IUPAC name for each aldehyde.
A: Concept introduction: For naming an aldehyde in IUPAC nomenclature, the suffix –al is added to the…
Q: 8.36 What is the major stereoisomer formed when each alkyl halide is treated with KOC(CH3)3? Br a.…
A: In chemistry, dehydrohalogenation is the process in which an elimination reaction removes a hydrogen…
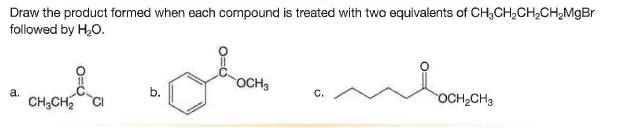
Q: Draw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some…
A:
Q: Draw the product formed when pentanal (CH;CH,CH2CH;CHO) is treated with each reagent. With some…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: What reagent is needed to convert (CH3)2CHCH2COCl to each compound?
A: a. The given compound is aldehyde. Acid chlorides on reaction with mild reducing agent like lithium…
Q: Draw the products formed when each ether is treated with two equivalents of HBr. a.…
A: The nucleophilic attack of the HBr on the electrophilic carbon favored the reaction. Ethers are…
Q: Givé the organic product: 1 KON D
A: Michael Addition - This is a versatile carbon-carbon (C-C) bond forming reaction which is also known…
Q: What
A: First OH- attack on carbonyl carbon and then π bond transfer towards more electronegative atom…
Q: Draw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH…
A:
Q: 6. Draw the products formed from nitration of each compound он он a. CH3
A:
Q: What Wittig reagent and carbonyl compound are needed to prepare each alkene? When two routes are…
A: a.
Q: β-D-Glucose, a hemiacetal, can be converted to a mixture of acetals on treatment with CH3OH in the…
A: A stepwise mechanism for this reaction has to be drawn. The reason two acetals are formed from a…
Q: 2 NaOH Hz propan-2-ol CH;NH, но CH3 PCI5 propan-2-ol NaOH(aq) Tollen's reagent LIAIH,
A: Organic reaction mechanisms:
Q: Draw the product formed when C6H5N2 +Cl− reacts with each compound.
A: a) Diazonium salt C6H5N2 +Cl− reacts with aniline to give p-aniline azo benzene It is an…
Q: Draw the major stereoisomer formed when attached compound is treatedwith NaOH.
A: Stereochemical requirement of E2 elemnationn reaction is that the leaving group and the beta H…
Q: 2 CH;CH,-OH) → + HC CHs
A: Ketone on treatment with alcohol in the presence of acid catalyst produces ketal.
Q: Draw the products formed from the acid–base reaction of H2SO4 with each compound.
A:
Q: Draw the organic products formed when allylic alcohol A is treated with each reagent.a.H2 + Pd-C…
A: (a). Catalytic hydrogenation of allylic alcohol reduces the double bond to single bond giving…
Q: Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. e.…
A: FeBr3 acts as a Lewis acid and it reacts with Br2 and produces Br+ Phenol reacts with Br+ and…
Q: NO2 reagent E reagent A reagent B reagent D CI reagent C OH
A: Reaction A is known as the nitration of benzene because the product forming is a having a nitrate…
Q: Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b.…
A:
Q: Even though B contains three ester groups, a single Dieckmann product results when B is treated with…
A: To find: The structure of the product and explanation
Q: Which compound is hydrated more rapidly?
A: The compound which will have more stable intermediate i.e carbocation formed during the addition of…
Q: Draw the products formed when CH;CH,C= CCH,CH, is treated with each reagent: (a) Br2 (2 equiv): (b)…
A: What will be the products for the bromination and chlorination of .
Q: Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each…
A: Wittig reaction involves synthesis of alkene from aldehyde and phosphorus ylide. Ylide on treatment…
Q: Draw the major stereoisomer formed when each compound is treated with NaOH.
A: A species with a larger size can easily accommodate a negative charge. In other words, in a large…
Q: What is the product formed in a-b when treated with one equivalent of HBr?
A: The addition of HBr to an alkene shows an electrophilic substitution reaction. In this reaction,…
Q: 31. (8) Provide one reagent that will complete each transf (a) (b) OH -OH ? он ? (c) ∞ -CH₂OH ? 요요 a…
A: You have posted multiple questions but I am solving only first three .if You want solution of…
Q: halide with H2O? benzyl alcohol Ph;COH cydohexanol (CH3)CHOH 1-propanol A B D E
A: Unimolecular nucleophilic substitution (SN1) reaction proceeds via formation of carbocation…
Q: Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each…
A: Wittig reaction involves synthesis of alkene from aldehyde and phosphorus ylide. Ylide on treatment…
Q: Draw the products formed when each ether is treated with two equivalents of HBr.
A: Note: Since you have asked multiple questions, we will solve the first for you. If you want any…
Q: Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d.…
A: Phenol reacts with an acid chloride in the presence of Lewis acid to give Fridel-Kraft acylation…
Q: Draw the product formed when C6H5N2+Cl− reacts with each compound.
A: Diazonium salt is formed by the reaction of aniline with NaNO2 in the presence of HCl and this…
Step by step
Solved in 4 steps with 7 images

- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2ODraw the product formed when (CH3)2CHOH is treated with each reagent. a.SOCl2, pyridine b. TsCl, pyridine c.H2SO4 d.HBr e.PBr3, then NaCN f.POCl3, pyridineDraw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridine
- Label the α and β carbons in each alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.Draw the products formed when CH3CH2C=C Na+ reacts with each compound. a. CH3CH2CH2Br b.(CH3)2CHCH2CH2Cl c.(CH3CH2)3CCl d.BrCH2CH2CH2CH2OH e. ethylene oxide followed by H2O f.propene oxide followed by H2O1. Draw the product formed when phenylacetaldehyde (C6H5CH2CHO) is treated with each reagent:
- Draw the products formed when A is treated with each reagent: (a) H2 + Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H2SO4, H2O; (e) Sharpless reagent with (+)-DET.Draw the product formed when (CH3)2CHOH is treated with following reagent. H2SO4What is the major stereoisomer formed when each alkyl halide is treated with KOC(CH3)3?
- Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.Draw the products formed when (CH3)2C=CH2 is treated with each reagent.a. HBrb. H2OH2SO4c. CH3CH2OH, H2SO4d. Cl2e. Br2, H2Of. NBS (aqueous DMSO)g. [1]BH3;[2]H2O2, HO-Draw the products (including stereoisomers) formed when benzaldehyde (C6H5CHO) is treated with each Wittig reagent.

