Q: The best true statement for voltaic cell? E for the cell is always positive. O The standard reductio...
A: Electrochemical cell are those cell which converts electrical energy to chemical energy or vice vers...
Q: A clear, colorless solution forms a pale yellow precipitate when KI(aq) is added. The same solution ...
A: The solubility of an ionic compound in water depends on the nature of the ions present in the ionic ...
Q: How much heat (kJ) is evolved when 4.50 g of Fe203 is reacted with excess carbon monoxide using the ...
A: Given Data :
Q: There are two steps in the extraction of copper metal from chalcocite, a copper ore. In the first st...
A: Chemical reactions can be added, subtracted, or multiplied like an algebraic equation.
Q: What is the average mass, in grams, of one Zna Multiple Choice O 1661 x 10 24 O 65.39 amu O 65.39 g ...
A: Molar mass of Zn = 65.38 g/mol
Q: If 87 J of heat are added to a balloon, causing it to expand from 1.0 L to 1.4 L under 0.97 atm of p...
A:
Q: Complete the following net ionic equations by choosing the correct chemical formula for the product ...
A: Ag^+(aq). + Br^-(aq). -->
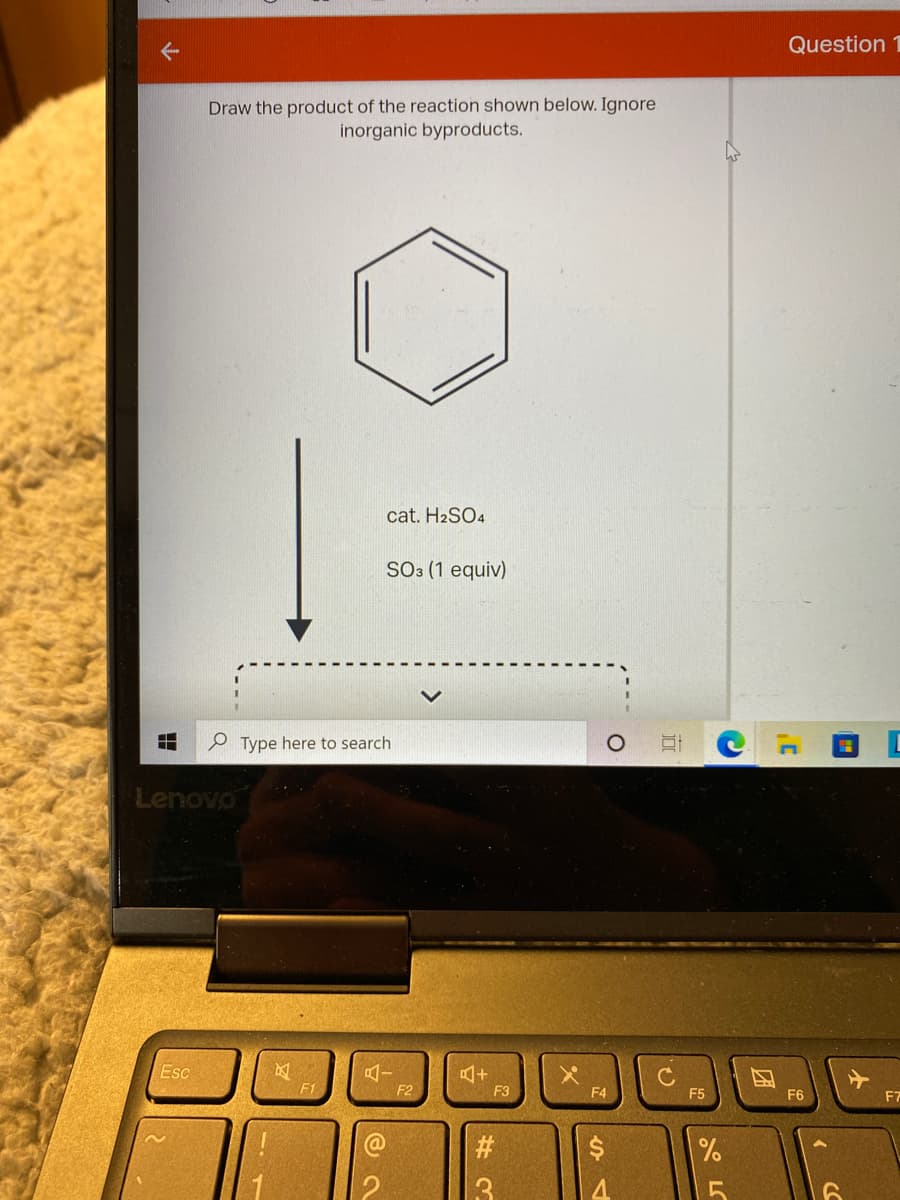
Q: 3. Determine the product of the following sequence. ZnCl2, HCI OH 1) TSCI, pyr 2) KOTBU Он socl2, py...
A: Few questions based on introduction to organic chemistry that is to be accomplished.
Q: An analytical chemist is titrating 139.8 mL of a 0.1600M solution of aniline (C,H,NH,) with a 0.6500...
A: Given that, 139.8 mL of 0.1600 M of aniline was titrated with 0.6500 M solution of HNO3. The pKb of...
Q: mass
A:
Q: Complete and balance the equation Co(C2H3O2)2•4H2O
A: A balanced equation can be defined as the equation in which equal number of atoms of each element is...
Q: Alcohols A and B formed layers with Lucas reagent within 10 minutes. However, compound A formed brow...
A: Lucas reagent is a mixture of ZnCl2 in presence of HCl.
Q: Propose a synthetic scheme for the synthesis of 3-pentanone from ethyl bromide and cyanide as your c...
A: To synthesize the ketone product detail solution is given below
Q: 2. How many signals in the 13C NMR spectrum would you expect for each of the two isomers of azulene
A: Azulene is an organic compound and an isomer of naphthalene. These both are aromatic compound.
Q: Name the highlighted chemical group in each molecule. Lewis structure name of highlighted group Н—о:...
A: We have to identify the highlighted group.
Q: make
A: Given statement is true.
Q: If the pressure exerted on a liquid is increased, while the temperature is held constant, what type ...
A: A phase diagram is given. We have to tell if the pressure exerted on a liquid is increased, while th...
Q: Can you solve the problems below, there's 2 pages. The page with the number 1 on the top corner is t...
A: Salt-1 Na2CO3 Mass of empty beaker = 25.468 g Mass of beake...
Q: Please answer fast There is a 20 cm-1 distance between two consecutive lines in the microwave spect...
A: The solution of the question is given below:
Q: Please give a clear handwritten answer of all reactions sub parts give net ionic equations:- 2Fe...
A: Interpretation- We have to write the net ionic equation of the given molecular equations as given ab...
Q: energy, in J, is
A:
Q: In SOAP Experiment, why did saponification reaction require the long period of stirring?
A: Saponification is the process of reacting oil/fat with sodium hydroxide or potassium hydroxide base....
Q: 3- Qualitative analysis : establishes the chemical identity of the species in the sample like (solid...
A: (1) Qualitative analysis :- Qualitative analysis concern with detection of elements or radicals in ...
Q: e mass percent
A:
Q: Mass of beaker 50.5g Mass of marble before reaction 3.0g Mass of marble after reaction 1....
A: Formula of marble CaCO3 Chemical reaction CaCO3 (s) + 2 HCl (aq) →CaCl2 (aq) + CO2 (g) + H2O (l) M...
Q: Compute for the numerical value of the gravimetric factor required to convert the given precipitate ...
A: 1. Solution - According to the question - Given - Gravimetric factor of P in (MgNH4PO4) = Molar mass...
Q: 32. Chloroform, CHC13, is used in a laboratory experiment to extract bromine. What is the percent of...
A: The atomic mass of H is 1 and Cl is 35.45 and C is 12
Q: 5.32 A sample of sodium-24 with an activity of 12 mCi is used to study the rate of blood flow in the...
A: Initial activity of sodium-24 = 12 mCi Half-life of sodium-24 = 15 h The question requires to calc...
Q: 6) Using arrows to represent electrons, Construct the orbital diagram of the Cr and Cr3+ ion. Cr Cr3...
A: The question is regarding electronic configuration of elements. General trends of size and electron ...
Q: 14. One 1iter of a solution contains 0.010 mole of AgNO and 2.0 mole of NHOH. (a) 0.010 mole of NaCl...
A: Solution -
Q: Explain the reason that the conversion of liquid water to gaseous water has a positive change in ent...
A: Given statement is : Explain the reason that the conversion of liquid water to gaseous water has po...
Q: Write an informative introduction about Matter.and Mixtures.
A: A substance can be defined as a material that has physical properties.It is basically a form of matt...
Q: How would you carry out the following reactions? (a) CHJCH2C=CH CH3CH3CCH, (bi CH,CH2C=CH CH3CH2CH2C...
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for you...
Q: Acetic acid (HC2H3O2) is a weak acid (Kg 1.8 x10). Calculate the pH of e 17.6 M HC2H3O2 solution. 1 ...
A: Acetic acid is a weak acid which dissociates partially .Alpha is degree of dissociation.
Q: The diagram below represents an operating electrolytic cell used to plate silver onto a nickel key. ...
A:
Q: Given a solution of 0.1 M Ag+ and O.2M of Pb2+, (Ksp for silver oxalate = 3.5x10^-11 and ksp for lea...
A: Solubility product Ksp is the product of ionic concentration required to precipitate any insoluble s...
Q: Illustrate which is the stronger acid using Lewis structures: а. HNO; or HCI4 b. CH2FCOOH or CH2BrCO...
A:
Q: A 10.00-L vessel at 515 K initially contains HI(g) at a pressure of 5.60 atm; at equilibrium, it is ...
A:
Q: The organic bases cocaine (pKb = 8.41) and codeine (pKb = 7.95) react with hydrochloric acid to form...
A: Given, Codaiene pKb=7.95 Cocaine , pKb=8.41
Q: What is the solubility of PbF₂ in a solution that contains 0.0550 M F⁻ ions? (Ksp of PbF₂ is 3.60 × ...
A: The solubility of any salt is defined as the how much the given solute can be dissolved completely i...
Q: A chemistry graduate student is given 250. mL of a 1.40M trimethylamine ((CH,) N) solution. Trimethy...
A:
Q: In the potential energy diagram, which letter represents the potential energy of the activated compl...
A: A chemical reaction occur only if reactant posses enough energy that lead to crossing activation ene...
Q: Consider the reaction: H + ¿n → 2¾H + ¿n + _}H. Calculate the energy change for this reaction. Is er...
A: Energy relesed in a nuclear reaction is, E= E=Δm×931MV
Q: spontaneous. Write an equation for the coupled process and calculate the equilibrium constant for th...
A:
Q: What is the absolute error on the calculated milligrams of iron in this sample? a. ± 10 mg b. ±3 mg ...
A: No of moles = Molarity * Volume (L) Mass = Moles * Molar mass
Q: The diagram below represents an operating electrolytic cell used to plate silver onto a nickel key. ...
A: Electroplating as the name suggests is plating of a surface using electricity
Q: corresponding to your answer. a. Ammonia and ammonium chloride b. Acetic acid and potassium acetate ...
A:
Q: Answer the following with complete solution.
A:
Q: Hydrofluoric acid and water react to form fluoride anion and hydronium cation, like this: HF(aq)+H,O...
A: Given data,Amount of HF at equilibrium=3.83gAmount of F- at equilibrium=0.293gAmount of H3O+ at equi...
Q: Describes the differences and similarities between solids, liquids and gases on a molecular level fo...
A: Solid - - > The substances which are having very high intermolecular forces between thier molecul...
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- 1. Why are solutions of KMnO4 and Na2S2O3 generally stored in dark reagent bottles?Need help on last question. The reaction is a fisher esterification. My unknown ester is pentyl acetate (amyl acetate) and my unknown alcohol is 1-pentanol. 1.5 mL of 1-pentanol used giving us 0.013806 moles used. 3.0 mL of acetic acid used giving us 0.052604 moles used. Please show all work (including identification of limiting reactant)2 A. could there be secondary product? B. Indicate why mud crystal is used in the reaction.
- Write short note on distereomers properties with a suitable example ?What is the balanced equation for the 4-ChIorobenzyI AIcohoI into 4-ChIorobenzoic Acid. Illustrate the haIf reactions then balance it just like how the other redox reactions are balanced.Please help with both parts! 41 and 42. An explanation for each one would be super!
- What is Solubility of phenazopyridine in water?acidic media? basic media ?and what is most common solvent?please don't provide handwrittin solution.. What is name of this structure? Give explantion and steps for supporting.Chapter #12, Question #4: Water from a lake in an area with limestone bedrock has a chemical composition as on p. 310. If the organic matter (HM) content of the water is 12 mg L-1, show that charge balance is adequately maintained. ***If you find that charge balance is not maintained from the tabulated concentrations, calculate the carboxylate concentration (CCOO−) in the HM.
- Compute the amount of reagents which will be used for the preparation of the following reagents. Show your computation neatly. 2 L of 5% (NH4)2C2O4 e. 1.5 L of 0.04 M KMnO4 500 mL of 0.1% (NH4)2C2O4 f. 3 L of 1 M H2SO4 1 L of 6 M NH4OH g. 500 mL of 0.1 M HCl 2 L of 0.1 M NaOH Describe the preparation and storage of each reagents.What happens when concentrated NH3 is added to (NH4) 2CO3? and What happens after washing with [Co (NH3) 4CO3] NO3 ethanol? please answer the two questionPlease answer the question at the bottom, Thanks. LAB NOTES FOR PREPARATION OF BUTYL MAGNESIUM BROMIDEAND ITS SUBSEQUENT CONVERSION TO AN ALCOHOL To the reaction flask were added 2.5967 gram of magnesium turnings, about 20 mlof diglyme, and a solution of 13.5942 gram of butyl bromide in about 25 mldiglyme.The procedure as per the lab manual was initiated. After the required heating timewas completed the reaction mixture was allowed to cool to room temperature.Then a solution of 6.1123 gram of acetone in about 15 ml of diglyme was addeddropwise as per the lab manual.The rest of the lab manual procedure was completed, ultimately affording 2.5532gram of an oily, clear and slightly yellow liquid, presumed to be 2-methyl-2-hexanol. Boiling point as measured by distillation was 138 – 141 deg C.About 0.8 ml of the presumed 2-methyl-2-hexanol was placed in an NMR tube towhich was added 2 drops of TMS. The tube was capped, then inverted and righted20 times to thoroughly mix the product and…



