Q: CI. OH 6.6 Draw the structure of the product.
A:
Q: What volume in L of a 0.320 M Mg(NO3)2 solution contains 45.0 g of Mg(NO3)2 ?
A: Since, Molarity is the number of moles of solute dissolve in one litre of the solution. Thus,
Q: While in Europe, if you drive 115 km per day, how much money would you spend on gas in one week if…
A: Money spend in one week can be calculated from the cost of one day.
Q: ow many milliliters of an 80% (v/v) solution of isopropyl alcohol would you need to make 382 mL of a…
A: The dilution formula is: C1V1=C2V2 ......(1) Here, C1 is the concentration of concentrated solution,…
Q: In the titration of 220.0 mL of 0.4000 M HONH, with 0.2000 M HBr, how many mL of HBr are required to…
A:
Q: Select the correct IUPAC name for the following organic substrate, including the R or S designation…
A:
Q: According to Schwabe and Wagner, the heats of combustion in a constant-volume calorimeter for…
A:
Q: Can someone help me to know the detailed mechanism of this transesterification reaction for…
A:
Q: Write the full and abbreviated electron configurations for bromine. How many core and valance…
A: we have to write electron configuration of Br and determine the number of core and valence electrons…
Q: broad singlet, 1H (a) NMR Spectrum A quartet, 1H (b) 3 PPM multiplet, 7H (c) -~ 2 1 doublet, 3H (d)
A: Labile protons come as broad peak in 1H NMR Number of signal = number of sets of protons in…
Q: A compound formula with brackets is called a coordination complex, and the structure looks like a…
A:
Q: 13. Write the full and abbreviated electron configurations for calcium dication. How many core and…
A: Given : ion = calcium fixation Tip : atomic no. Of Ca = 20
Q: Name the following compounds: 6. 7. 8. 9. 10. CH₂-CH₂ HỌC-C-CH, CH CH3 CI CI Br-CH-CH-CH₂…
A: All the compounds given are hydrocarbons except the second one having single, double, and triple…
Q: Follow the curved arrows and draw the product of an SN2 reaction shown below. Include all lone…
A:
Q: What is the classification for the following halide? $ Br A quaternary halide B primary halide C)…
A: When halogen atoms are attached with carbon atoms it forms alkyl halides .
Q: Name the following compounds:
A: We have to determine the IUPAC name of the structures given
Q: Which of the following processes are endothermic? O None of the above are endothermic. O2 Br(g)…
A: The energy difference between the products and reactants is known as the energy change of a…
Q: uestion 5 Which of these structures represent the same compound? CH₂ H3C CH₂ CH₂ CH3 H3C H3C CH₂ (B)…
A:
Q: Which number is smaller? 9.997 x 10-6 km or 9.997 x 10.2 cm
A:
Q: A newborn kitten weighs 0.158 kg. What is the kittens mass in ounces ? Show your work
A:
Q: For the reaction KCIO4 → KCI + 20₂ assign oxidation numbers to each element on each side of the…
A: Here we have to determine the oxidation number of the atoms in the redox reaction.
Q: Question 21 of 21 300.0 mL of a 0.400 M solution of Nal is diluted to 700.0 mL. What is the new…
A:
Q: Explain whether or not it will result in a usable standard solution, and why. (Assume appropriate…
A: To explain whether the solution prepared on mixing of 0.500 moles of Fe+3 and 0.00500 moles of SCN-1…
Q: 2Hg²+ (aq) + 2e Hg₂²+ (aq) + 2e¯ calculate AG Hg₂+ (aq) Mac Hg₂ 2+ (aq) 2Hg(1) = 0.92 V Eº = 0.85 V…
A:
Q: A 65.0 mL solution of 0.121 M sulfurous acid (H₂SO3) is titrated with 0.121 M NaOH. The pK₁ values…
A: According to the question we have, The concentration of the sulfurous acid H2SO3 is given by = 0.121…
Q: Draw a structural formula for the product formed upon hydroboration/oxidation of the alkene below.…
A:
Q: A solution contains 7.02x10-3 M barium acetate and 1.38x102 M calcium nitrate. Solid sodium chromate…
A: Concentration of barium acetate = 7.02×10–3 M Concentration of calcium nitrate = 1.38×10–2 M
Q: Provide the structure(s) of the expected major organic product(s). Unless mentioned otherwise, you…
A: In SN2 reaction, Inversion of configuration occurs. In SN1 reaction, more stable carbocation leads…
Q: Cd(s) + Fe²+ (aq) yes - 0 E (Fe²+ / Fe) = -0.4400 V, E° (Cd²+ / Cd) = -0.4000 V Calculate the emf…
A:
Q: Using the thermodynamic information, calculate the standard reaction free energy for the reaction…
A:
Q: The following equation describes the oxidation and ethanol to acetic acid by potassium permanganate…
A: First we will find the limiting reagent and using the balance reaction we will calculate the…
Q: Using the appropriate electron affinities and ionization energies, estimate the enthalpy of reaction…
A: Enthalpy of reaction is defined as the difference in enthalpy that occurs when products are formed…
Q: Part A Provide the major organic product of the following reaction. Br NaOCH3 CH₂OH Draw the…
A: Alkyl halide in the presence of a strong base undergo elimination reaction to give alkene as the…
Q: Draw structural formulas for the isomeric carbocation intermediates formed on treat- ment of each…
A: Carbocation (carbon + cation) is defined as the carbon having +ve charge along with the three bonds.…
Q: When 1 mole of CO₂ is formed from the elements at 25 °C and 1 atm, 94,272 cal is liberated. The…
A:
Q: During a Blood Donation 1.00pint (pt) of blood is removed from a donor. What is The volume of this…
A: Pint is unit for volume
Q: 3.2) The energy required to dislod effect is 275kJ/mol. What wave photon to dislodge an election…
A:
Q: 2. Buffer capacity has a rather loose definition, yet it is an important property of buffers. A…
A: We have to answer questions 2, 3a. and 3b. For that, the data table given is:
Q: 1 10J 1 MJ 1 m J = = 11 = 10 -1, 10 3 10 0 J J 100 J w by filling in the missing prefix μ X
A: Energy is represented in terms of joule(J). Different representation of unit of energy: Decijoule…
Q: Calculate the temperature change of imole of a perfect diatomic gas contained in a piston. q = 65J…
A: Given, The gas is dia atomic Number of moles = 1 mol q = 65 J w = 210 J
Q: Based on the thermodynamic properties provided for water, determine the amount of energy needed for…
A:
Q: a mechanism for the formation of the latter product. Propose Propose a mechanism for the reaction. &…
A: OH attached with most substituted carbon in reaction
Q: Count the significant digits in each of these measurements: measurement - 7.0 x 10 kJ/mol -2 1.3 x…
A: Rule of significant digit-1) In non decimal number, 0 comes after non-zero not count in significant…
Q: When a sample of a hydrated CaBr was heated to a constant weight, 3.24 g of the anhydrous (dry) CaBr…
A:
Q: Identify the type of below reaction: 2 NaCl + F. F₂ Decomposition Single-displacement Combination…
A: The different type of reactions are defined as : 1) Decomposition reaction: In this reaction is…
Q: Chapter 8 Problem 171 Provide the structure of the major organic product of the following reaction.…
A: Base can abstract beta hydrogen and form alkene as elimination product .But if more than one beta…
Q: A chemistry student needs to standardize a fresh solution of sodium hydroxide. He carefully weighs…
A: Given: Mass of oxalic acid = 10 mg Volume of oxalic acid = 250 mL Volume of NaOH solution = 12.2 mL…
Q: What is the percent concentration of a solution containing 68945 mg of solute dissolved in 67.77 mL…
A:
Q: Fill in the missing information: symbol Ge atom or ion? check all that apply O neutral atom neutral…
A:
Q: 21. Order the following atoms in increasing first ionization energy: Cr, O, P, Rb
A: Given : atoms Tip : on moving top to bottom ionization energy decreases.
Draw the skeletal formula of A, B and C
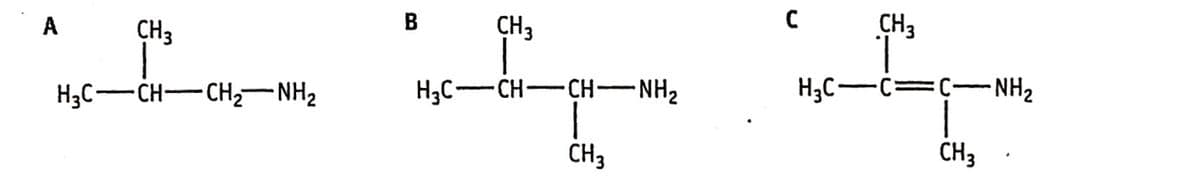
Step by step
Solved in 2 steps with 1 images


