Chapter20: Nucleophilic Substitution Reactions: Competing Nucleophiles
Section: Chapter Questions
Problem 9Q
Related questions
Question
100%
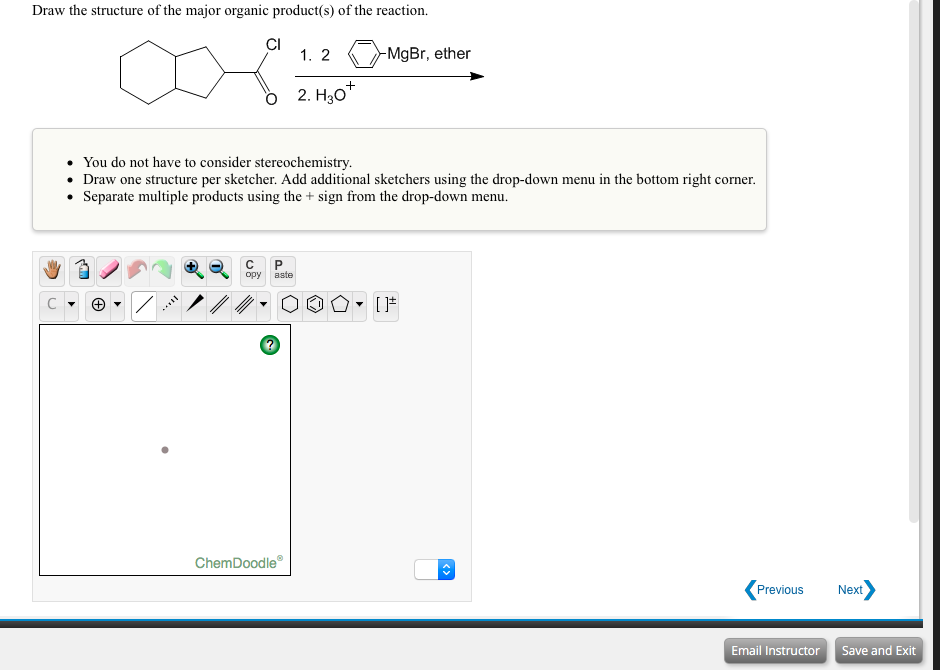
Transcribed Image Text:Draw the structure of the major organic product(s) of the reaction.
CI
1. 2
-MgBr, ether
+
2. H30*
You do not have to consider stereochemistry.
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
• Separate multiple products using the + sign from the drop-down menu.
opy
aste
ChemDoodle
Previous
Next
Email Instructor
Save and Exit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT