Q: Match the unknowns with their correct class of organic compounds based on the given qualitative…
A:
Q: Rank the following compounds in order of increasing basicity in aqueous solution, east basic first…
A:
Q: Draw the products formed when each attached amine is treated with [1] CH3I(excess); [2] Ag2O; [3] Δ.…
A: In the given question we have to do the stepwise Reaction. 1,2-dimethylcyclopentanamine reacts with…
Q: 4. Why must the pH of imine formation be controlled at 4.5? i
A:
Q: Why is acetamide a much weaker base than ammonia? Select one: a. The adjacent C-O makes the lone…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: cOOEt -014 resovcinol ethyl accteacebate methylumbelliferone 04 conduyote acid . -0lt Ho. -OH olt Ho…
A: Chem sketch of given diagram has to be given,
Q: Identify the most and the least acidic compound in each of the following sets. Leave the remaining…
A: Carboxylic acids are weak acidic in nature. If an electron-withdrawing group attached to the…
Q: Determine the expected result of each amine test for the following compounds. Draw the resulting…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Show how you would use appropriate acyl chlorides and amines to synthesize thefollowing amides.(a)…
A: (a) Amines are undergoing a condensation reaction with acyl chlorides to give amide. The…
Q: Given that the pKa of carbonic acid (H2CO3) is 6.4, is sodium bicarbonate strong enough of a base to…
A: Sodium bicarbonate, NaHCO3(aq) is a conjugate base of carbonic acid, H2CO3(aq). For carbonic acid,…
Q: What aldehyde or ketone and nitrogen component are needed to synthesize N-ethylcyclohexanamine by a…
A: There are two conditions by which the synthesis of N-ethylcyclohexanamine can be done by a reductive…
Q: Diethylstilbestrol (DES) was given to pregnant women to prevent miscarriage, until it was found that…
A: Estradiol has four rings fused together. Diethylstilbesterol (DES) has only two rings joined by…
Q: (ii) Give the structure and general name of 2-aminopropanoic acid when in a neutral solution of pH =…
A: 2 amino propanoic acid at 7 pH exist in neutral form
Q: 4. Calculate the ratio of the concentrations of a thiolate (A) and its corresponding thiol (HA) at a…
A: a) Given : pH of solution of HA = 5.5 And pKa of HA = 10.3 => Ka = 10-pKa = 10-10.3 = 5.012 X…
Q: What is the purpose of the amine in the following reaction: ROH + SOCI, + R'3N – RCI + SO2 + HCI a.…
A: When base reacts with acid forms salts, these salts are soluble in water, after completion of…
Q: Which compound is not shown? NHCCH3 A. aspirin OB. saccharin OC. acetanilide O D. acetaminophen O E.…
A: The structure which is not shown is .
Q: The following compound is derived from one of the twenty mammaliam "amino acids". It is the ethyl…
A: Highly basic Nitrogen protonated. Guanidine moiety is highly basic than secondary amine so…
Q: 2. Identify the protection group on the AMINE of the amino acid. Вос Fmoc МОМ TMS Benzyl
A: A protecting group or protective group is introduced into a molecule by chemical modification of a…
Q: Isoprenaline is an adrenergic agonist. The isoprenaline analogue shown below demonstrates a dramatic…
A: The answer is given below.
Q: Which of the following statements is correct regarding aliphatic amines? Select one: O a. aliphatic…
A: Amine are the compounds having -NH2 as a functional group in them.
Q: The pkb of methylamine, CH3NH2, is 3.36. Calculate the pk, of its conjugate acid, CH3NH3*. pka =
A:
Q: The pKa values for the carboxy and ammonium protons of phenylalanine are 2.58 and 9.24,…
A: The compounds in which both amino (NH2) and carboxylic acid (COOH) group are present are known as…
Q: The pKa of acetamide (CH3CONH2) is 16. Draw the structure for its conjugate base and explain why…
A: In the acetamide, nitrogen is more electronegative than carbon. Thus, the conjugate base is formed…
Q: When a amide is treated with SOCl2 ? What arises ?
A: Amides are the organic compounds which have the general formula RCONH2. It undergo dehydration in…
Q: 4. Ciprofloxacin is a quinolone antibiotic that functions by inhibiting the bacterial enzymes DNA…
A: PKa = -log Ka Where Ka is the dissociation constant. With increasing in the dissociation ka value…
Q: Following is the structure of pyridoxamine. CH2NH2 CH2OH но. H3C N. a) Which nitrogen atom of…
A: Please find your solution below : (a) Amines are Lewis base and hence act as electron donor. The…
Q: Aniline (conjugate acid pKa 4.63) is a considerably stronger base than diphenylamine (pKa 0.79).…
A: In aniline the lone pair of nitrogen atom is delocalized on one benzene ring only.
Q: Rank the attached compounds in order of increasing basicity andexplain the order you chose.
A: The resonance structures of pyrrole are shown below. The above structures imply that lone pair of…
Q: Protonation of 1,1,3,3-tetramethyl-2-X-guanidine has been shown to occur on the imino nitrogen when…
A: In Hammett plots, ρ is given as the slope of the curve of log k versus the substituent constants, σ.
Q: Explain the Mechanism - Amide Hydrolysis in Base ?
A: The organic compound in which the carbonyl carbon atom is bonded to the nitrogen atom by a single…
Q: Show how you would use appropriate acyl chlorides and amines to synthesize thefollowing amides.(a)…
A: (a) Amines are undergoing a condensation reaction with acyl chlorides to give amide. The…
Q: 22. The stability of carbocation can be accounted by which of the following structural effect? a.…
A: Please find your solution below : 22. The stability of carbocation can be accounted by both…
Q: Name the following compound: [(NH3)5Co(NH2)Co(NH3)5]Cl5 H-aminobis [tetraamminecobalt(III)] chloride…
A: Co-ordination compounds are formed when the metal ion is bonded to the ligand which donates its…
Q: How does methyl 2-acetoxybenzoate or Aspirin methyl ester hydrolise. Please show detailes.
A:
Q: Calculate the pKa of a acid at 25°C if its conjugate base has a pKb = 1.19
A: The relation between pKa and pKb is given as: pKa + pKb = pKw ..........(1) Also, pKa = -logKa pKb =…
Q: Which form of amphetamine would you expect to be present at pH 1.0?
A: The structure of amphetamine is given as:-
Q: What are A and B in the given sequence, ?respectively conc. HNO3 i) Sn, conc. HCI B A ) NaOH sol…
A: Organic reactions are chemical reactions which involve organic compounds. The example of organic…
Q: Which group is a stronger activator between the amino group of aniline and the acetamido group of…
A: Ring activators are groups that increase the electron density on the benzene ring making the ring…
Q: For amines A, B, and C below: NH₂ A Problem viewing the image, Click Preview Here NH₂ B C 1.1) Rank…
A:
Q: What percent of acetic acid is present in the acidic form at pH 5.0, assuming a pKa of 4.8?
A: Given,pH = 5.0pka = 4.8
Q: Sijande par av aminer (esketamin ocl som är mest basisk. Förutsätt att ett protiskt lösningsmedel a:…
A: Basicity of amines is based on +I, Inductive effect and solvation effect in case of protic solvents.…
Q: Why is the conjugate acid of morpholine more acidic than the conjugate acid of piperidine?
A: According to Bronsted-Lowry acid base theory, a conjugate acid of a base is obtained by gaining a…
Q: Although it was initially sold as a rat poison, warfarin is an effective anticoagulant used to…
A:
Q: (a) Explain why phentermine [PhCH2C(CH3)2NH2] can’t be made by a reductive amination reaction.(b)…
A: (a)The structure of phentermine is given below.
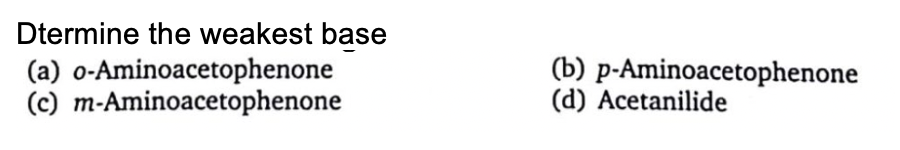
Step by step
Solved in 2 steps with 1 images

- Explain the observed difference in the pKa values of the conjugate acids of amines A and B.Given that the pKa of carbonic acid (H2CO3) is 6.4, is sodium bicarbonate strong enough of a base to deprotonate benzoic acid (i.e., push the acid-base equilibrium to the right)? What information led you to the answer to this question? Provide a short explanationThe following compound is derived from one of the twenty mammaliam "amino acids". It is the ethyl ester of arginine . If you offer a proton (H+) to this molecule, what is the expected conjugate acid that is formed. Draw its structure, thank you!
- Carnosine, found in muscle and brain tissue, acts as a buffer to neutralize small amounts of acid. The pKa of the conjugate acid of carnosine is close to 7.0. What is its structure?What is the ratio of ionized:unionized amine of glycine (pKa 9.60) in a solution at pH 8.0?Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions. b. Explain why it can cross-link DNA.
- Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions.b. Explain why it can cross-link DNA.Hypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?Although it was initially sold as a rat poison, warfarin is an effectiveanticoagulant used to prevent blood clots. Label the most acidic protonin warfarin, and explain why its pKa is comparable to the pKa of acarboxylic acid.
- When a amide is treated with SOCl2 ? What arises ?Although tryptophan contains a heterocyclic amine, it is considered a neutral aminoacid. Explain why the indole nitrogen of tryptophan is more weakly basic than one ofthe imidazole nitrogens of histidine.Starting with benzene synthesize the amino acid phenylalanine 1. using the strecker approach 2. by reductive amination

