Q: How many grams of silver can be plated onto an object in 16.28 minutes at 7.54 A of current? Agt(aq)…
A:
Q: 19) What species if added will allow the separation of neutral molecules in capillary…
A: Capillary electrophoresis (CE) is a separation method which separates charged molecules on the basis…
Q: Calculate the pressure in pascals exerted on a tabletop by a cube of iron that is 2.20 centimeters…
A: Given mass of iron cube = 83.8 g Length of each side of iron cube = 2.20 cm The pressure (P)…
Q: <Electrochemistry Exercise 20.79 - Enhanced with Feedback MISSED THIS? Watch KCV: Cell Potential and…
A:
Q: Explain why the conductivity of mixture A is higher than that of mixure B, , yet the pH of A is…
A: We have to analyse the given conductivity data and pH data.
Q: Saccharin, an artificial sweetener, is pictured below. Please note that this is a mostly structural…
A:
Q: If these products were dissolved in methylene chloride and ran on a TLC plate in 20% ethyl acetate/…
A: Reduction of ketone to a secondary alcohol is done using NaBH4.
Q: Consider Auger electron spectroscopy of gold atoms in which a highly monochromatic X-Ray source is…
A: Auger electron spectroscopy is a technique that involves bombarding a sample with high-energy…
Q: 18). What is the major product of the following reaction? (A) NH2 -NHz (D) Br₂, NaOH, H₂O -Br Bro…
A: Convertion of amides into amines are called as Hoffmann rearrangement reaction.
Q: A voltaic cell is constructed from a standard Fe²+ Fe half cell (E° red = -0.440V) and a standard…
A: Answer: Voltaic cell is a type of cell that converts chemical energy into electrical energy via…
Q: Identify the reactant, reagents, and intermediates in the synthetic sequence shown. Reagents may be…
A: A question based on reactions of aldehyde and ketone. A reaction scheme of ketone into alcohol…
Q: Sodium hydrogen carbonate (NaHCO3), also known as sodium bicarbonate or "baking soda", can be used…
A:
Q: Classify each of these chemical compounds: compound (CH₂)₂0 HBr Mg(OH)₂ type of compound (check all…
A: We need to classify the compounds in molecular , ionic ,organic,inorganic and hydrocarbon.
Q: MISSED THIS? Watch KCV: Cell Potential, Free Energy, and the Equilibrium Constant Read Section 20.5.…
A:
Q: The isotope of plutonium 238Pu is used to make thermoelectric power sources for spacecraft. Suppose…
A: Initial amount = Q0 = 3.0 kg For first order reactions, general relation is: Qt = Q0e-kt…
Q: What is the net ionic equation for 2NaOH+H2SO4>Na2SO4+2H2O
A: Steps to write a net ionic equation and complete ionic equation for a molecular reaction :…
Q: Consider the Lewis dot structure below (which means it does not have to indicate the true…
A: VSEPR (Valence Shell Electron Pair Repulsion) theory is used to determine the shape of the…
Q: Calculate the change in joules for vaporization of 4.00 g of water at 100 degrees Celsius.
A: Answer: During the vaporization process, a substance from its liquid form gets converted into its…
Q: Which symbols show that the contents of a product are flammable and corrosive? A. W y mobom exs www…
A: we have to select the sign that denotes the product contents are flammable and corrosive
Q: Identify each of the following statements as True or False concerning the following reaction. LDA…
A: A question based on reactions of ketone. Alkylation of ketone in the presence of LDA is given that…
Q: Use molecular orbital theory to predict which of the species below is paramagnetic. A) C₂²- B) B₂²-…
A: Since, If in a molecular orbital, if electron is not paired then species known as paramagnetic and…
Q: Draw the Lewis structure for chloroethane (C₂H₂CI). Be certain you include any lone pairs.
A: Given- Molecule-C2H5Cl We need to draw the Lewis structure of C2H5Cl.
Q: D D Question 4 Match HNO3 HF CH3CH2CH2COOH KOH NH3 Question 5 Match ✓ [Choose] oxyacid h neutral…
A: 1) HNO3 is oxyacid (nitric acid)Any oxygen containing acid is oxyacid. 2) HF is binary acid. (Binary…
Q: Questions 1. In the reaction Cl₂ + 21-→ 2Cl + I, which species (Cl, or I-) is the oxidizing agent?…
A: We have the redox reaction Cl2 + 2I-→ 2Cl- + I2 We have to discuss the oxidizing agent.
Q: 13. Choose the product of the following reaction A В ОН 0 0 H ОН НО. HO PCC dioxane C Н D О Н
A:
Q: What volume (in liters, at 568 K and 3.39 atm) of chlorine gas is required to react with 4.74 g of…
A: To solve this problem, we first need to use stoichiometry to determine how much chlorine gas is…
Q: For the following reaction scheme, match the correct reagent to each reaction (A, B, C, D and E).
A: Here a sequence of reaction is given and reaction contains side chain oxidation, halogenation,…
Q: 4. The product of the following reaction is: o. (1 mcl) COCH₂CH₂CH3 COH IV O + CH₂CH₂CH₂OH (1 mol)…
A:
Q: Use the bond energies listed below to estimate the enthalpy of formation of ammonia, NH3(g) NEN (946…
A: we have to calculate the enthalpy of formation of ammonia
Q: What is the structure for product (A) in the reaction sequence shown? OH PBr3 Mg D₂O ether O…
A:
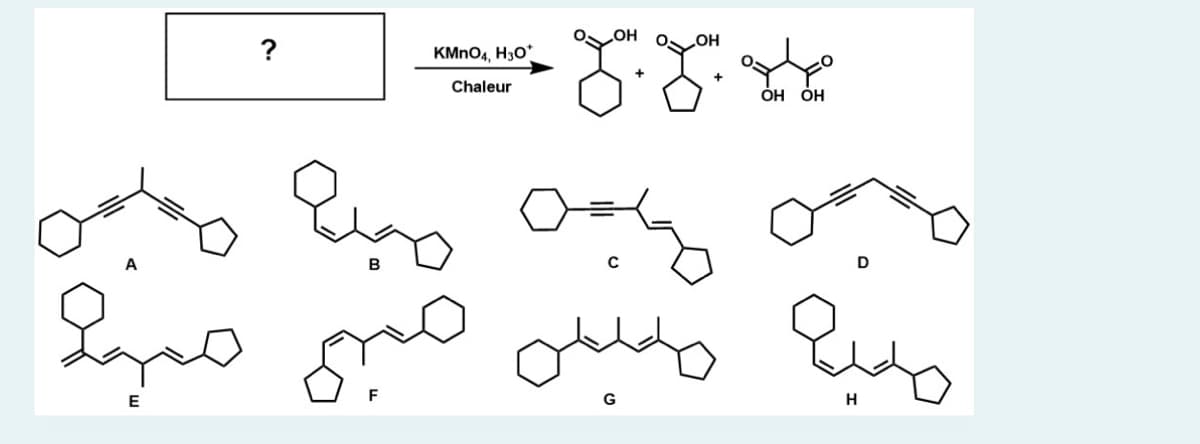
Q: Predict the organic products, in any order, when the alkene shown is subjected to ozonolysis with…
A: Ozonolysis is a simple reaction used for cleaving of double bond of alkene using O3 and metal ( as…
Q: 20.00 mL of a 0.13 M solution of weak acid (Ka = 4.00 × 10−4) was titrated with 0.12 M sodium…
A:
Q: Consider the following equilibrium: 2NO(g) + Cl₂ (g) → 2NOC1 (g) AG=-41. KJ Now suppose a reaction…
A: The given reaction is : 2NO(g) + Cl2(g) ⇌ 2NOCl(g) ΔG°=−41.kJ The pressure of Cl2 = 0.575…
Q: Name the following organic compounds: CH3 | CH | CH3 - compound CH- | CH₂ - CH₂ - CH3 I CH₂ - CH₂ -…
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Consider the reaction between aqueous solutions of baking soda, NaHCO3, and vinegar, HC₂H₂O2.…
A: Answer: Work done during a chemical reaction is calculated by the formula shown below: W=-∆ngRT…
Q: Determine the mass of precipitate, in grams, that forms when 76.4 mL of 0.0554 M Ba(ClO4)2 reacts…
A: Molarity is given as: Molarity=Number of moles of soluteVolume of solution (mL)×1000
Q: Toxic Cr(VI) can be precipitated from an aqueous solution by bubbling SO₂ through the solution. How…
A: The question is based on quantitative analysis. we need to calculate quantity of Sulphur dioxide…
Q: 20.0 mL of 0.250 M KOH is titrated with 0.100 M of HNO3. What volume of HNO3 (in mL) is needed to…
A: The Molarity of KOH solution M1= 0.250 M The volume of KOH solution V1= 20.0 mL The Molarity of HNO3…
Q: Consider the Lewis dot structure below (which means it does not have to indicate the true…
A: VSEPR (Valence Shell Electron Pair Repulsion) theory is used to determine the shape of the…
Q: What is the major organic product obtained from the following reaction? HBr peroxides 2
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide. In the…
Q: [Review Topics) References Use the References to access important values if needed for this…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: 18) What can be added in a capillary electrophoresis separation that will result in charge reversal…
A: To identify the reagent used to reverse the direction of osmotic flow in capillary electrophoresis.…
Q: The azide ion, N3, is a symmetrical ion, all of whose contributing structures have formal charges.…
A: We need to draw three Lewis structure of azide ion and to show formal charge .
Q: CN B12 FeBr3
A:
Q: In HPLC, the is always a liquid. a) mobile phase b) stationary phase c) solute
A: HPLC (high performance liquid chromatography) is an analytical technique to separate mixture of…
Q: Convert the following to the unit specified. Show your dimensional analysis (SHOW YOUR WORK!)…
A: In this type of questions we need to identify the relationship between the given two units and then…
Q: 12. Draw the product of the following Diels-Alder reaction. Indicate stereochemistry where…
A: A question based on reactions of alkene. Two reactant are given whose product is to be answered.
Q: An analytical chemist is titrating 50.2 mL of a 0.5100M solution of trimethylamine ((CH3)N) with a…
A: The question is based on the titrations. we are titrating a weak base with a strong acid. we need…
Q: Using the data in the table, determine the rate constant of the reaction and select the appropriate…
A:
Q: Are CH3+ and CH3 ̄ symmetrical or unsymmetrical molecules? Explain your reasoning.
A: We have to explain if CH3+ and CH3 ̄ are symmetrical or unsymmetrical molecules
Step by step
Solved in 3 steps with 1 images

- In the following equation: HNO2+ NaHCO3 (yields) NaNO2+H2O+CO2 Why does this result in water and CO2 instead of H2CO3?Explain A One-Step Mechanism (bond breaking and bond making occur at the same time) ?Chemistry of Fire Extinguishing Agents: Review and answer the below scenerios. a. The OSHA regulation at 29 C.F.R. §1910.162(b)(5) requires employers to provide a predischarge-employee alarm system to alert employees if a carbon dioxide concen-tration of 4% or greater is released from a fixed extinguishing system in a workplace. What is the most likely reason OSHA enacted this regulation? b. At 14 C.F.R. §121.265, FAA allows carbon dioxide to be used as a fire extinguish-ing agent onboard aircraft, provided that provisions are made to prevent harmful concentrations from entering any personnel compartment. Use Table 5.6 to deter-mine whether adverse health effects are likely to be experienced by a crew member who is confined to a compartment in which the air contains 8, carbon dioxide. c. The chemical formula of methyl bromide is CH3Br. Why does methyl bromide per-form effectively as a fire extinguishing agent? d. A potential risk of inhalation toxicity may occur to individuals that…
- Questiom:- The energy required to destabilize existing chemical bonds is called ____ energy. a. activation b. kinetic c. free d. potentialPeroxynitric acid (HOONO2) is an unstable molecule that decomposes to nitric acid and oxygen: 2HOONO2(aq) → 2HNO3(aq) + O2(g)When the concentration of peroxynitic acid is graphed against time, the resulting plot is curved, but if the logarithm of this concentration is plotted, we instead get a straight line. Based on this, which statement is true? a) This decay is a second order in peroxynitric acid. b) The slope of the straight-line graph is the rate constant. c) One needs the concentration of peroxynitric acid to calculate its half-life. d) The rate law appears to be of the form -Δ[HOONO2]/Δt = k[HOONO2].Define racemic mixture? And briefly explain the Racemic modification and its types?

