Q: Q2. A. Compound Which is composed of 57.14 % C, 6.16% H, 9.52% N, 27.18% O by mass and has a molar m...
A:
Q: 2. Consider the given reaction. 2BRF,(g) = Br,(g) + 5F,(g) If the respective equilibrium concentrati...
A:
Q: a rate of effusion th
A:
Q: Determine the formula and name for a compound that is 42.9% carbon and 57.1% Oxygen.
A:
Q: B. What mass of CaCL: is needed to react with sufficient silver nitrate to make 15 g of AgCl ? (M.wt...
A:
Q: d.A solution with a density of 0.876 g/mL contains 2.5 g of substance A with a molar mass of 92.14 g...
A:
Q: e.What is the molarity and normality of a solution which is prepared by dissolving 75.0 g of Ba(OH)₂...
A:
Q: a) Predict the structure of the major product(s) formed in each of the following reactions: (i) Br2 ...
A: Since you have posted multiple sun part questions, the answer for first subpart is given below. Kind...
Q: the concentrations of all substances present in the equilibrium mixture, if 2.35 mol of H2 and 2.35 ...
A: Given- Moles of H2 =2.35mol Moles of I2=2.35 mole Volume= 10 .0L Moles of HI= 3.76 mol
Q: Shown in the image is a diagram for the voltaic cell with the overall cell reaction being Al + Pb2+ ...
A: In the given cell Al electrode is anode because oxidation of Al is taking place while Pb electrode i...
Q: B. What mass of CACL2 is needed to react with sufficient silver nitrate to make 15 g of AgCl ? (M.wt...
A: We need to find the amount of CaCl2 required
Q: Problem 26: • Draw the mechanism for the meta-nitration of phenol. Indicate all the possible resonan...
A: Mechanism of meta-nitration of benzene is shown below:
Q: 1. Balance the equation: - C2H2 + O2 → CO2 + H2O 2. The chemical name for AAL2(SO4) 3. How many prot...
A: Since you have posted multiple questions, the answer for first three question is given below. Kindly...
Q: Directions: Complete the following blanks below with the appropriate data. Base your answers on the ...
A:
Q: Using the table for lonic Radii for Several Cations and Anions c. Cell edge length of ferrous oxide ...
A: Given, Ferrous oxide has rock salt structure . density of ferrous oxide =5.75gcm3
Q: diffusion
A:
Q: What should be taken in consideration when designing an enzyme kinetics experiment so that the react...
A: Correct option is C.
Q: 1.Substance X is mixed with substance Y. The state of resulting solution is liquid. Which of the fol...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: basic equilibrium equation
A:
Q: What is the pH of an aqueous solution that is initially prepared as 0.35 M NH4Br ? Note: Kb fo...
A: NH4Br is a salt formed from a strong acid HBr and a weak acid NH3. Therefore the salt is acidic.The ...
Q: A sample of a compound contains 30.46% N and 69.54% O by mass, as determined by a mass spectrometer....
A:
Q: what is the polymer formed when resorcinol and formaldehyde are combined with naoh? Shows complete ...
A: Please find the below attachment.
Q: 3. 24Na is produced through the reaction 26Mg(d,a)4Na. A 0.2 mm thick magnesium foil is irradiated f...
A:
Q: Sally has constructed a concentration cell to measure Ksp for MCln. She constructs the cell by addin...
A: Sally has constructed a concentration cell to measure Ksp for MCln. She constructs the cell by addin...
Q: 1. Balance the equation: - Cl: + KOH - KCI + KCIO, + HO 2. The chemical name for Ag:CrO. 3. How many...
A: Since you have posted multiple questions, the answer for first three question is given below. Kindly...
Q: Analysis of a sample showed that it contained 68.3% lead, 10.6% sulfur, and 21.1% oxygen. What is th...
A:
Q: Write the equilibrium constant expressions (K, and K) for the following reactions 4NH,(g) + 5O,(K) =...
A: Eqilibrium constant can be defined as the ratio of concentration of products to concentration of rea...
Q: What is the density (in g/mL) of an object if 7.237 mL has a mass of 5.443 grams?
A: Given data : Mass (m) = 5.443 g Volume (v) = 7.237 mL
Q: Identify the most polar part of the molecule
A:
Q: Write Half Redox reaction Sn = Sn02
A: In a balanced redox reaction two conditions are satisfied i.e atom balance and charge balance.
Q: 6. -čOCH,CH, 7. CH;CH:CH:CH;CH2NH2 S. CH:(CH:);CHO 9. HC CHẠCH,CH;-CH-CH, 10.
A: IUPAC Rules : Identify the longest carbon chain as parent chain Start the numbering from the end ne...
Q: Q3. A sample consisting of mono atomic ideal gas at I atm and 25C' expand adiabatically and reversib...
A: First law of thermodynamics says that internal energy change is equal to the sum of heat energy chan...
Q: What is the [H3O+] of a 0.1982 M CH3(CH2)2CH2NH3Br? Answers are expressed in molarity. Kb for CH3(CH...
A: Given : CH3(CH2)2CH2NH3Br is salt of strong acid and weak base. [CH3(CH2)2CH2NH3Br] = 0.1982 M Kb...
Q: HO H3CO. X Y IR MS 'H NMR 13C NMR
A:
Q: plalh the difference between the split and splitless mode of operation in a GO jector. Consider prop...
A:
Q: Rank the solubility of the 3 bases: Na2CO3, K2cO3, NaOH
A: Since salts dissolve into electrically charged species (as do acids and bases) they fall in the gene...
Q: Organic compounds are usually liquids and low melting solids because they have relatively intermolec...
A: There are various types of intermolecular forces present in compound such as hydrogen bonding, Londo...
Q: B. What mass of CACL: is needed to react with sufficient silver nitrate to make 15 g of AgCl ? (M.wt...
A:
Q: Determine the pH and percent ionization of a .80 M HNO2 solution. Given Ka for HNO2 = 4.5*10^-4
A: pH is determined by the concentration of H+ ions that will be formed after ionization of Nitrous aci...
Q: The group 17 elements cannot be found free in nature because they are reactive. they are in gaseous ...
A: 1. Option A 2. Option A 3. Option A
Q: Hydrides are compounds that contain Nitrogen Hydrogen Oxygen Halogens Water (H2S) has enormously hig...
A: Answer Q=1 hydrides are compound that contain Hydrides are formed when hy...
Q: Q3. A sample consisting of mono atomic ideal gas at I atm and 25C" expand adiabatically and reversib...
A: Introduction: For an adiabatic process, the change in heat, ∆Q = 0 Therefore, from the 1st law of Th...
Q: a) Determine the grams of reagent in excess if 4.19 moles BiCl3 reacted with 0.970 moles water. b) W...
A:
Q: 3. Which of the following statement/s is/are correct? 1. 6.00 N H2C03 is equivalent to 3.00M H2CO3. ...
A:
Q: Which of the following molecules can be a starting material in producing 4- Methyl-1-pentyne? Br Br ...
A: We have to find out the molecule which can be a starting material in producing 4- methyl 1- pentyne?...
Q: One ingredient in some antacids is Al(OH)3 which reacts with stomach acid, HCl . Complete and bala...
A: Neutralization reaction: The hydrogen ion of acid reacts with the hydroxide ion of a base to form sa...
Q: A combustion reaction released heat at a constant pressure. Which of the following statements is COR...
A: According to the first law of thermodynamics, the enthalpy change of a system is related by the inte...
Q: What volume of 0.500M NaOH is required to react completely with 5.00 g Al(NO3)3
A:
Q: otentiometry Topic Briefly discuss the following: 1.different types of membrane indicator elec...
A: 1. Solution - According to the question - membrane electrodes that produce an electrical potential ...
Q: Which molecules are more likely in the course of a collision to reach the transition state and overc...
A: The solution of the question is given below:
Step by step
Solved in 2 steps with 2 images

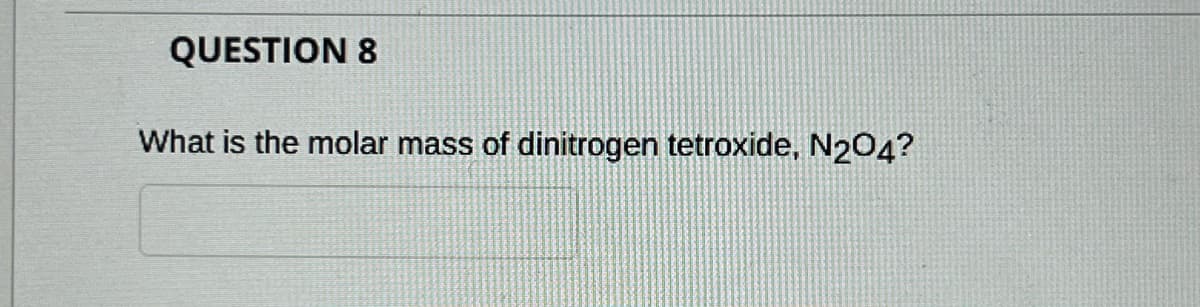
- I needing help how to solve Average mass % NaHCO3 in Alka-Seltzer, pleaseA compound NiX3 is 19.67% (by mass) nickel. What is the molar mass of the compound? What is the name of the compound?9 Calculate the mass of K10, (molar mass = 21402 @/mol) required for par A 1. Sc Prelaboraur, Assienment.question-2.
- What is the number of grams in 5.6x10^-9 molecules of diiodine dichloride?If a human is 80.0% water, by mass, how many molecules of water (H20) are there in a body weighing 46.57kg? Needed help on this one and its not in the text book.1. Define Avogadro's number? explain w/ applications 2. What is a mole? 3. How are Avogadro's number and mole related, for element and/or compound?

