Q: Describe the preparation of the following reagents. 3. 375 mL of 40 ppt Cl" from 0.01 M P6CI2
A: Solution -
Q: e mole fraction of C2H
A:
Q: For each addition route of the reaction, draw the structure(s) of the major product(s), including st...
A: Answer of the question given below,
Q: 6. The following drawing shows the association constants of three different complexes in chloroform,...
A: Introduction : Hydrogen bond is an intermolecular force that form a dipole- dipole attraction when ...
Q: omplete the table below for calculating the molar mass of the compound sulfur tetrafluoride. Molar m...
A: Given , Sulfur tetrafluoride
Q: Some measurements of the initial rate of a certain reaction are given in the table below. [H.] 12 in...
A: Rate constant is proportionality constant of rate law expression, rate law is experimental analysis ...
Q: An ionic compound of formula XY2 (X = cation with two positive charges, Y = anion with one negative ...
A:
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A:
Q: volume
A:
Q: Why do enzymes get denatured by organic reagents?
A: Enzymes are proteins (except Ribozyme) function in catalysis of biochemical reactions.Denaturation i...
Q: A student heats a 5.0 g sample of an unknown metal to a temperature of 207∘C, and then drops the sam...
A:
Q: ST4B.1- Estimate the CHANGE in the boiling point [in Kelvin units or Celsius degrees] of a liquid if...
A: To estimate change in boiling point for the given set of data: P1 = 1.00 atm P2 = 1.00 atm + 5% incr...
Q: onstant is given for two a B(g) Kc = ?
A:
Q: Nitrogen dioxide dimerizes according to the following reaction: NO2 (g) = N204 (g) Kp = 6.7 at 298 K...
A:
Q: Balance the given molecular equation using redox (electron-transfer method). Identify the element ox...
A:
Q: Choose three different C-C disconnections for this ketoester and write out the structures of the sta...
A: Retrosynthesis is the process of "deconstructing" a target molecule into starting material.
Q: 3. Suggest a reaction sequence for preparing the following compound from the given starting material...
A:
Q: Decomposition reactions like carbonate decomposition is a common organic chemistry reaction. True Fa...
A: Orgaanic compounds can be defined as the compounds containing carbon bonded with hydrogen.These are ...
Q: NiCl2 + 6 NH3 + 2 KClO4 ® Ni(NH3)6(ClO4)2 + 2 KCl if you mix 1.0406 g of NiCl2, 6.0092 g NH3 and 2.0...
A: The given chemical equation is: NiCl2 + 6 NH3 + 2 KClO4 → Ni(NH3)6(ClO4)2 + 2 KCl Given mass of NiCl...
Q: Draw the kinetic and the thermodynamic addition products formed when one equivalent of HBr reacts wi...
A: The thermodynamic and kinetic product is given below
Q: the equilibrium constant for t
A: Equilibrium constant is defined as a constant which explains the relationship between the concentrat...
Q: Chapter 7: Reactions in Aqueous Solutions When predicting a product for a reaction between two ionic...
A: net Ionic equation is obtained by removing spectator ions from the solutions, because it not form f...
Q: .coH coM What kind of reaction is occurring?
A: Radical reactions that involes the symmetrical bond clevages and bond formation reaction. In this re...
Q: 4. Are these two compounds: a) the same b) enantiomers OR c) diastereomers? HÖ +-OH Explain how you ...
A:
Q: Consider the following reaction: SO2 Cl2 (g) = SO2 (g) + Cl2 (g) K. = 2.99 x 10-7 at 227 °C You may ...
A: Given reaction: SO2Cl2 (g) ⇌ SO2 (g) + Cl2 (g) Given, Kc = 2.99 × 10-7 Given, the initial concentrat...
Q: How does the following affect the basicity of compounds? Explain clearly and thoroughly. a. Bond le...
A: Basicity is the number of protons given by an acid when it is dissolved in water. Acidic strength or...
Q: A new temperature scale, know as the Ungoogable (U) scale is related to Fahrenheit (F) in this way: ...
A:
Q: complete ionic equation for A. 3CaCl2(aq)+2Na3PO4
A:
Q: Several brands of antacids use aluminum hydroxide to react with stomach acid (which is primarily hyd...
A:
Q: Part B Calculate the mass of water produced when 9.54 g of butane reacts with excess oxygen. Express...
A: Given, Mass of butane (C4H10) reacts = 9.54 g Excess reactant - O2 (Oxygen gas) Limiting reactant -...
Q: how would you prepare 25mL of a 15% dye solution beginning with a 20% stock dye solution? find mL of...
A:
Q: you have 10mL of a 60% stock dye solution. what is the maximum amount of a 12% dye solution you coul...
A: Initial concentration of solution, C1 = 60% Initial volume of 60% solution, V1 = 10 mL Final concent...
Q: For the reaction X(g) + 2Y(g) = 3Z(g) Kp = 2.08x10-2 at a temperature of 325 °C . Calculate the valu...
A: Given reaction: X (g) + 2Y (g) ⇌ 3Z (g) Given, Kp = 2.08×10-2 at 325 ∘C We have to find the value of...
Q: Lewis Structure
A:
Q: Specify the formal charges (both sign and magnitude) on the atoms labeled a-c. H c/ H2C=CH-C: a b 1)...
A: Formal charge is an electric charge over the atom in molecules.
Q: how many dozen bagels do you have (assume that 1 bagel weighs 4oz
A: I have 20 lbs of bagles. 1 bagles weighs 4 oz. This oz is actually ounce which is nearly 1/16 pounds...
Q: A compound with a conjugate base that is more stable because of electron delocalization or resonance...
A: No, it is a false statement. right statement is- A compound with a conjugate base that is more stabl...
Q: Calculate the pH of a 0.100 M solution of lactic acid, for which the Ka value is 1.40 x 10-4.
A:
Q: Q4: Identify the structural effect present on the given molecule.
A: The structural effect present in compound depend on the functional group present in present
Q: Rank the isomers in order of increasing heat of formation. Highest heat of formation Lowest heat of ...
A: The order of heat of formation of diene is given below
Q: Example 3 A sample of zinc anode corrodes uniformly with a current density of 4.27 x 107 A/cm² aqueo...
A: Current density = 4.27 × 10-7 A/cm2
Q: Which statement is correct? I. The carbons in a chair conformation will have two axial or two equato...
A:
Q: "СООН v Pi delocalization A. Present CH hyperconjugation B. Absent Lone-pair delocalization Inductiv...
A:
Q: Rank the isomers in order of increasing heat of formation. Highest heat of formation Lowest heat of ...
A: The alkenes are compound which composed of carbon and hydrogen and contains at least one double bond...
Q: - the Diels-Alder reaction.
A:
Q: The structures Pentan-2-one and Pentan-3-one are isomers. A Positional B Stereoisomers Functional Sk...
A: Answer :
Q: Which atom is used as a reference in classifying the strength of electron-attracting and -withdrawin...
A: Electron attracting and withdrawing effect involve inductive effect, mesomeric effect. These effect...
Q: How many moles are in 33.36 grams of Ec,H5O2?
A: Given :- mass of Ec6H5O2 = 33.36 g Molar mass of Ec = 31.79 g/mol To calculate :- num...
Q: 2) Discuss the molecular basis for the reaction of tollen's reagent with acetic acid that leads to t...
A: carboxylic acids have -COOH functional group.
Q: Explain the reasoning for the baking soda and vingear balloon experiment. Fill in the blank. If baki...
A: When baking soda and vinegar are mixed together then the formation of carbon dioxide gas take place ...
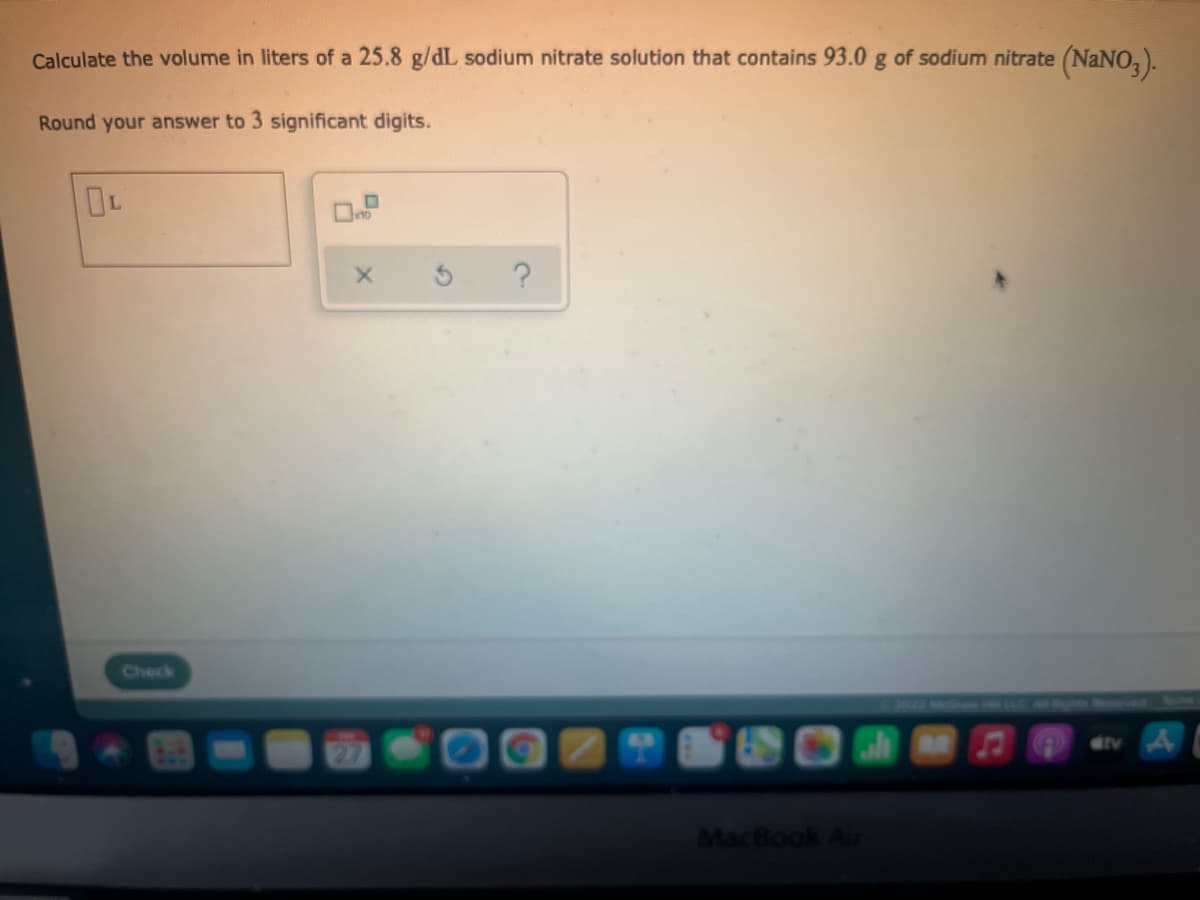
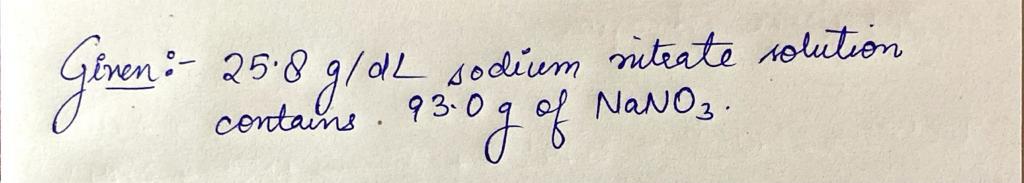
Step by step
Solved in 2 steps with 2 images

- Calculate the volume in liters of a /6.17gdL aluminum chloride solution that contains 81.0g of aluminum chloride AlCl3 . Round your answer to 3 significant digits.You have a stock NaCl solution of 8 g/L. You want to prepare 357 mL of a 5 mg/L NaCl solution. How many mL of the stock solution will you need? Give your answer with two digits after the decimalA chemist prepares a solution of sodium chloride NaCl by measuring out 6.51mol of sodium chloride into a 150.mL volumetric flask and filling the flask to the mark with water. Calculate the concentration of the chemist's sodium chloride solution. Round your answer to 3 significant digits.
- A chemist prepares a solution of sodium carbonate Na2CO3 by weighing out 147.g of sodium carbonate into a 500.mL volumetric flask and filling the flask to the mark with water. Calculate the concentration in g/mL of the chemist's sodium carbonate solution. Round your answer to 3 significant digits.You have a stock NaCI of 5 g/L. You want to prepare 259 mL of a 1 mg/L NaCI solution. How many mL of the stock solution will you need? Give your answer with two digits after the decimal.Calculate the volume in milliliters of a 3.6M sodium nitrate solution that contains 500.mmol of sodium nitrate NaNO3. Round your answer to 2 significant digits.
- You have a 16.0 M stock solution of Superstressin (something that little nervous dogs are known to drink every morning). You need to prepare 500. mL of 0.358 M solution of Superstressin. What volume of your stock solution do you need to use for this? In the box below enter the volume in milliliters (numeric value only) with a sufficient number of significant digits.Calculate the volume in liters of a ×2.2910−6/μmolL mercury(I) chloride solution that contains 800.mg of mercury(I) chloride Hg2Cl2 . Round your answer to 3 significant digits.A chemist must dilute 82.6mL of 51.0 mM aqueous potassium permanganate KMnO4 solution until the concentration falls to 24.0mM. He'll do this by adding distilled water to the solution until it reaches a certain final volume. Calculate this final volume, in liters. Be sure your answer has the correct number of significant digits.
- A chemist prepares a solution of sodium nitrate by measuring out 2.1 *10^2 of sodium nitrate into 450 ml a volumetric flask and filling the flask to the mark with water.Calculate the concentration in the chemist's sodium nitrate solution. Round your answer to significant digits.Hydrocortisone is used as an anti-inflammatory for localized pain. You need to prepare a 45 mg/mL solution of hydrocortisone for an injection via a dilution. You have 15 mL of a 175 mg/mL stock solution of hydrocortisone available. How many mL of the 45 mg/mL solution can you prepare? Note: Provide your answer using the correct number of significant figures and remember to include your units.Calculate the volume in mL of 7.0 M HNO3 needed to make 675 mL of 1.5 M HNO3. Report your answer with the correct number of significant figures. Enter only the number. There will be a penalty if you enter the units.

