Effect of Common lons on the Solubility of Ca(OH)2 Answer the concentrations and solubility in 3 significant figure. You can use scientific notation using "e". Example: 0.0010 = 1.0e-3 [Ca²*hotal = [Ca2"lrom Ca(OH)2 + [Ca2*Irom Caci2 (This will be clarified in the Postlab Module) n Caci20 Assume that 0.01M Caci2 medium is exact. Trial 1 Volume of filtrate (mL) 50 50 50 Net volume of HCI (mL) 9.1 9.0 9.1 [OH-] [Ca 2+kotal Solubility of Ca(OH)2 Average solubility
Effect of Common lons on the Solubility of Ca(OH)2 Answer the concentrations and solubility in 3 significant figure. You can use scientific notation using "e". Example: 0.0010 = 1.0e-3 [Ca²*hotal = [Ca2"lrom Ca(OH)2 + [Ca2*Irom Caci2 (This will be clarified in the Postlab Module) n Caci20 Assume that 0.01M Caci2 medium is exact. Trial 1 Volume of filtrate (mL) 50 50 50 Net volume of HCI (mL) 9.1 9.0 9.1 [OH-] [Ca 2+kotal Solubility of Ca(OH)2 Average solubility
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter15: Equilibria Of Other Reaction Classes
Section: Chapter Questions
Problem 11E: The Handbook of Chemistry and Physics (http://openstaxcollege.org/l/16Handbook) gives solubilities...
Related questions
Question
pls complete the table using given data
Effect of Common Ions on the Solubility of Ca(OH)2
1. Add Ca(OH)2 to 250.0 mL of 0.010 M CaCl2 with stirring until equilibrium is achieved.
2. Filter the undissolved precipitate. Measure out 50.0 mL of the filtrate in a 250-mL Erlenmeyer flask.
3. Add a few drops of phenolphthalein indicator and titrate with standardized HCl solution until endpoint
is achieved.
4. Record the volume of HCl solution used. Perform two more trials.
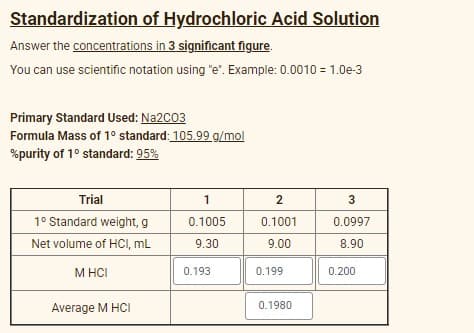
Transcribed Image Text:Standardization of Hydrochloric Acid Solution
Answer the concentrations in 3 significant figure.
You can use scientific notation using "e". Example: 0.0010 = 1.0e-3
Primary Standard Used: Na2C03
Formula Mass of 1° standard: 105.99 g/mol
%purity of 1° standard: 95%
Trial
1
2
3
1° Standard weight, g
0.1005
0.1001
0.0997
Net volume of HCI, mL
9.30
9.00
8.90
M HCI
0.193
0.199
0.200
Average M HCI
0.1980
![Effect of Common lons on the Solubility of Ca(OH)2
Answer the concentrations and solubility in 3 significant figure.
You can use scientific notation using "e". Example: 0.0010 = 1.0e-3
[Ca²*hotai = [Ca2*lrom Ce(OH)2 * [Ca?*lfom CeCi2 (This will be clarified in the Postlab Module)
Assume that 0.01M CaC12 medium is exact.
Trial
2
Volume of filtrate (mL)
50
50
50
Net volume of HCI (mL)
9.1
9.0
9.1
[OH-]
[Ca 2+kotal
Solubility of Ca(OH)2
Average solubility](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F55815ef3-1f3f-422e-b33b-6261d3029e90%2Fad32ffe6-44fc-4d61-8d2f-18c722998d8a%2Fetn526_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Effect of Common lons on the Solubility of Ca(OH)2
Answer the concentrations and solubility in 3 significant figure.
You can use scientific notation using "e". Example: 0.0010 = 1.0e-3
[Ca²*hotai = [Ca2*lrom Ce(OH)2 * [Ca?*lfom CeCi2 (This will be clarified in the Postlab Module)
Assume that 0.01M CaC12 medium is exact.
Trial
2
Volume of filtrate (mL)
50
50
50
Net volume of HCI (mL)
9.1
9.0
9.1
[OH-]
[Ca 2+kotal
Solubility of Ca(OH)2
Average solubility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning