Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 7P
Related questions
Question
Transcribed Image Text:(58) A Ch - A fun
A Ch 4 A Lab
O Ix G Whi
G Whi
G An a G Mas G is Br
G Pari
Ch 2
Мар
G acc
G pen
G iron
+
Thir
Goo
O File
/home/chronos/u-89894221a1237b56532567cf04377caaald4971d/MyFiles/Downloads/Observation%20protocol.pdf
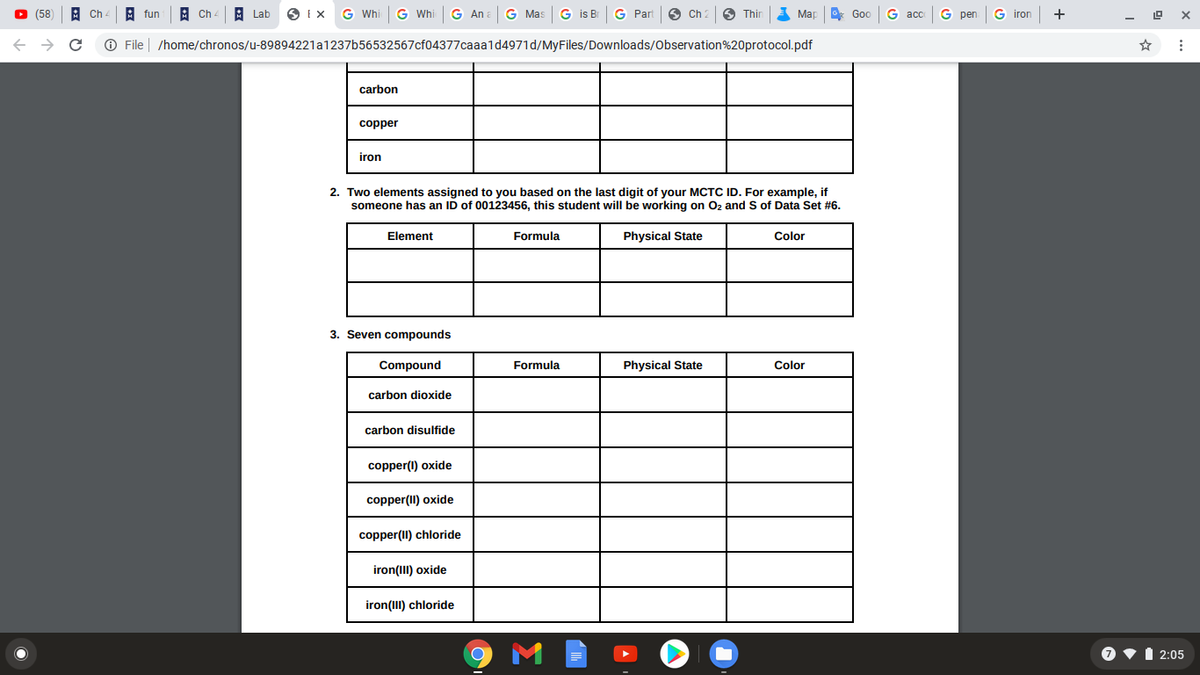
carbon
copper
iron
2. Two elements assigned to you based on the last digit of your MCTC ID. For example, if
someone has an ID of 00123456, this student will be working on Oz and S of Data Set #6.
Element
Formula
Physical State
Color
3. Seven compounds
Compound
Formula
Physical State
Color
carbon dioxide
carbon disulfide
copper(1) oxide
copper(II) oxide
copper(II) chloride
iron(III) oxide
iron(III) chloride
2:05
Transcribed Image Text:(58) A Ch - A fun
A Ch
A Lab
O Ix G Whi
G Whi
G An a G Mas G is BI
G Part
Ch 2
O Thir
Map
G acc
G pen
G iron
+
Goo
O File
/home/chronos/u-89894221a1237b56532567cf04377caaald4971d/MyFiles/Downloads/Observation%20protocol.pdf
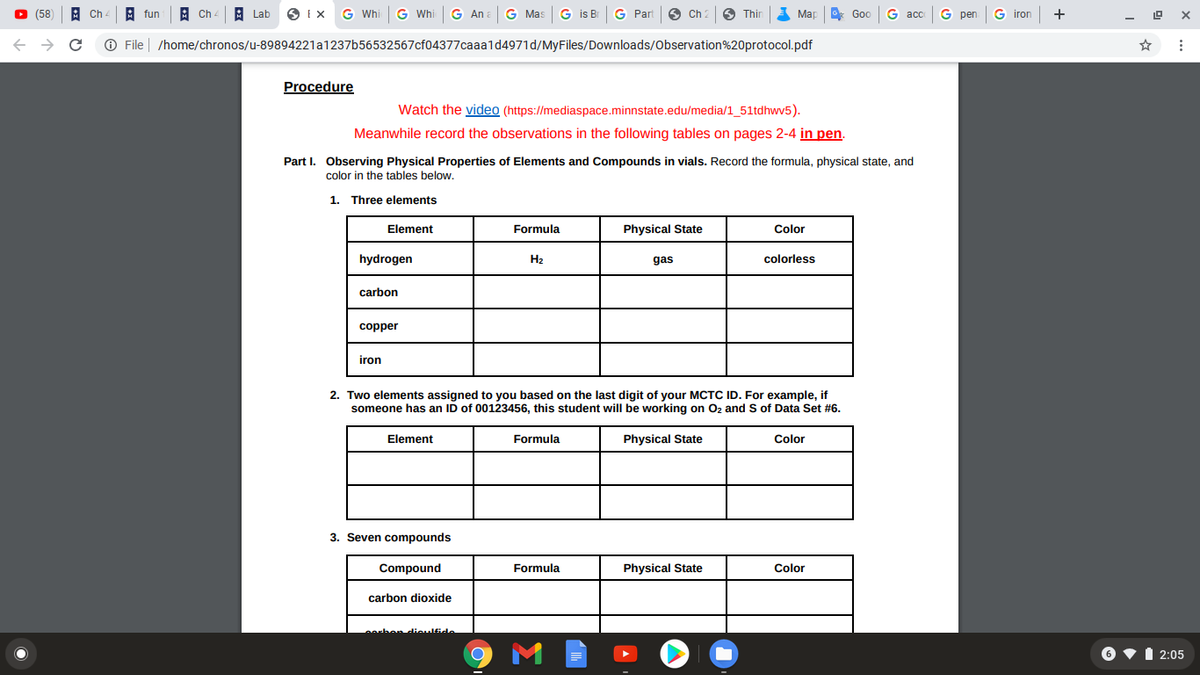
Procedure
Watch the video (https://mediaspace.minnstate.edu/media/1_51tdhwv5).
Meanwhile record the observations in the following tables on pages 2-4 in pen.
Part I. Observing Physical Properties of Elements and Compounds in vials. Record the formula, physical state, and
color in the tables below.
1. Three elements
Element
Formula
Physical State
Color
hydrogen
H2
gas
colorless
carbon
copper
iron
2. Two elements assigned to you based on the last digit of your MCTC ID. For example, if
someone has an ID of 00123456, this student will be working on Oz and S of Data Set #6.
Element
Formula
Physical State
Color
3. Seven compounds
Compound
Formula
Physical State
Color
carbon dioxide
hen dieulfide
2:05
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



