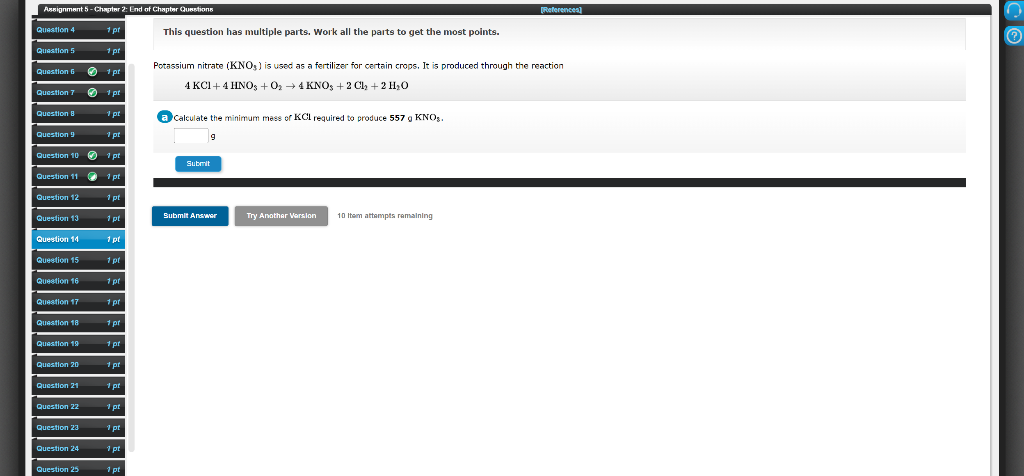
End of Chapter Quetions References) This question has multiple parts. Work all the parts to get the most points. Potassium nitrate (KNO; ) is used as a fertilizer for certain crops. It is produced through the reaction 4 KCI+ 4 HNO; +0, → 4 KNO, + 2 C, + 2 H,0 Cakulate the minimum mess of KCl required to produce 557 g KNO,. Submit Submit Answor Try Anothar arsion 10 Itam attempts ramaining
End of Chapter Quetions References) This question has multiple parts. Work all the parts to get the most points. Potassium nitrate (KNO; ) is used as a fertilizer for certain crops. It is produced through the reaction 4 KCI+ 4 HNO; +0, → 4 KNO, + 2 C, + 2 H,0 Cakulate the minimum mess of KCl required to produce 557 g KNO,. Submit Submit Answor Try Anothar arsion 10 Itam attempts ramaining
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 6ALQ: onsider separate aqueous solutions of HCI and H2S04 with the same concentrations in terms of...
Related questions
Question
9
Transcribed Image Text:Assignment 5-Chapter 2: End of Chapter Queetions
References)
Question 4
1 pt
This question has multiple parts. Work all the parts to get the most points.
Question 5
1 pt
Potassium nitrate (KNO:) is used as a fertilizer for certain crops. It is produced through the reaction
Question 6 O 1 pt
4 KCI + 4 HNO; + 0, → 4 KNO, + 2 Cl, + 2 H;0
Question 7 O pr
1 pt
Question 8
1 pt
Calculate the minimum mass of KCl required to produce 557 g KNOS.
1 pt
Question 9
Question 10 O 1 pt
Submit
Question 11 1 pt
Question 12
1 pt
Question 13
1 pt
Submit Answer
Try Anothar Verslon
10 Itam attampts ramaining
Question 14
1 pt
Question 15
1 pt
Question 16
1 pt
Question 17
1 pr
Question 18
1 pr
Question 19
1 pt
Question 20
1 pt
Question 21
1 pt
Question 22
1 pt
Question 23
1 pt
Question 24
1 pt
Question 25
1 pt
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning