Chapter13: Acids And Bases: The Molecules Responsible For Sour And Bitter
Section: Chapter Questions
Problem 20E: What causes acid indigestion? List some common antacid ingredients and their side effects.
Related questions
Question
Transcribed Image Text:00
LL
LEGO
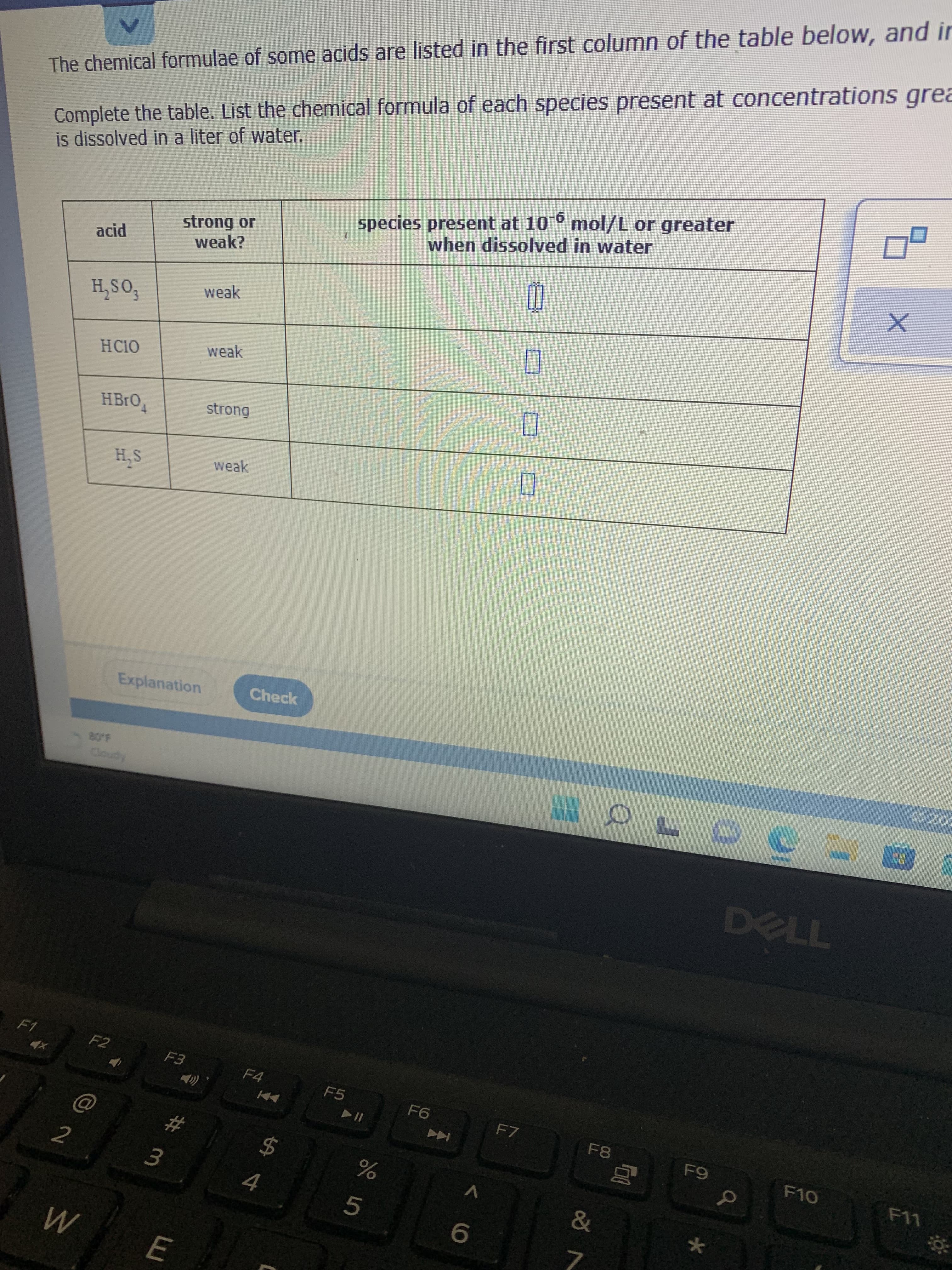
Complete the table. List the chemical formula of each species present at concentrations grea
is dissolved in a liter of water.
The chemical formulae of some acids are listed in the first column of the table below, and ir
9-
species present at 10° mol/L or greater
when dissolved in water
strong or
weak?
acid
weak
fos'H
HCI0
weak
strong
weak
S'H
Explanation
Check
Coudy
DELL
F1
F2
F4
F5
F6
F7
F10
2$
4.
5.
7.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

