environment that may cause problems on the system. musculoskeletal O Physical hazard O Biological hazard O None of the choices O Ergonomic hazard O Safety hazard
environment that may cause problems on the system. musculoskeletal O Physical hazard O Biological hazard O None of the choices O Ergonomic hazard O Safety hazard
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter31: Immunochemistry
Section: Chapter Questions
Problem 31.72P
Related questions
Question
Please send me the question in 20 minutes it's very urgent plz
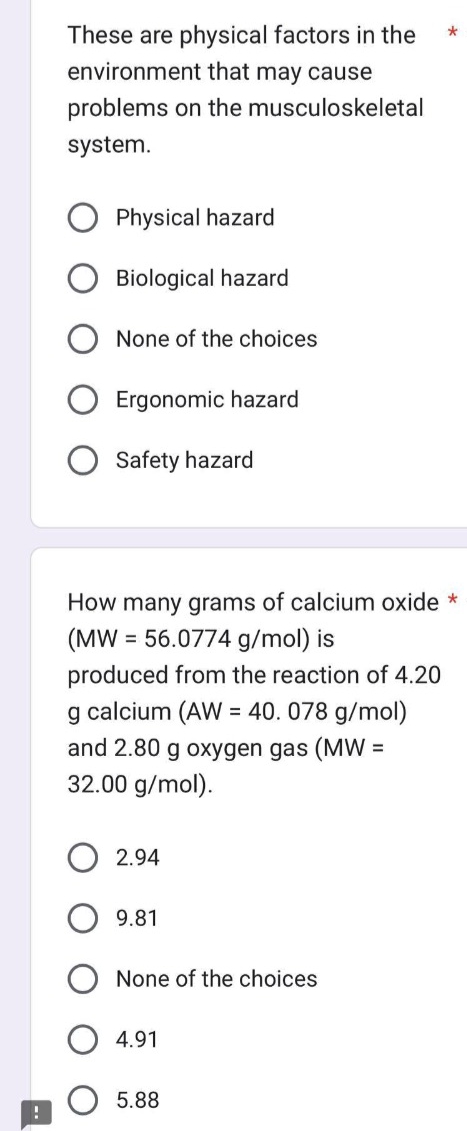
Transcribed Image Text:!
These are physical factors in the
environment that may cause
problems on the musculoskeletal
system.
Physical hazard
Biological hazard
None of the choices
Ergonomic hazard
Safety hazard
How many grams of calcium oxide *
(MW = 56.0774 g/mol) is
produced from the reaction of 4.20
g calcium (AW = 40.078 g/mol)
and 2.80 g oxygen gas (MW =
32.00 g/mol).
2.94
9.81
None of the choices
4.91
*
5.88
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning