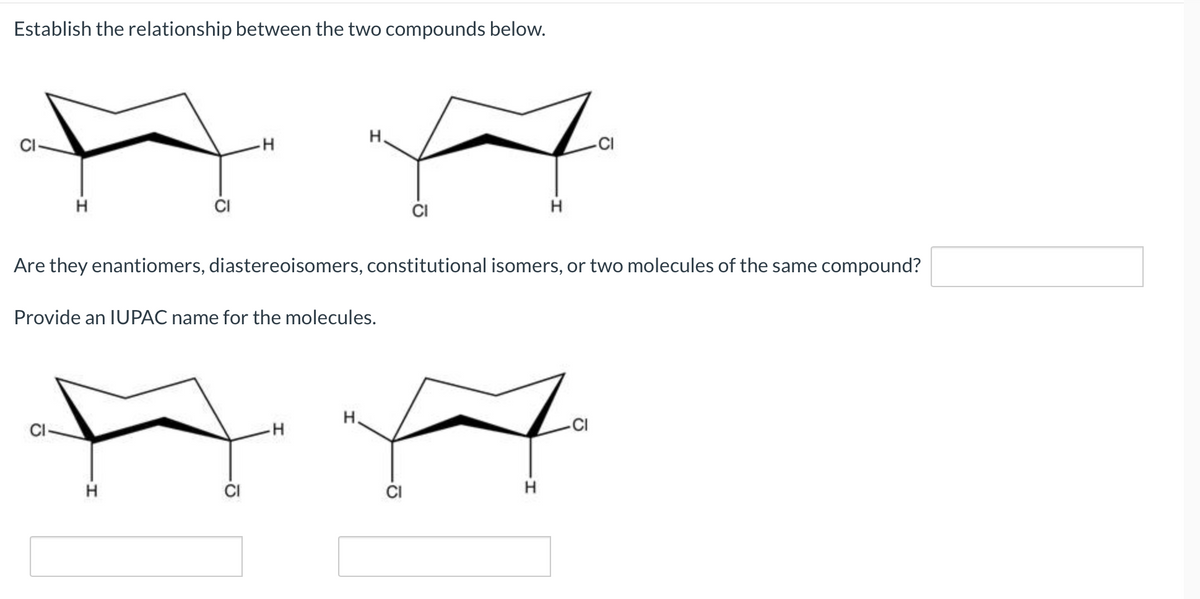
Establish the relationship between the two compounds below. H. AA H H Are they enantiomers, diastereoisomers, constitutional isomers, or two molecules of the same compound? Provide an IUPAC name for the molecules. H H. CI
Q: 4355.5 kJ/mol 16,091 kJ/mol
A: Ionization energy is the amount of energy required to remove an electron from the outermost shell of…
Q: . Calculate AHº, AS°, and AG° at 298K for the following reactions that produce acetic acid: CH4(g) +…
A: The objective of the question is to calculate the standard enthalpy change (ΔHº), standard entropy…
Q: Calculate the pH of a buffer solution made by mixing a 0.37 M formic acid (HCOOH) with 0.23 M sodium…
A: pH of buffer solution=3.54Explanation:So,pH of buffer solution=3.54
Q: What is the effect of mobile phase on the selectivity factor (☐ ) for benzonitrile and p-…
A: The objective of the question is to understand the effect of the mobile phase on the selectivity…
Q: -NHCH2CH3 + HBr
A: Given reaction:
Q: slove Part BFor the same procedure described in the chemical equilibrium lab handout for determining…
A: The objective of this question is to calculate the equilibrium constant, K, for the reaction I2(aq)…
Q: 47) What is the common name of this compound? n CH3-C-O-CH3 A) ethyl methyl ester B) diethyl ester…
A:
Q: Draw structural formulas for the a,ẞ-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: 1. What is the IUPAC name for the compound given below? H. C. HIN: Н CH3
A: The IUPAC rules for naming amides typically involve identifying the parent alkane, replacing the -e…
Q: Macmillan Lea In a coffee cup calorimeter, 16.0 mL of 0.47 M nitric acid (HNO,) and 16.0 mL of 0.47…
A: The objective of the question is to calculate the enthalpy of neutralization for the reaction.
Q: Draw the structure of the product for the following reaction and identify the type of reaction: 0 ||…
A: The reactions in which an atom or group is replaced by another atom or group are known as…
Q: Explain why K has more negative electron affinity than Ca. Explain why Ge has more negative electron…
A:
Q: Propose a reaction mechanism and fill in the product of the following transformation: 1. NaH MeO₂C-…
A:
Q: What [Ag+] (in M) is required to reduce the [CrO42] in a solution to 8.1× 10-4 M by precipitation of…
A: The objective of the question is to find the concentration of Ag+ ions required to reduce the…
Q: lonic equations for each of the reactions indicated belo rion loth before and be Face part and list…
A: Net Ionic Equation:The balanced net ionic equation includes only species that are involved in the…
Q: Which electron-dot diagram best represents a binary ionic compound? OA. H:S: OB. K+[:Br:] OC. 12- Ca…
A: The objective of the question is to identify the electron-dot diagram that best represents a binary…
Q: What is the pH of 0.015 M aqueous solution of barium hydroxide? 1.82 12.48 10.40 1.52 12.17
A: Given:Concentration of barium hydroxide, Ba(OH)2 = 0.015 MWe have to determine the pH of the…
Q: ease provide the retrosynthetic analysis of the compound below. Start with cyclohexanone. Write a…
A: The objective of the question is to predict the synthesis pathway for the given molecule.The…
Q: Starting with the following reaction a more iron(II) chloride? Choose all th FeS(s) + 2 HCl(g) =…
A: Le Chatelier principle says that equilibrium will shift in such a way so as to undo the effect of…
Q: Sketch the shape and orientation of the following types of orbitals. a. px b. dyz
A: There is need to find shape and orientation of Px and dyz orbitals .
Q: What is the product formed in the following reaction? O B) 2-methylcyclohexene OA) Cyclohexene OD)…
A:
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A: Diels-Alder reaction: It is a [4+2] cyclo addition reaction between a diene and dienophile to form a…
Q: How many atoms from the following molecule will be detected in a H NMR experiment? We are not asking…
A: We have to find the total number of atoms that will be detected in a 1H NMR experiment from the…
Q: Give the reagents and conditions to provide the following transformations, usethe 1. 2. etc.…
A: The required reagents to carry out the reaction are clearly mentioned. Please find the attachment…
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A: The Diels-Alder reaction is a type of [4+2] cycloaddition reaction, where four π-electrons of the…
Q: Give the major organic product of the following reaction:
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: If the frequency of the light is 5.1 x 1014 s-1, what is the wavelength for it in nanometers?
A: The objective of this question is to calculate the wavelength of light given its frequency. The…
Q: Calculate the concentration of the hydroxide ion in a solution that contains 5.5 × 10- 5 M of…
A: [H3O+] = 5.510-5 MKw = 1.010-14
Q: The electron in a hydrogen atom absorbs a photon causing the electron to jump from the state n = 2…
A:
Q: 4. During Part 1, which reactant (Fe3+ or SCN-) is in excess and which is limiting? What is the…
A: The question is asking about a chemical reaction involving iron (III) ions (Fe3+) and thiocyanate…
Q: Suppose a 250. mL flask is filled with 0.70 mol of H2 and 1.1 mol of I2. The following reaction…
A: Find the initial concentration of hydrogen and iodide.Molarity = moles /volume Molarity of H2=…
Q: Calculate the pH change that results when 15 mL of 2.0 M HCI is added to 580. mL of each of the…
A: The objective of the question is to calculate the change.
Q: raw the structures for the carbonyl compound and amine required to synthesize benzyl methylamine via…
A: The chemical reaction in which the condensation product of carbonyl compound and amine undergoes…
Q: a. Predict the 13C NMR (# of peaks, integration of each peak, and relative chemical shift of each…
A: The signals in the NMR based on the chemical environments of the atoms present in the compounds.If…
Q: In the reaction below, which species is the electrophile? CI B H₂O OH AC B D H3O+ cr ဗ
A: Electrophiles are electron-deficient species that seek electrons to complete their octet or…
Q: b Draw curved arrows to show the rearrangement of electrons in the second step leading to the…
A: The reaction involves elimination of water molecule to result in 1-butene.
Q: Provide a name for the following compound: 3-sec-Butyl-2,5-dimethylhexane O…
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Give the explanation of formula. Calculate the pH after 0.034 mole of NaOH is added to 1.00 L of…
A: The objective of this question is to calculate the pH of different solutions after the addition of a…
Q: How might you carry out the following selective transformations? Use the letters from the table to…
A: The reagent selectively reacts only one functional group in presence of other potential functional…
Q: The Solubility Product Constant for silver phosphate is 1.3 x 10-20. The maximum amount of silver…
A: Answer:Ionic compounds that have low solubility in water are called sparingly soluble compounds. For…
Q: Provide the proper IUPAC Name for the alkene below Br Br
A: To solve this problem we have to write the IUPAC name of the given compound.
Q: entify the stronger and weaker Bronsted acid, assign the smaller and larger pKa, identify the…
A: The objective of this question is to identify and compare Bronsted acids, assess acid strength based…
Q: = NaOH E2
A: The Wolff-Kishner reduction is a chemical reaction that converts carbonyl compounds like ketones and…
Q: Use appendix E : Thermodynamic data from p. 203 workbook, to calculate the enthalpy of formation…
A: Given:Carbon monoxide (g) reacts with hydrogen gas to produce carbon tetrahydride (g) and water…
Q: What is the pH at the equivalence point when 30. mL of 0.310 M hydroxylamine is titrated with 0.150…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: A 8.06 g sample of benzoic acid was dissolved in water to give 60.6 mL of solution. This solution…
A: Mass of benzoic acid = 8.06 gVolume of solution = 60.6 mLConverting to liters:Molarity of NaOH =…
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A: Diels-Alder reaction is a pericyclic reaction in which a conjugated diene (either substituted or…
Q: What element is represented by X in the atomic symbol notation 195 is over 78X ? Select one: A.…
A: The objective of the question is to identify the element represented by X in the atomic symbol…
Q: Draw the major elimination and substitution products formed in this reaction. Use a dash or wedge…
A: Substitution reaction is an organic reaction where an incoming nucleophile replaces the functional…
Q: A sample is run on a TLC plate and gives a single spot as shown below on the left. However, the…
A: TLC chromatography
Step by step
Solved in 1 steps with 1 images

- Give the relationship between the pair of molecules below by selecting from the choices below: A. Enantiomers B. Diastereomers (Geometric) C. Diastereomers (Configurational) D. ConstitutionalIsomers–SkeletalE. Constitutional Isomers – Functional F. Constitutional Isomers – Position G. Identical moleculesRank the following groups in order of decreasing priority. −F, −NH2, −CH3, −OH10. Draw all isomers of difluorochlorobutane including stereoisomers
- How much of the R enantiomer is present in 10g of a mixture that has an enantiomeric excess of 30% of the S isomer?Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry to the molecules on part a and b in the pictureWhat is the relationship between stereoisomers and enantioners? Give a real life example?
- For the molecule with formula C7H14O please draw the constitutional isomers with the properties as indicated below. Contains a 6-membered ring (indicate stereo chemistry using wedge/dash if necessary) Contains a proton with the pKa of approximately ~44 Contains a carbonyl group and no double bonds Is a meso compoundThe LD50 (“Lethal Dose, 50 %”) is a value used in toxicology which gives the amount of a substance required to kill 50 % of a test population. It is usually reported based on body weight. For racemic carvone LD50 is reported as 1640 mg/kg. 1. Would it consider carvone to be toxic to an average person? Explain. 2. Would it be expected this the LD50 value to be the same for each enantiomer? Explain at the molecular level in stereochemical terms.Draw all the isomeres of a thioester with molecular formula C4H8OS
- Need help differentiating enantiomers,diastereomers,constitutional isomers and whether it is a same compoundHow many stereoisomers does ephedrine have?4-Chloro-2-pentene has a double bond that can have either the E or the Z configuration and a stereogenic center that can have either the R or the S configuration. How many stereoisomers arepossible altogether? Draw the structure of each, and group the pairs of enantiomers.

