Exp 5 Separation Develop a separation scheme for the following compounds. Make sure that you end with each compound in an organic phase. Use the following solvents; water, methanol, hexane, CH2C12, Ether, 1M NaOH, 6M NaOH, 1M HCI, 6M HCI. Oc-OH H2N. H3C CH3 Acid Base weutral te Le ecp at
Exp 5 Separation Develop a separation scheme for the following compounds. Make sure that you end with each compound in an organic phase. Use the following solvents; water, methanol, hexane, CH2C12, Ether, 1M NaOH, 6M NaOH, 1M HCI, 6M HCI. Oc-OH H2N. H3C CH3 Acid Base weutral te Le ecp at
Chapter89: Thin-layer Chromatography
Section: Chapter Questions
Problem 3P
Related questions
Question
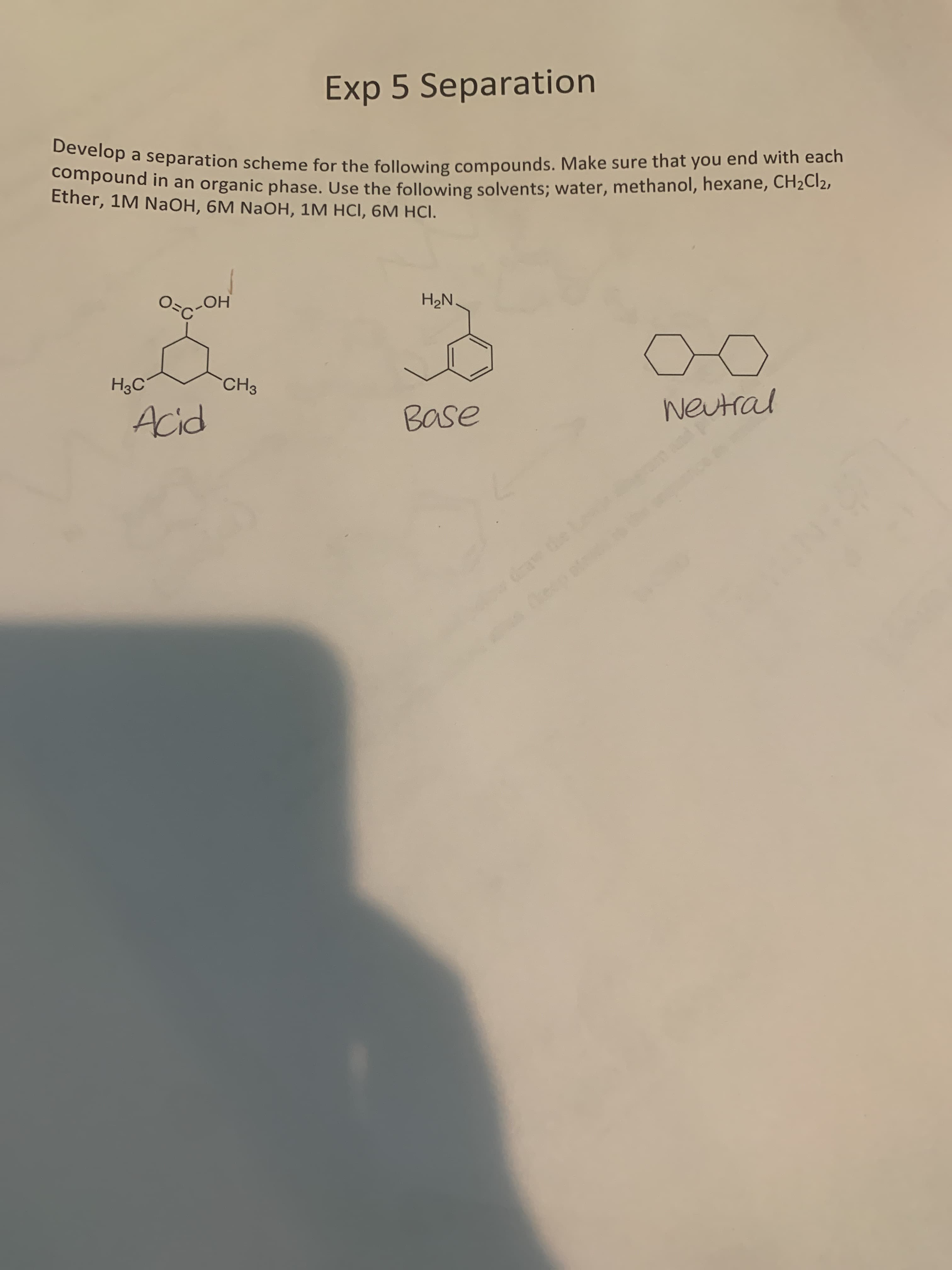
Transcribed Image Text:Exp 5 Separation
Develop a separation scheme for the following compounds. Make sure that you end with each
compound in an organic phase. Use the following solvents; water, methanol, hexane, CH2C12,
Ether, 1M NaOH, 6M NaOH, 1M HCI, 6M HCI.
Oc-OH
H2N.
H3C
CH3
Acid
Base
weutral
te Le
ecp at
Expert Solution
Step 1
Given problem is based on ternary mixture separation .
Here, three types of compound give and we can separate them using solvent such as ether.
Let's see flowchart of separation.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT