Find all functions o for which u(x, t) = 4(x – ct) is a solution of the heat equation 1 ди - o < x < 0 dx² k dt where k and c are constants.
Q: A sample of an ideal gas in a cylinder of volume 4.02 L at 298 K and 2.51 atm expands to 8.75L by…
A:
Q: 5.0 kg of steam at 0.7 MPa and 250°C enters a pipeline and flows steadily along it at constant…
A: By using the properties of steam table The enthalpy of the steam at the pressure of 0.7 MPa and…
Q: 1) One mole of a van der Waals fluid with empirical constants a and b is contained within a cylinder…
A:
Q: It is difficult to extinguish a fire on a crude oil tanker, because each liter of crude oil releases…
A:
Q: Combustion gases enter the nozzle of a jet engine at 3.8 bar and 820 o C. The nozzle is well…
A:
Q: piston cylinder assembly initially contains 25g of saturated water vapor at 600kpa pressure. Then,…
A:
Q: For a certain gas the critical temperature is 20 K and reduced temperature is 40 K, then the actual…
A: Option (c) 800K is correct option
Q: The cooling system of a car engine contains 20.0 L of water (1L(1L of water has a mass of 1kg1kg ).…
A: Given Data Heat transfer, Q=836 KJ Specific heat of water, C=4180 J/Kg 1L has 1Kg 20L has 20 Kg…
Q: Wet steam at 200°C and 20% moisture is compressed in an irreversible adiabatic process to a process…
A:
Q: A sample of an ideal gas in a cylinder of volume 4.02 L at 298 K and 2.51 atm expands to 8.75 L by…
A: Given data: The initial volume of the ideal gas is V1=4.02 L=4.02×10-3 m3 The initial temperature of…
Q: 5.0 How much heat is needed to completely vaporized 100 kg of ice at-10 °C if the pressure is held…
A: Given:m=100 kgT=-10°CP=200 kPaLHF=320 kJ/kgLHV=2202 kJ/kg
Q: find the relation between the work done in the isothermal expansion and the work done in the…
A: Work done is isothermal expansion : Process 1-2: The temperature will be constant in the isothermal…
Q: A 19.2 g piece of dry ice at T = 195 K is dropped into a beaker containing water at T = 273.15 K.…
A: Given, ∆Hsys=25.2 kJ/moleT=195 KTsurr=293.15 K
Q: A 6-cm-diameter steel rod at 38°C is suddenly immersed in a liquid at 93°C with h =110 W/m²·K. How…
A: given; ⇒conductivity of carbon steel (K1)=49.8w/m. K⇒conductivity of stainless steel (K2)=13w/m.…
Q: c. Saturated air at 50oF db is heated to 80oF dry bulb without the addition of moisture. If 1000 cfm…
A: We are suppose to solve only one question. Please post other question as a separate question.
Q: 2. A Carnot engine operating between 775 R and 305 K produces 54 kJ of work, Determine (a) QA (BAS…
A: η= 1-T2T1 , T1=775k and T2= 305kη= 1-305775η=0.6065 (c)work done = 54kJAlso,…
Q: A turbine is installed between a feed line at 1800 psia and 250 ° F and the outlet line at 800 psia…
A: It is asked to find the temperature at the exit of the valve
Q: B/A container which has a volume of 0.1 m' is fitted with a plunger enclosing 0.5 kg of steam at 0.4…
A:
Q: perheated steam at 0.5 MPa and 400 °C enters an adiabatic nozzle with a low velocity and leaves at…
A:
Q: A 25.5-g aluminum block is warmed to 65.4 °C and plunged into an insulated beaker containing 55.2 g…
A: Given data: Heat capacities are:
Q: 1. Determine Z and V for steam (HzU) at 250 °C (523.15 K) and 1800 kPa by the following: PV a. The…
A: Given, P = 1800 kPa T = 250oC = 523.15 K We know that, PV = RT V = RT/P = 0.287 * 523.15 / 1800 =…
Q: 5.0 How much heat is needed to completely vaporized 100 kg of ice at -10 °C if the pressure is held…
A:
Q: The electromotive force, EMF of the galvanic cell Pb(s)| PbCl2(s) | HCI(aq) | AgCl(s) | Ag(s) where…
A:
Q: Find the mass in metric tons per day of at -5 °C that would theoretically made in a refrigerating…
A: Let's first calculate heat absorbed (Qabs) by CO2 in a day: Qabs = m˙(h1 - h2) h1 = specific…
Q: Find the temperature at any time using separation of variables (k =- P) Ug %3D U(0,t) = U(L,t) =0…
A:
Q: Determine the MW and specific heat ratio of a gas whose Cv=1.386 kJ/kg.K and R= 0.485 kJ/Kg.K…
A:
Q: Derive the thermodynamic identity for G (equation 5.23), and from it the three partial derivative…
A: The gibs free energy of the system is given as, G=U-TS+PV
Q: Find the mass in metric tons per day of at -5 °C that would theoretically made in a refrigerating…
A: Answer: The mass in metric tons per day is 32.8340 tons/day.
Q: Q.1) An insulated piston-cylinder de vice contai ns 10 L of saturated liquid water at a constant…
A:
Q: 2. 5.0 kg of steam at 0.7 MPa and 250°C enters a pipeline and flows steadily along it at constant…
A: (A) The saturation temperature at the pressure of 0.7 MPa is Tsat=164.95°C The droplets of the water…
Q: 2. A Carnot engine operating between 775 R ar 305 K produces 54 kJ of work. Determine a QA (BAS…
A: Given, T1=775K, T2=305K, W=54kJ
Q: Estimate the specific heat relationship R = Cp - Cv (kJ/kg°C), for R134a at 0.24MPa and 40°C. Using…
A: Given Data⇒ Refrigerant R-134a Pressure(P)=0.24 MPa Temperature(T)=400C
Q: The evaporative condenser of an ammonia refrigeration plant has a water flow rate of 100 kg/s and…
A: To find : The percent of make up water needed in the cooling tower. Given: The inlet temperature…
Q: Steam enters a diffuser steadily at 300 kPa and 300 °C having a velocity of 80 m/s. The inlet…
A:
Q: At 1 kg copper part, initially a 500K is plunged into a tank containing 4 kg of liquid water…
A:
Q: By circulating water inside a copper tube with an internal diameter of 5 cm, it is desired to heat…
A: Internal diameter of the tube, d=5 cm =0.05 mMass flow rate of water, m˙w=100 kg/min=10060kg/sWater…
Q: PRACTICE: 1 mole of a monoatomic ideal gas undergoes an isothermal expansion from 0.01 m3 to 0.05…
A:
Q: Q1) 1kg saturated liquid water-vapor mixture at a pressure P = 250kPa with a quality of x = 0.6 is…
A:
Q: An inventor proposes to make a heat engine using water/ice as the working substance, taking…
A: The efficiency of the heat engine is, Here, is the work done…
Q: (d) Two mole of an ideal monatomic gas goes from a to c along the diagonal path in the figure below.…
A: Ideal gas equation relates the temperature, volume and pressure of an ideal gas. It is given as…
Q: Determine the MW and specific heat ratio of a gas whose Cv=1.386 kJ/kg.K and R= 0.4
A:
Q: 1. Combustion gases enter the nozzle of a jet engine at 3.8 bar and 820°C. The nozzle is well…
A:
Q: 4.26 A cylinder of free volume 0.283 m is divided into two compartments by a piston. Initially the…
A: Given data: The final pressure of the steam is equal to the pressure of the supply line because of…
Q: 4. Refrigerant R-134a is used as the working fluid in a Carnot refrigerator. The refrigerator…
A: Given data: TH=313.15 KTL=273.15 K Need to determine the quality of the refrigerant at the entry and…
Q: b) Cousider a typical enclosed room in a house that is very well thermally insulated with constant…
A: Typical enclosed room in a house is thermally insulated that is Q=0 Volume V=4×6×3=72 m3 Initial…
Q: Carbon dioxide (Cp = 871 J/kg.K and Cv = 678 J/kg.K) is compressed from 0.4 m3 at 100 kPa and 14 oC…
A:
Q: Steam at 20 MPa and 620°C enters a steam turbine and expands to a condenser pressure of 100 kPa. An…
A:
Q: 2 moles of ideal gas has the transformations shown in the diagram below where the 2-3 is an…
A:
Q: Fluid falls from a 50 m elevation. a. Determine the velocity of the fluid at the surface of the…
A: Given data: H= 50 m C = 4.19 kj/kg* °C Need to determine the velocity of the fluid at the surface of…
I need help with this question and also show your work
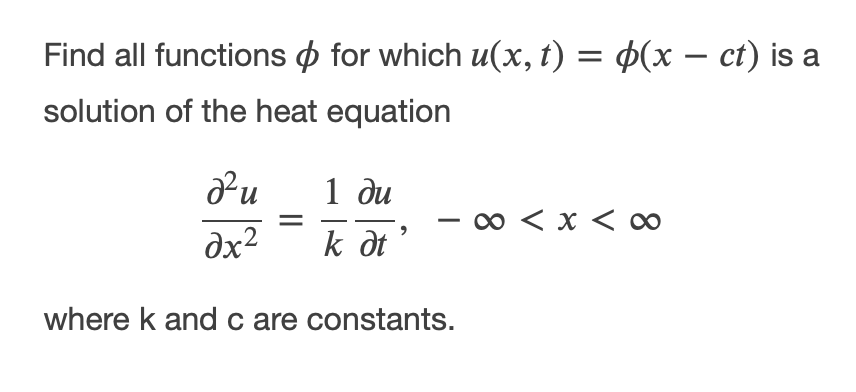
Step by step
Solved in 2 steps with 2 images

- J 6 A semi-infinite solid, with a boundary condition of convection on the surface (Tfluid = 100°, h = 220 W/m2-°C) has at t=0s, a uniform temperature of Ti = 0°C, draw the isotherm of 60°C, at time t = 2h. Consider the solid to be cast iron.SHOW COMPLETE AND LEGIBLE SOLUTION. Assume the surroundings to be at 25OC and 100 kPa.A block of ice at a temperature of 0∘C is crushed by a 5.2 kg mass piston being dropped a distance of 0.35 m onto the ice. This act is repeated 2,500 times. The piston has an initial temperature of 20C, a final temperature of 18C, and a specific heat capacity of 550 J/kg⋅K. Determine how much ice is melted. Assume none of the work done by the ice on the piston heats it. Use a value of 336 kJ/kg for the specific latent heat of fusion of ice. Give your answer to the nearest gram. please help
- A block of ice at a temperature of 0∘C is crushed by a 5.2 kg mass piston being dropped a distance of 0.35 m onto the ice. This act is repeated 2,500 times. The piston has an initial temperature of 20C, a final temperature of 18C, and a specific heat capacity of 550 J/kg⋅K. Determine how much ice is melted. Assume none of the work done by the ice on the piston heats it. Use a value of 336 kJ/kg for the specific latent heat of fusion of ice. Give your answer to the nearest gram.A 0.4 kg metal bar initially at 1000 K is removed from an oven and quenchedby immersing it in a closed tank containing 10 kg of water initially at 295 K. Eachsubstance can be modeled as incompressible. An appropriate constant specific heat valuefor water is 4.22 kJ/kg-K and an appropriate value for the metal is 0.42 kJ/kg-K. Heattransfer from the tank structure can be neglected. Determine (a) the final equilibriumtemperature of the metal bar and the water in K, and (b) the amount of entropy producedin kJ/K. List any key assumptions. [ Ans. (a) T = 297 K ]find the driving pressure for a stack that has a height of 18 m and carry a hot gas mixture with an average density of 1.2 k/m3. If you know that the total heat rejection by the stack is 1450 KJ and the c, of the hot gas is 1.8 KJ/Kg.K while the hot gas inlet and outlet temperature are 650 K and 500 K respectively. Assume the hot gas pressure as 2.3 bar while the ambient pressure is 1 bar.
- Derive an equation for work done by the system during isothermal expansion process.Heat is added to fixed 0.15 m3 volume of steam initially at a pressure of 400 kPa and a quality of 0.5. Estimate the final temperature if 800 kJ of heat is added.In an aluminum container weighing 0.30 kg are 0.70 kg of water in thermal equilibrium at a temperature of 70°C. Container and water temperature together cool at a rate of 2.5 K/min. What is the heat flux leaking from the container?
- Derive the thermodynamic identity for G (equation 5.23), and from it the three partial derivative relations 5.24.2 m^3/min of a light oil are to be heated from 20C to 100C (with zero vaporization) in an exchanger using 250 kPa steam of 90% quality. The heat losses to the surrounding air have been estimated to be 5% of the heat transferred from the condensing steam to the oil. If the steam condensate leaves at its saturation point, what mass of steam perhour will be used in the exchanger? For light oil, Specific gravity= 0.88Specific heat= 2.00 kJ/kg-KFor steam at 250 kPa,Saturated liquid: hf= 535.49 kJ/kgvf=0.001067 m^3/kgSaturated vapor: hg= 2716.8 kJ/kgvg= 0.7188 m^3/kgA carbon steel pipe (thermal conductivity of 38 W/m oC) with an external diameter of 5 cm and a wall thickness of 4 mm, transports saturated water vapor to an equipment in which water evaporates from the sucrose solution to produce sugar. There are few curves in the system, so you decide to neglect their effects. The ambient temperature in which the pipe is located is 28°C and the temperature of the steam for the pressure in which it is in the line is 120°C. By using an empirical correlation to estimate the convection coefficient of the cooling law of Newton for steam condensing inside tubes you find the value of 15000 W/m 2 oC. This value is so high that we can neglect the resistance to convective transport inside the pipe, if you want, calculate the resistance in question and check. Considering an external convective coefficient of 25 W(m2 oC), we estimate that the water will lose ____________ W/m of pipe. If we want to insulate the pipe with urethane foam (conductivity between 0.016…

