Following is the mass spectrum of 1,2-dichloroethane. Propose a structural formula for the cation of m/2 63. X? D 100- 80- 60- 40- 20 10 20 60 m/2 . CI can represent either or C • Br can represent either 79Br or 18 . In cases where there is more than one answer, just draw one You do not have to include radical electrons in your answer. 70 MO 100 99 H₂C-CH₂ MW-98 Spectrs images are reproduced by permission from Signa-Aldrich and the National Institute of Advanced Industrial Science and Technology (Soesweb 2/7/09, 2/23/09, 3/10/09) 100 110 120 1.00, 8/26/S
Following is the mass spectrum of 1,2-dichloroethane. Propose a structural formula for the cation of m/2 63. X? D 100- 80- 60- 40- 20 10 20 60 m/2 . CI can represent either or C • Br can represent either 79Br or 18 . In cases where there is more than one answer, just draw one You do not have to include radical electrons in your answer. 70 MO 100 99 H₂C-CH₂ MW-98 Spectrs images are reproduced by permission from Signa-Aldrich and the National Institute of Advanced Industrial Science and Technology (Soesweb 2/7/09, 2/23/09, 3/10/09) 100 110 120 1.00, 8/26/S
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter14: Mass Spectrometry
Section: Chapter Questions
Problem 14.29P
Related questions
Question
4
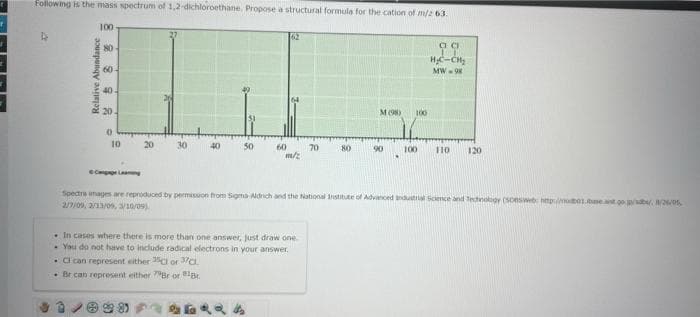
Transcribed Image Text:Following is the mass spectrum of 1,2-dichloroethane. Propose a structural formula for the cation of m/2 63.
2
100
80
60
40-
20.
0
10
20
30
40
50
60
62
24
m/z
. In cases where there is more than one answer, just draw one.
You do not have to include radical electrons in your answer.
• Cl can represent either 35Cl or 37C.
• Br can represent either 79Br or 818r.
70
80
M (98) 100
90
99
H₂C-CH₂
MW-98
Spectra images are reproduced by permission from Sigma-Aldrich and the National Institute of Advanced Industrial Science and Technology (Sonsweb: http://mob01buse antodb. 11/26/05
2/7/09, 2/13/09, 3/10/09)
100 110 120
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
