For each chemical reaction in the table below, decide whether the highlighted reactant is a Brønsted-Lowry acid, a Brønsted-Lowry base, or neither. highlighted reactant reaction Bronsted-Lowry acid Bronsted-Lowry base neither HBr(aq) + NH3(aq) → Br (aq) + NH (aq) HBr(aq) + NH,(aq) → Br (aq) + NH (aq) Br (aq) + NH (aq) → HBr(aq) + NH,(aq) Br (aq) + NH (aq) → HBr(aq) + NH,(aq)
For each chemical reaction in the table below, decide whether the highlighted reactant is a Brønsted-Lowry acid, a Brønsted-Lowry base, or neither. highlighted reactant reaction Bronsted-Lowry acid Bronsted-Lowry base neither HBr(aq) + NH3(aq) → Br (aq) + NH (aq) HBr(aq) + NH,(aq) → Br (aq) + NH (aq) Br (aq) + NH (aq) → HBr(aq) + NH,(aq) Br (aq) + NH (aq) → HBr(aq) + NH,(aq)
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter17: Acid-base(proton Transfer) Reactions
Section: Chapter Questions
Problem 14E: In the following net ionic reaction, identify each species as either a Brnsted-Lowry acid or a...
Related questions
Question
Thank you!

Transcribed Image Text:Tes
Identifying Bronsted-Lowry acids and bases
For each chemical reaction in the table below, decide whether the highlighted reactant is a Brønsted-Lowry acid, a Brønsted-Lowry base, or neither.
highlighted reactant
reaction
Bronsted-Lowry
acid
Bronsted-Lowry
base
neither
+
HBr(aq) + NH,(aq)
Br (aq) + NH, (aq)
HBr(aq) + NH,(aq)
Br (aq) + NH (aq)
Br (aq) + NH (aq) → HBr(aq) + NH,(aq)
4
Br (aq) + NH (aq)
HBr(aq) + NH3(aq)
4
||
Expert Solution
Step 1
Bronsted-Lowry acid-base theory:
According to the Bronsted-Lowry acid-base theory, acid is a substance that gives H+ ions and base is a substance that accepts the H+ ions.
The species which are differing by the H+ called conjugate acid-base pairs.
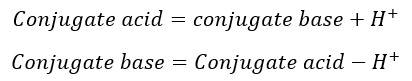
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning