For each reaction in the table below, write the chemical formulae of any reactants that will be oxidized in the second column of the table. Write the chemical formulae of any reactants that will be reduced in the third column. reactants oxidized reactants reduced reaction 8Mg(s) + S,(s)8MgS (s) 2Cu(s) + CL, (g) - 2CUC1(s) Ni(s) + 1, ()- Nil,(6) Aの
For each reaction in the table below, write the chemical formulae of any reactants that will be oxidized in the second column of the table. Write the chemical formulae of any reactants that will be reduced in the third column. reactants oxidized reactants reduced reaction 8Mg(s) + S,(s)8MgS (s) 2Cu(s) + CL, (g) - 2CUC1(s) Ni(s) + 1, ()- Nil,(6) Aの
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter19: Electrochemistry
Section: Chapter Questions
Problem 19.99QP: Some metals, such as iron, can be oxidized to more than one oxidation state. Obtain the balanced net...
Related questions
Question
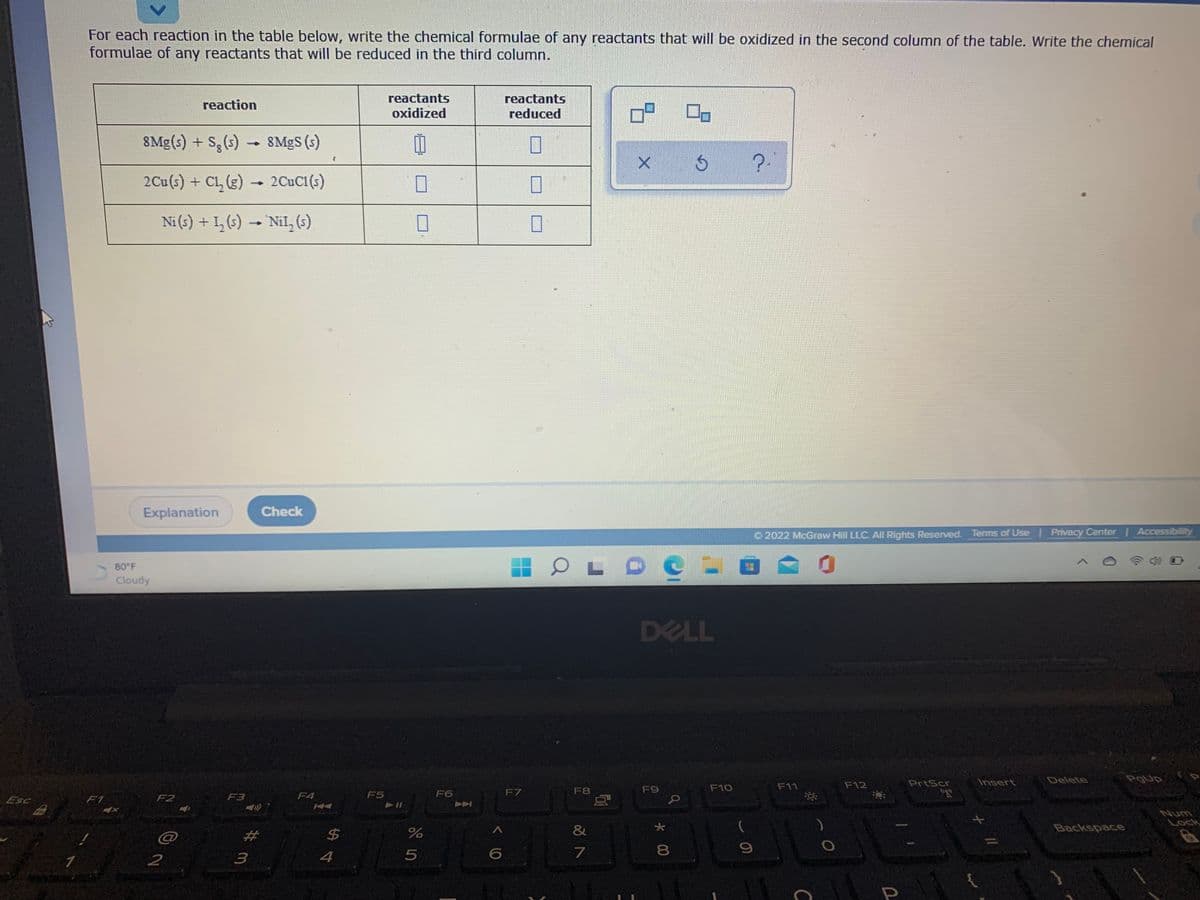
Transcribed Image Text:For each reaction in the table below, write the chemical formulae of any reactants that will be oxidized in the second column of the table. Write the chemical
formulae of any reactants that will be reduced in the third column.
reactants
oxidized
reactants
reaction
reduced
8Mg(s) + S,(s) SMGS (s)
?-
2Cu(s) + Cl, (g)
2CuC1 (s)
Ni (s) + 1, (s) - Nil, (6)
Explanation
Check
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility
80 F
Cloudy
DELL
PrtScr
Insert
Delete
PgUp
F9
F10
F11
F12
F3
F4
F5
F6
F7
F8
Esc
F1
F2
Lock
%24
&
Backspace
6.
7
8.
69
2
3
4
C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning