Q: The preparation of homeopathic "remedies" involves repeated dilution of solutions containing active…
A: The mass of arsenic trioxide is 18.0 g. The moles of arsenic trioxide is calculated as,…
Q: A50 ml. sample of a 48 0 % (m/v) acetic acid (HC,H3O2) solution is added to water to give a final…
A:
Q: A 10.5%(w/w) NiCl2 solution has a density of 1.149 g/mL. What is the mass in grams of Ni2+ contained…
A: Given : 10.5%(w/w) means 10.5 grams of NiCl2 (solute) present in 100 grams of solution. Mass of 1 L…
Q: What is the mass % of propylene glycol in a 2.77 M solution of propylene glycol (MM = 76.09 g/mol)…
A: Mass percent shows the mass of solute present during a given mass of solution. Mass percent is…
Q: Many remote villages depend solely on wells to supply their drinking water. A ground water analyst…
A: we have to calculate volume of sample of well water
Q: Molecular Weight of Zinc Sulfate Heptahydrate 287.6 g/mol 7.199 Mass of Zinc Sulfate Heptahydrate…
A: Molar concentration (also known as molarity, quantity concentration, or material concentration) is a…
Q: An aqueous solution of potassium sulfate, K2SO4 , is made by dissolving 15.1 grams of potassium…
A: Given: Mass of K2SO4 = 15.1 g. And volume of solution filled = 250 mL = 0.250 L…
Q: Is water available in ethanol?
A: Ethanol is an organic chemical compound. It is a primary alcohol. Its formula is CH3−CH2−OH. It…
Q: How much water ( in mL) must be added to a 4.01 M sulfuric acid solution to obtain 193.1 mL of 2.00…
A:
Q: A solution of cobalt(II) chloride is analyzed, and the concentration of cobalt(II) ions is found to…
A: Given, Concentration of cobalt(II) ions(Co2+) = 0.27 mol/L Concentration of Chloride ion (Cl^-1) =…
Q: For the chemical equation 2HBr (aq) + Ba(OH)2 (aq) yields to 2H2O (L) + BaBr2 (aq) write the net…
A: Net ionic equation involves only the ions that actually take part in the reaction excluding the…
Q: A 66.7 mL sample of 18.0 M sulfuric acid was diluted with enough water in a volumetric flask to make…
A: The solution is as follows:
Q: What volume, in mL, of a 0.35 M Mg(NO3)2 aqueous solution contains 87 g of Mg(NO3)2?
A: Definition of Molarity is, Molarity is the number of moles of solute dissolve in one litre of the…
Q: A chemist needs 225 mL of 3.0 M HCl. What volume of 12 M HCl must be dissolved in water to form this…
A: Given: Volume of HCl (V1)=225 ml Molarity of HCl (M1)=3.0 M M2=12 M To find: V2 Solution: According…
Q: 1. A solution of sodium bicarbonate is prepared by adding 45.00 g of sodium bicarbonate to a 1.00-L…
A:
Q: 11.A sample of 10 g of potassium iodide is dissolved in a beaker. Then it is poured in a volumetric…
A: Given : A sample of 10 g of potassium iodide is dissolved in a beaker. Then it is poured in a…
Q: A 30.54-g sample of compound X (Molar Mass 171 g/mol) is dissolved in water for a total volume of…
A:
Q: Calculate the normality of 12.5 g of potassium permanganate (KMnO4) in 475 mL solution
A: The number, n = 5 for KMnO4 as it accepts 5 electrons to become Mn2+
Q: Sodium carbonate is often added to laundry detergents to soften hard water and make the detergent…
A: Given,Concentration of Ca2+ = 3.8×10−3 M = 3.8×10−3 mol/LConcentration of Mg2+ = 1.1×10−3 M =…
Q: In a flask, you heat a mixture of 735.4 g of sodium nitrate and 700.0 grams of water until all of…
A: Answer:- this question is answered by using the simple concept of calculation of solubility of the…
Q: An aqueous solution is 6.00 % methanol CH3OH by mass with a density = 0.988 g/mL. What is the…
A: The molarity is the concentration term defined as the number of moles of solute present per liter of…
Q: An aqueous solution of manganese (II) iodide,MnI2,is made by dissolving 21.1 grams of manganese(II)…
A: Weight/Volume percentage = it tells about amount of solute present per 100 mL of solutionWeight of…
Q: A 15.2-g sample of sodium carbonate is added to a solution of acetic acid weighing 19.4 g. The two…
A: Soln
Q: The fish canning industry has a waste effluent with the specification as follows: BOD5 = 150 mg/L,…
A: High levels of BOD and COD can be treated with the help of ozone. Ozone is an oxidant and is soluble…
Q: In the laboratory you dessolve 22.3g of iron (III) fluoride in a volumetric flask and add water to a…
A: The number of moles of solute to the volume of solution in liters is known as molarity.
Q: How many liters of a 1.85 M KNO3 solution can be prepared from 53.3 g of KNO3? Express your answer…
A: First convert the given KNO3 mass to moles. Moles=MassMolar mass=53.3 g101.10 g/mol=0.527 mol
Q: You are a work study student in our chemistry department. Amy, your supervisor has just asked you to…
A: Molarity (M) of a solution is defined as the number of moles of solute dissolved per volume of the…
Q: Calculate the normality of 0.321 g sodium carbonate when it is mixed in a 250 mL solution
A:
Q: What mass of salt (NaCI) in grams (g) is required to prepare 2.552 L ofD51/¼NS solution? Give your…
A: Given that : The volume of D514NS solution = 2.552 L We have to calculate the mass of NaCl required…
Q: Use the data below for the concentrated acids and bases. Reagent Concentration %w/w 99.7 29.0 37…
A: we can prepare a diluted solution from concentrated stock solution using dilution law
Q: Water and acetonitrile, CH3CN, are miscible (can be mixed in any proportions). However, when water…
A: The given statement indicates that the solution of water and acetonitrile shows deviation from…
Q: What volume of 1.24 M KMnO4, in liters, contains 422 g of solute? Volume %3D
A: In order to find the volume of the solvent required we first write down the given data: Molarity=…
Q: Calculate the mass of NaOH in 150 cm^3 of a 0.1M solution provided.
A: no of moles = mass/molar mass molarity= no of moles of solute/volume of solution in liter molarity…
Q: Write the net ionic equation (Including phases) for: Pb(ClO4)2(aq)+K2CO3(aq)⟶PbCO3(s)+
A:
Q: How many milliliters of a 0.900% (m/v) normal saline solution can be prepared from 2.00 g of sodium…
A: Percentage (mass/volume) can be defined as the weight of solute in g per volume of solution in mL,…
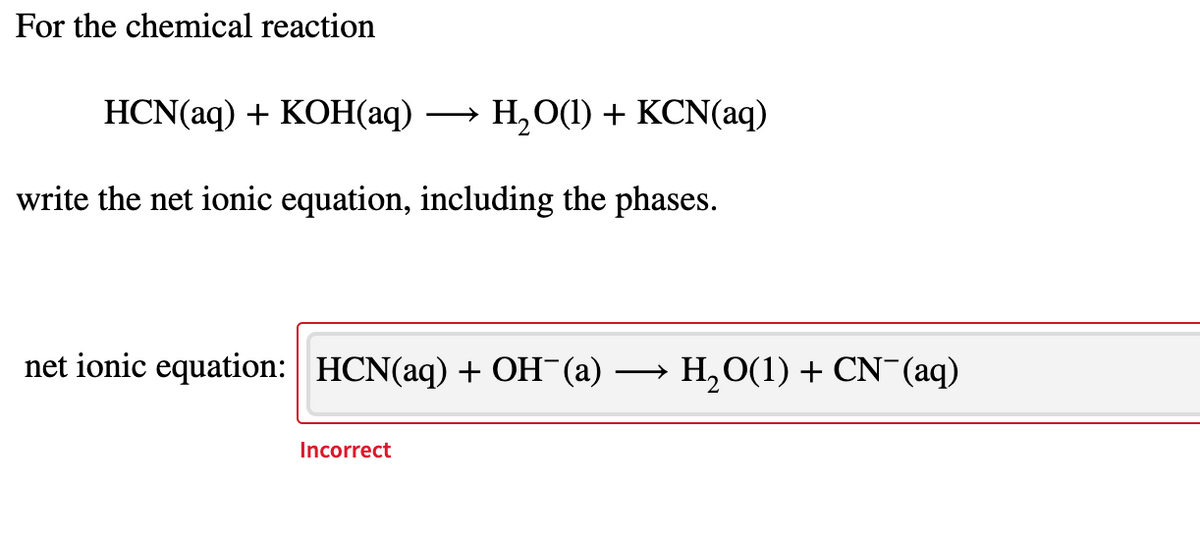
Q: For the chemical reaction…
A: Ionic reaction means ions are participating in reaction. Given reaction is in molecular form…
Q: From the graph of temperature vs. volume data from a fractional distillation of 100 mL…
A: Methanol and water fractional distillation is an analytical process used to separate compounds…
Q: An aqueous solution of sodium chloride,NaCl , is made by dissolving 1.01 grams of sodium chloride in…
A: Weight/volume percentage= Mass of soluteVolume of solution×100%
Q: Calculate the normality of 0.321 g sodium carbonate when it mixes in a 250 mL solution.
A: Given, Mass of Sodium carbonate (Na2CO3) = 0.321 g Molat mass of Na2CO3 = 106 g/mol Mole = mass/MM…
Q: Conversion of units: ✓ Blood Sodium 100 mmol/mL to mEq/L ✓ Blood CO2 15 vol % to mmol/L ✓ Blood…
A: We have to convert blood sodium 100 mmol/mL to mEq/L. For sodium 1 mole = 1 equivalent. So, 100…
Q: An aqueous solution of acetone is made by transferring 6.86 mL of liquor acetone to a 100 mL…
A:
Q: An aqueous solution of nickel(II) bromide, NiBr2, is made by dissolving 11.2 grams of nickel(II)…
A: Given-> Weight of nickel(2)bromide = 11.2 gm Volume = 250 ml
Q: If 426 mL of 0.204 M HCI solution is needed to neutraize a solution of Ca(OH)2. how many grams of…
A:
Q: Sodium carbonate is often added to laundry detergents to soften hard water and make the detergent…
A: Given,3.8×10−3 M in Ca2+ and 1.1×10−3 M in Mg2+ The average capacity of a washing machine is 24.5…
Q: Lead ions (Pb2+)can be removed from solution by precipitation with SO42− Suppose that a solution…
A: A chemical equation in which electrolytes of an aqueous solution are represented in the form of…
Q: Write balanced net ionic equation for KOH(aq)+HC2H3O2(aq)→H2O(l)+KC2H3O2(aq). Express your answer…
A: Balanced chemical equation of a reaction is written according to law of conservation of…
Q: One of the Ionic compounds in sports drinks is potassium di hydrogen phosphate. The label on one of…
A: 30mg of potassium in present in 240ml of the solution. The weight of potassium dihydrogen phosphate…
Q: calculate the molarity. (a) 8.0 g of sodium hydroxide, NaOH, dissolved in 150 cm3 of water.
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- When HCl(aq) is added to calclum carbonate solid, carbon dioxide gas, liquid water and aqueous calcium chloride form: Caco,(s) + 2HCI(aq) - CO,lg) • CaCl, faq) + H,00) This reaction is used by geologists to confirm the presence of limestone (calcium carbonate) in minerals. In this experiment you are trying to determine the effect of concentration of HCI(aq) on the production of carbon dioxide. a. Based upon the information above, come up with a question to answer. (C:1) You can use the following equipment to answer this question: calcium carbonate chips, 1.90 molL HClaq). pH meter (measures hydrogen ion concentration), beaker, timer, apparatus to measure gas produced b. Based upon the equipment provided above, design a procedure to answer your question. Ensure you provide numbered steps. (1:3, C:2) c. Hypothesize an answer to your question. (I:1) Give reasons for your hypothesis. Ensure you are very specific. (A3) From your experiment, you collect the following data: Reactant…Hi,can you slove Q.no 1 and Q.no. 2 , all Please. Thank You 1. A student is asked to perform a neutralization reaction between hydrochloric acid, HCl and sodium hydroxide, NaOH. The student transfers 5.00 mL of HCl solution to the titration flask and then adds 25.0 mL of distilled water and 3 drops of phenolphthalein indicator to the solution. The resulting solution uses 35.5 mL of 0.125M NaOH solution to reach the end point.a. Write a balanced chemical equation for the neutralization process.b. What will be the color of solution at the end point?c. Calculate the number of moles of NaOH used for complete neutralization.d. Calculate the strength in terms of molarity for HCl solutione. Calculate the m/v percent strength of the HCl solution 2. If a student calculated that 0.0035 moles of acetic acid present in a 5.0 mL sample of vinegar. Then calculate the mass/volume percent of acetic acid in vinegarA student prepares a dilute solution of sodium hydroxide, NaOH (aq), starting with 6 M sodium hydroxide. She then titrates a 1.372 g sample of KHP with the dilute sodium hydroxide solution, NaOH (aq), to a phenolphthalein end point. a) If the titration required 21.84 mL of sodium hydroxide, NaOH (aq), calculate the molar concentration of the sodium hydroxide solution, NaOH (aq). (Remember that KHP is potassium hydrogen phthalate, KHC8H4O4, NOT potassium hydrogen phosphorus!) b) The student uses the same sodium hydroxide to titrate 10.00 mL of vinegar to a phenolphthalein end point. If the titration required 27.48 mL of sodium hydroxide, NaOH (aq), calculate the molar concentration of acetic acid, HC2H3O2 (aq), in the vinegar. c) Calculate the mass percent of acetic acid, HC2H3O2(aq), in the vinegar using the molar concentration for acetic acid, HC2H3O2 (aq), determined in part b and assuming the density of the solution is 1.01 g/mL.
- A student prepares a dilute solution of sodium hydroxide, NaOH (aq), starting with 6 M sodium hydroxide. She then titrates a 1.372 g sample of KHP with the dilute sodium hydroxide solution, NaOH (aq), to a phenolphthalein end point. A.) If the titration required 21.84 mL of sodium hydroxide, NaOH (aq), calculate the molar concentration of the sodium hydroxide solution, NaOH (aq). (Remember that KHP is potassium hydrogen phthalate, KHC8H4O4, NOT potassium hydrogen phosphorus!) B.) The student uses the same sodium hydroxide to titrate 10.00 mL of vinegar to a phenolphthalein end point. If the titration required 27.48 mL of sodium hydroxide, NaOH (aq), calculate the molar concentration of acetic acid, HC2H3O2 (aq), in the vinegar. C.) Calculate the mass percent of acetic acid, HC2H3O2 (aq), in the vinegar using the molar concentration for acetic acid, HC2H3O2 (aq), determined in part b and assuming the density of the solution is 1.01 g/mL.To measure the amount of calcium carbonate CaCO3 in a seashell, an analytical chemist crushes a 4.200g sample of the shell to a fine powder and titrates it to the endpoint with 179.mL of 0.370M hydrogen chloride HCl solution. The balanced chemical equation for the reaction is: →+2HClaq CO2−3aq + H2CO3aq 2Cl−aq What kind of reaction is this? precipitation acid-base redox If you said this was a precipitation reaction, enter the chemical formula of the precipitate. If you said this was an acid-base reaction, enter the chemical formula of the reactant that is acting as the base. If you said this was a redox reaction, enter the chemical symbol of the element that is oxidized. Calculate the mass percent of CaCO3 in the sample. Be sure your answer has the correct number of significant digits. %For a precipitation reaction to be useful in a gravimetric analysis, the product of the reaction must be insoluble. Is Kc > 1, < 1, or ≈ 1 for a useful precipitation reaction?
- The fertilizer ammonium sulfate, (NH4)2SO4, is prepared by the rxn of ammonia with sulfuric acid. Compile the balanced molecular chemical eqn of this reaction: This reaction type is: A Combination B single displacement C Double displacement D oxidation-reductionCopper (II) chloride, CuCl2(aq) solution is reacted with aluminum hydroxide, Al(OH)3(aq) solution. Write a BALANCED EQUATION for this reaction.For the chemical reaction HCN(aq)+KOH(aq)⟶H2O(l)+KCN(aq)HCN(aq)+KOH(aq)⟶H2O(l)+KCN(aq) write the net ionic equation, including the phases.
- A solution of citric acid (H₃C₆H₅O₇) with a known concentration of 0.200 M H₃C₆H₅O₇ is titrated with a 0.750 M NaOH solution. How many mL of NaOH are required to reach the third equivalence point with a starting volume of 25.0 mL H₃C₆H₅O₇ , according to the following balanced chemical equation: H₃C₆H₅O₇ + 3 NaOH → Na₃C₆H₅O₇ + 3 H₂OThe Kc for the reaction 2A (aq) ⇌ 3 B (aq) is 0.085. What is Kc for the reaction 3 B (aq) ⇌ 2A (aq)?How could you distinguish solution of sodium chloride (table salt) from a solution of sodium iodide (a poison) using an AgNO3 solution? Show reactions. Express your answers as balanced chemical equations. Identify all of the phases in your answer. Seperate the two equations by a comma. hint: Begin each with AgNO3(aq) and add to it either NaCl(aq) or NaI(aq)

